Advanced Synthesis of Bisphenol F Tetraacid Dianhydride for High-Performance Polyimides
The landscape of high-performance polymer chemistry is constantly evolving, driven by the demand for materials that can withstand increasingly rigorous operational environments. A pivotal advancement in this sector is detailed in Chinese Patent CN102276563A, which discloses a novel and efficient preparation method for bisphenol F tetraacid dianhydride. This specific class of monomers serves as the critical building block for next-generation bis-ether type polyimides, offering a substantial upgrade over traditional bisphenol A-based systems. The patent highlights the synthesis of three distinct isomeric forms, designated as formula (I), formula (II), and formula (III), each providing unique structural nuances that influence the final polymer's thermal and mechanical properties. By leveraging a nucleophilic substitution strategy between bisphenol F and specifically substituted phthalimides, this technology addresses long-standing challenges regarding yield, purity, and process complexity. For R&D teams focused on electronic materials, understanding this synthetic pathway is essential for developing resins with enhanced solvent resistance and dimensional stability.
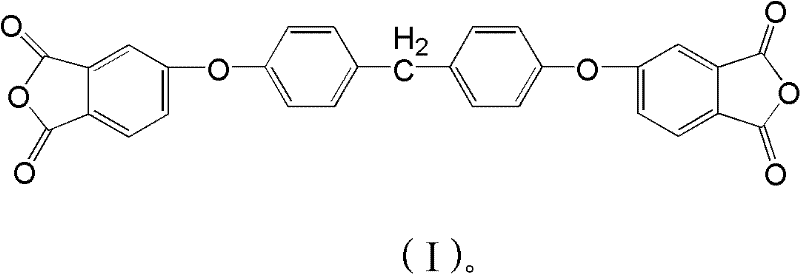
The transition from legacy manufacturing techniques to this innovative approach represents a significant leap in process engineering for specialty chemical intermediates. Conventional methods for producing bis-ether dianhydrides often rely on bisphenol A as the core scaffold. While historically prevalent, polyimides derived from bisphenol A frequently suffer from inadequate solvent resistance, limiting their application in advanced aerospace and microelectronics sectors where exposure to aggressive chemicals is common. Furthermore, prior art methods, such as those utilizing 4-nitrophthalonitrile, are plagued by high raw material costs and suboptimal yields, creating bottlenecks in supply chains. The existing technologies often require harsh reaction conditions and complex downstream purification to remove stubborn by-products, which escalates both the environmental footprint and the overall production cost. These limitations have necessitated a search for more robust and economically viable synthetic routes that do not compromise on the quality of the final monomer.
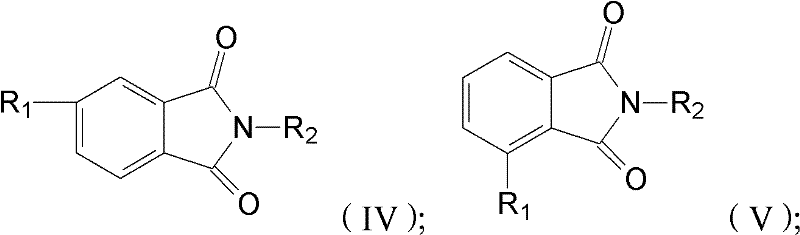
In stark contrast, the novel approach outlined in the patent utilizes bisphenol F and substituted phthalimides (structures IV and V) to achieve a streamlined synthesis with superior outcomes. This method capitalizes on the inherent reactivity of halogenated or nitro-substituted phthalimides in the presence of a base, facilitating a clean nucleophilic attack on the phenolic hydroxyl groups of bisphenol F. The result is a highly efficient formation of N,N-disubstituted bisphenol F bisphthalimide intermediates, which are then converted to the target dianhydride through straightforward hydrolysis and dehydration steps. This pathway eliminates the need for expensive catalysts and reduces the generation of hazardous waste, aligning with modern green chemistry principles. The ability to selectively produce specific isomers or controlled mixtures allows manufacturers to tailor the properties of the resulting polyimide, offering a level of customization that was previously difficult to achieve at scale.
Mechanistic Insights into Base-Catalyzed Nucleophilic Substitution
The core of this synthesis lies in the precise control of the nucleophilic substitution reaction, where the phenoxide anion of bisphenol F attacks the electron-deficient aromatic ring of the phthalimide derivative. The choice of solvent plays a critical role in this mechanism; polar aprotic solvents such as dimethyl sulfoxide (DMSO), sulfolane, or N-methylpyrrolidone are employed to stabilize the transition state and enhance the nucleophilicity of the phenoxide ion without solvating it too strongly. The reaction temperature, typically maintained between 100°C and 200°C, provides the necessary activation energy to overcome the steric hindrance associated with the bulky bisphenol F molecule. Additionally, the use of a water-carrying solvent like toluene or xylene during the initial salt formation step ensures the complete removal of water, driving the equilibrium towards the formation of the reactive phenoxide species. This meticulous control over reaction parameters minimizes side reactions, such as ether cleavage or polymerization, ensuring a high-purity intermediate.
