Advanced Synthesis of 1,6-Bis(2,6-Dinitro-4-Trifluoromethylphenoxy)Naphthalene for High-Performance Polyimides
The global demand for high-performance aromatic polyimides in the aerospace and electronic display sectors has necessitated the development of robust synthetic routes for their key monomeric precursors. Patent CN101328126A, filed in late 2008, discloses a highly efficient preparation method for 1,6-bis(2,6-dinitro-4-trifluoromethylphenoxy)naphthalene, a critical intermediate used to synthesize highly branched aromatic polyimide monomers. This specific chemical architecture imparts superior thermal stability, mechanical strength, and electrical insulation properties to the final polymer matrix, making it indispensable for applications ranging from liquid crystal displays (LCDs) to radiation-hardened components in space exploration. The patented process addresses significant gaps in the prior art by offering a scalable, atmospheric pressure route that achieves exceptional purity levels exceeding 99.4% without the need for complex chromatographic purification steps.
For R&D directors and process chemists evaluating new supply chains, the significance of this technology lies in its ability to consistently produce high-purity electronic chemical intermediates with minimal impurity profiles. The method leverages a nucleophilic aromatic substitution mechanism that is both chemically elegant and industrially pragmatic. By optimizing the molar ratios of reactants and utilizing a dual-solvent system, the process ensures high conversion rates while maintaining operational simplicity. This technical breakthrough provides a reliable foundation for the commercial scale-up of complex polyimide monomers, ensuring that downstream manufacturers can access materials that meet the rigorous specifications required for next-generation optoelectronic devices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the disclosure of this invention, the specific synthesis of 1,6-bis(2,6-dinitro-4-trifluoromethylphenoxy)naphthalene lacked documented industrial protocols, forcing manufacturers to rely on ad-hoc laboratory methods that were often inefficient and difficult to scale. Conventional approaches to synthesizing similar diaryl ether structures frequently suffered from low yields due to incomplete conversion or the formation of stubborn by-products that were difficult to separate. Traditional methods often required harsh reaction conditions, such as extremely high temperatures or the use of expensive transition metal catalysts, which introduced heavy metal contamination risks that are unacceptable in high-end electronic applications. Furthermore, many legacy processes utilized single-solvent systems that made water removal difficult, leading to equilibrium limitations that stalled the reaction progress and resulted in significant amounts of unreacted starting materials remaining in the final product mixture.
The Novel Approach
The novel approach detailed in the patent overcomes these historical barriers through a sophisticated yet simple biphasic solvent strategy combined with precise stoichiometric control. By employing a mixture of a water-insoluble organic solvent, such as toluene or benzene, and a strong polar aprotic solvent like N-methyl-2-pyrrolidone (NMP) or dimethylformamide (DMF), the reaction system facilitates effective phase transfer and solubility for both the organic salts and the neutral reactants. This dual-solvent environment allows for efficient azeotropic removal of water generated during the salt formation and substitution steps, driving the equilibrium towards the desired product. The result is a streamlined process that operates under normal atmospheric pressure, eliminating the need for expensive high-pressure reactors while delivering yields that consistently surpass 90% in optimized examples, thereby establishing a new benchmark for cost reduction in electronic chemical manufacturing.
Mechanistic Insights into Nucleophilic Aromatic Substitution
The core chemical transformation driving this synthesis is a classic nucleophilic aromatic substitution (SnAr) reaction, facilitated by the strong electron-withdrawing nature of the substituents on the benzene ring. In this mechanism, the 1,6-dihydroxynaphthalene acts as the nucleophile after being deprotonated by a base such as potassium carbonate or sodium hydroxide to form the corresponding naphthoxide anion. This anion is highly reactive and attacks the electron-deficient aromatic ring of the 2,6-dinitro-4-trifluoromethyl halobenzene. The presence of two nitro groups and a trifluoromethyl group ortho and para to the leaving halogen atom significantly activates the ring towards nucleophilic attack by stabilizing the negatively charged Meisenheimer complex intermediate. This electronic activation is crucial, as it allows the reaction to proceed smoothly even with less reactive halogens like chlorine or fluorine, providing flexibility in raw material sourcing.
