Advanced Synthesis of 4,4'-Bis(2,4-Dinitrophenoxy)Diphenyl Sulfone for High-Performance Polyimide Manufacturing
The global demand for high-performance polymers, particularly aromatic polyimides used in aerospace, microelectronics, and liquid crystal display (LCD) technologies, has necessitated the development of robust synthetic routes for their key monomers. Patent CN101230026A introduces a groundbreaking preparation method for 4,4'-bis(2,4-dinitrophenoxy)diphenyl sulfone (44BDNPODPS), a critical precursor for highly branched aromatic polyimides. This technical insight report analyzes the proprietary synthesis strategy detailed in the patent, highlighting its potential to revolutionize the supply chain for electronic chemical manufacturing. By leveraging a novel nucleophilic aromatic substitution pathway under atmospheric pressure, this method addresses long-standing challenges regarding purity, yield, and environmental impact, positioning it as a vital technology for any reliable polyimide intermediate supplier aiming to serve the high-end materials market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations described in CN101230026A, the synthesis of complex bis-ether sulfones like 44BDNPODPS lacked standardized, industrially viable protocols, with no significant published literature detailing efficient production methods. Traditional approaches to similar aromatic ether syntheses often suffer from incomplete conversion due to the steric hindrance of bulky sulfone groups and the varying reactivity of halogenated precursors. Furthermore, conventional processes frequently rely on single-phase polar aprotic solvents which, while effective for solubility, make product isolation and solvent recovery energetically expensive and environmentally burdensome. The inability to effectively remove water generated during the phenol deprotonation step often shifts the equilibrium backward, leading to lower yields and requiring excessive amounts of expensive starting materials. Additionally, many historical methods involve harsh conditions or corrosive catalysts that degrade equipment integrity, increasing capital expenditure and maintenance downtime for chemical plants.
The Novel Approach
The patented methodology overcomes these barriers through a sophisticated biphasic solvent strategy combined with azeotropic water removal. By employing a specific mixture of water-insoluble organic solvents (such as toluene or xylene) and strong polar aprotic solvents (like DMF or NMP), the process creates an optimal environment for the nucleophilic attack while facilitating the continuous removal of reaction by-products. This dual-solvent system not only enhances the reaction kinetics by maintaining the reactants in solution but also allows for the efficient recycling of the bulk solvent phase, drastically simplifying the downstream processing. The use of readily available inorganic bases, such as potassium carbonate or sodium hydroxide, eliminates the need for expensive phase-transfer catalysts or hazardous metallic reagents. This approach ensures that the commercial scale-up of complex polymer additives becomes economically feasible, offering a streamlined pathway from laboratory bench to multi-ton production without compromising on the structural integrity of the sensitive nitro-functionalized product.
Mechanistic Insights into Nucleophilic Aromatic Substitution
The core chemical transformation driving this synthesis is a classic nucleophilic aromatic substitution ($S_NAr$), facilitated by the strong electron-withdrawing nature of the nitro groups on the benzene ring. In this mechanism, the 4,4'-dihydroxydiphenyl sulfone (BPS) acts as the nucleophile after being deprotonated by the inorganic base to form a phenoxide anion. This anion attacks the electron-deficient carbon atom bonded to the halogen on the 2,4-dinitrohalobenzene ring. The presence of two nitro groups, positioned ortho and para to the leaving halogen group, significantly stabilizes the negatively charged Meisenheimer complex intermediate, thereby lowering the activation energy required for the substitution. This electronic activation allows the reaction to proceed efficiently even with less reactive halogens like chlorine or bromine, providing flexibility in raw material sourcing. The subsequent elimination of the halide ion restores aromaticity, resulting in the formation of the stable ether linkage that characterizes the final 44BDNPODPS structure.
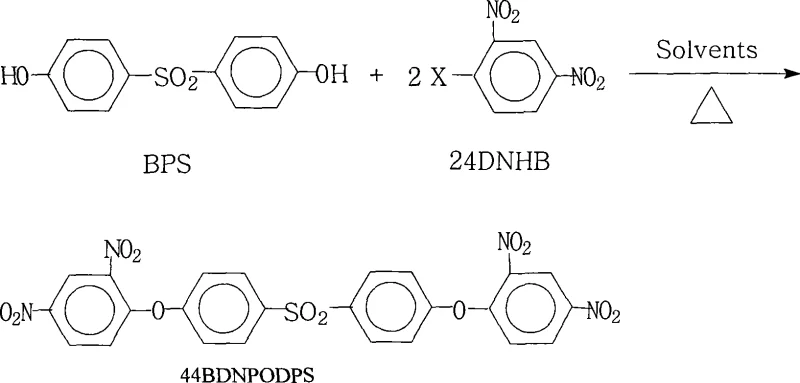
Controlling impurities in this reaction is paramount for ensuring the thermal stability of the resulting polyimide. The patent specifies a molar ratio of 1.0:2.0 to 2.2 for the diphenol to the dinitrohalobenzene, ensuring a slight excess of the electrophile to drive the reaction to completion and minimize the presence of mono-substituted intermediates. The choice of base and its molar ratio (0.10 to 8.00 relative to the diphenol) is critical; insufficient base leads to incomplete deprotonation, while excessive base can promote side reactions or complicate the workup. The heating reflux with water separation (80°C to 200°C) serves a dual purpose: it provides the thermal energy necessary to overcome the kinetic barrier of the substitution and continuously removes water formed during the salt formation, preventing the hydrolysis of the sensitive nitro groups or the reverse reaction. This precise control over reaction parameters ensures a clean impurity profile, which is essential for high-purity OLED material and advanced polyimide applications where trace ionic contaminants can degrade electrical performance.