Impurity control is further managed through the strategic selection of the leaving group on the phthalimide ring, which can be either a halogen (chlorine or bromine) or a nitro group. The subsequent hydrolysis step converts the imide rings into carboxylic acid salts under alkaline conditions, followed by acidification to precipitate the tetra-acid. The final dehydration step, often performed in a high-boiling solvent like o-dichlorobenzene or via acetic anhydride treatment, cyclizes the tetra-acid back into the dianhydride structure. This multi-stage process is designed to be robust against variations in raw material quality, as the recrystallization steps effectively purge residual salts and organic impurities. For quality assurance teams, this means the final product consistently meets stringent specifications for ash content and color, which are vital for optical and electronic applications where even trace contaminants can cause device failure.
How to Synthesize Bisphenol F Tetraacid Dianhydride Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and thermal management to maximize yield and safety. The process begins with the formation of the bisphenol F salt, followed by the addition of the phthalimide derivative under an inert atmosphere to prevent oxidation. Detailed operational parameters, including specific molar ratios and heating profiles, are critical for reproducibility. The following guide outlines the standardized procedure derived from the patent examples, serving as a foundational reference for process engineers looking to adapt this technology for pilot or commercial plants.
- React bisphenol F with substituted phthalimides (formula IV/V) in a polar aprotic solvent with a base at 100-200°C to form N,N-disubstituted bisphenol F bisphthalimide.
- Hydrolyze the intermediate bisphthalimide in an alkaline aqueous solution under reflux conditions to generate the tetra-acid salt.
- Acidify the solution to precipitate the tetra-acid, followed by dehydration or recrystallization to obtain the final dianhydride product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers compelling economic benefits driven by the accessibility of raw materials and the simplicity of the unit operations involved. Bisphenol F is a commodity chemical with a stable global supply chain, unlike some specialized nitrile precursors used in older methods which can be subject to volatile pricing and availability issues. The elimination of transition metal catalysts removes the need for expensive scavenging steps and complex wastewater treatment protocols associated with heavy metal disposal. This simplification of the downstream processing directly translates to reduced operational expenditures and a lower total cost of ownership for the manufacturing facility. Furthermore, the high yield reported in the patent examples suggests that less raw material is wasted per kilogram of finished product, enhancing overall resource efficiency.
Supply chain reliability is significantly enhanced by the robustness of the reaction conditions, which tolerate a degree of variability without compromising product integrity. The use of common industrial solvents and bases means that sourcing logistics are straightforward, reducing the risk of production stoppages due to specialized chemical shortages. The process is inherently scalable, moving seamlessly from laboratory glassware to large-scale reactors without requiring fundamental changes to the chemistry. This scalability ensures that suppliers can respond rapidly to fluctuations in market demand, reducing lead times for high-purity polyimide monomers. For supply chain heads, this predictability is invaluable for maintaining continuous production schedules in downstream polymerization facilities.
Environmental compliance and sustainability are also major advantages, as the process generates fewer by-products and avoids the use of toxic reagents. The ability to recycle solvents like toluene and xylene further reduces the environmental footprint and waste disposal costs. The final product's superior performance characteristics mean that less material may be required in end-use applications to achieve the same level of protection, contributing to a more sustainable lifecycle for the electronic components. These factors collectively position this technology as a future-proof solution for manufacturers aiming to meet increasingly strict regulatory standards while maintaining competitiveness in the global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of bisphenol F tetraacid dianhydride. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, offering clarity for stakeholders evaluating this technology for integration into their supply chains.
Q: What are the advantages of Bisphenol F dianhydride over Bisphenol A dianhydride?
A: Bisphenol F-based polyimides exhibit superior solvent resistance and processability compared to Bisphenol A analogues, making them ideal for harsh chemical environments in electronics.
Q: Is the synthesis process suitable for large-scale industrial production?
A: Yes, the patent describes a method using readily available raw materials and simple post-treatment processes like recrystallization, which facilitates commercial scale-up without complex purification steps.
Q: What specific isomers can be produced using this method?
A: The method allows for the selective synthesis of 4,4'-, 3,3'-, or mixed isomers of bisphenol F tetraacid dianhydride by choosing specific phthalimide starting materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bisphenol F Tetraacid Dianhydride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance monomers play in the development of advanced electronic materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of bisphenol F tetraacid dianhydride meets the exacting standards required for aerospace and semiconductor applications. Our commitment to quality ensures that your downstream polyimide synthesis proceeds without interruption or yield loss due to monomer impurities.
We invite you to collaborate with us to optimize your material sourcing strategy. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term growth and innovation goals in the polyimide sector.