How to Synthesize 4,4'-Bis(2,4-Dinitrophenoxy)Diphenyl Sulfone Efficiently
The synthesis protocol outlined in the patent offers a reproducible framework for producing 44BDNPODPS with consistent quality. The process begins with the precise charging of 4,4'-dihydroxydiphenyl sulfone and the selected 2,4-dinitrohalobenzene into a reactor equipped with a water separator. Following the addition of the base and the optimized solvent mixture, the system is heated to reflux, allowing the reaction to proceed for a duration of 6 to 18 hours depending on the specific halogen reactivity. Detailed standard operating procedures regarding temperature ramping, solvent ratios, and crystallization conditions are essential for maximizing yield. For a comprehensive breakdown of the specific experimental steps and parameter optimization, please refer to the standardized synthesis guide below.
- Charge the reactor with 4,4'-dihydroxydiphenyl sulfone and 2,4-dinitrohalobenzene in a molar ratio of 1.0: 2.0-2.2, along with a carbonate or hydroxide base.
- Add a mixed solvent system comprising a water-insoluble solvent (e.g., toluene) and a polar aprotic solvent (e.g., DMF), then heat to reflux (80-200°C) for 6-18 hours with water separation.
- Concentrate the reaction mixture, cool, precipitate the product with water, and filter to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible strategic advantages beyond mere technical feasibility. The process is designed with industrial scalability in mind, utilizing common chemical feedstocks and standard reactor configurations that do not require exotic metallurgy or high-pressure vessels. This inherent simplicity translates directly into reduced capital investment and lower operational risks, making it an attractive option for expanding production capacity in response to market demand. Furthermore, the emphasis on solvent recovery and recycling aligns perfectly with modern sustainability goals, potentially lowering waste disposal costs and enhancing the company's environmental, social, and governance (ESG) profile. By securing a supply of intermediates produced via this efficient method, organizations can mitigate the risks associated with volatile raw material markets and ensure a steady flow of critical components for their downstream polymer manufacturing.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the ability to recover and reuse the organic solvent system. Unlike traditional methods where solvents are often consumed or difficult to separate from the product, this biphasic approach allows for the distillation and recycling of the bulk solvent, leading to substantial reductions in raw material procurement costs. Additionally, the use of inexpensive inorganic bases like potassium carbonate instead of costly organometallic catalysts further drives down the variable cost per kilogram. The high yield reported in the patent examples minimizes the loss of valuable starting materials, ensuring that the overall cost of goods sold (COGS) remains competitive even when scaling to multi-ton quantities. These factors collectively contribute to a more lean and cost-effective manufacturing model.
- Enhanced Supply Chain Reliability: The reliance on widely available commodity chemicals such as toluene, xylene, and various carbonates ensures that the supply chain is resilient against disruptions. Since the process does not depend on rare earth metals or specialized reagents that might face geopolitical supply constraints, production continuity is greatly improved. The robustness of the reaction conditions, operating at atmospheric pressure and moderate temperatures, also means that the process can be easily transferred between different manufacturing sites or contract manufacturing organizations (CMOs) without extensive requalification. This flexibility allows supply chain heads to diversify their supplier base and reduce lead times for high-purity aromatic sulfone derivatives, ensuring that production schedules for end-user applications like LCD panels remain uninterrupted.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this method offers a cleaner alternative to older synthetic routes. The absence of corrosive by-products and the minimization of waste generation simplify the effluent treatment process, reducing the burden on wastewater treatment facilities. The ability to operate under atmospheric pressure eliminates the safety hazards associated with high-pressure reactors, lowering insurance premiums and safety compliance costs. As global regulations on volatile organic compounds (VOCs) and industrial waste become stricter, adopting a process that inherently supports solvent recycling and waste reduction future-proofs the manufacturing operation. This alignment with environmental standards facilitates smoother permitting processes and enhances the brand reputation of the manufacturer as a responsible producer of specialty chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 4,4'-bis(2,4-dinitrophenoxy)diphenyl sulfone. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the process capabilities and limitations. Understanding these details is crucial for R&D teams evaluating the material for new formulations and for procurement specialists negotiating supply agreements.
Q: What are the critical reaction conditions for synthesizing 44BDNPODPS?
A: The synthesis requires a molar ratio of 1.0:2.0-2.2 between the diphenol and the dinitrohalobenzene, utilizing a mixed solvent system (water-insoluble + polar aprotic) at temperatures between 80°C and 200°C for 6 to 18 hours.
Q: How does this method improve environmental compliance compared to traditional routes?
A: This method utilizes a solvent system that allows for convenient recovery and repeated recycling, significantly reducing waste generation (three wastes) and eliminating the need for corrosive reagents often found in older sulfonation or nitration processes.
Q: What is the expected purity and yield for this polyimide intermediate?
A: According to patent data, the process consistently achieves high purity levels (up to 99.6%) and substantial yields (ranging from 71% to over 93% depending on the specific halogen and solvent combination used).
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Bis(2,4-Dinitrophenoxy)Diphenyl Sulfone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation electronic materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of the global polyimide market. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs which utilize advanced analytical techniques to verify every batch. By leveraging the efficient synthesis strategies outlined in patents like CN101230026A, we offer a supply solution that balances technical excellence with commercial reliability, making us the preferred partner for industry leaders.
We invite you to collaborate with us to optimize your supply chain for high-performance polymers. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your innovation goals and drive value for your organization.