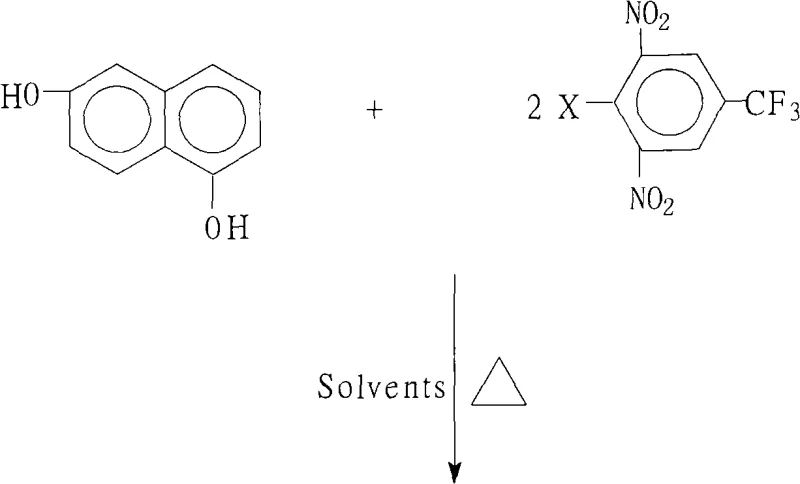
Following the formation of the intermediate complex, the expulsion of the halide leaving group restores aromaticity, resulting in the formation of the stable ether linkage. The choice of base and the molar ratio of the salt-forming agent to the dihydroxynaphthalene (ranging from 0.80:1.00 to 8.00:1.00) plays a pivotal role in controlling the reaction kinetics and minimizing side reactions. Excess base ensures complete deprotonation of the diol, while the specific solvent mixture helps to solvate the cation, increasing the nucleophilicity of the anion. This mechanistic understanding allows process engineers to fine-tune reaction parameters, such as temperature (80°C to 200°C) and time (6 to 18 hours), to maximize throughput while maintaining the stringent purity specifications required for high-purity aromatic organic compounds used in sensitive electronic applications.
How to Synthesize 1,6-Bis(2,6-Dinitro-4-Trifluoromethylphenoxy)Naphthalene Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for manufacturing this valuable intermediate with high reproducibility. The process begins by charging a reactor with the defined molar quantities of 1,6-dihydroxynaphthalene and the selected halobenzene derivative, followed by the addition of the base and the mixed solvent system. The mixture is then subjected to heating under reflux conditions with continuous water separation, typically using a Dean-Stark apparatus or similar setup, to drive the reaction to completion. Detailed standardized synthesis steps, including specific workup procedures like concentration, cooling, and water precipitation, are essential for achieving the reported purity levels of over 99%.
- Charge the reactor with 1,6-dihydroxynaphthalene and 2,6-dinitro-4-trifluoromethyl halobenzene in a molar ratio of 1.0: 2.0-2.2, along with a salt-forming agent like potassium carbonate.
- Add a mixed solvent system comprising a water-insoluble organic solvent (e.g., toluene) and a polar aprotic solvent (e.g., DMF or NMP), then heat to reflux (80°C-200°C) for 6-18 hours with water separation.
- Concentrate the reaction mixture, cool, add water to precipitate the solid product, then filter, wash with hot water, and dry to obtain crystals with >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers substantial strategic advantages by simplifying the manufacturing landscape and reducing dependency on exotic reagents. The reliance on commodity chemicals such as toluene, benzene, and common inorganic bases like potassium carbonate means that raw material sourcing is robust and less susceptible to market volatility compared to processes requiring specialized catalysts. Furthermore, the ability to recover and recycle the organic solvents repeatedly creates a closed-loop system that drastically lowers the variable costs associated with solvent consumption. This efficiency translates directly into a more competitive pricing structure for the final intermediate, allowing downstream polymer manufacturers to optimize their own cost structures without compromising on material quality.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of atmospheric pressure reactors significantly lower capital expenditure (CAPEX) and operational expenditure (OPEX). By avoiding the need for high-pressure equipment and complex catalyst removal steps, the process reduces energy consumption and labor hours, leading to substantial cost savings in the overall production budget. Additionally, the high yield minimizes raw material waste, ensuring that every kilogram of input generates maximum output value.
- Enhanced Supply Chain Reliability: The use of widely available starting materials like 1,6-dihydroxynaphthalene and various halobenzenes ensures a stable supply chain that is not vulnerable to single-source bottlenecks. The flexibility to use different halogens (F, Cl, Br, I) allows procurement teams to switch suppliers based on availability and price without altering the core process, thereby enhancing supply continuity. This adaptability is crucial for maintaining consistent production schedules in the face of global logistical challenges.
- Scalability and Environmental Compliance: The process is inherently scalable due to its simple operation and lack of corrosive by-products, making it suitable for large-scale industrial production from 100 kgs to multi-ton batches. The reduced generation of three wastes (waste water, gas, and solids) and the ability to recycle solvents align with increasingly strict environmental regulations, reducing the burden on waste treatment facilities and lowering compliance costs. This environmental friendliness enhances the corporate social responsibility profile of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized intermediate. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on process capabilities and material properties. Understanding these details is vital for stakeholders assessing the feasibility of integrating this material into their existing supply chains.
Q: What are the critical reaction conditions for synthesizing this polyimide intermediate?
A: The process requires a mixed solvent system of water-insoluble organics (like toluene) and polar aprotic solvents (like DMF), heated to reflux between 80°C and 200°C for 6 to 18 hours to ensure complete conversion.
Q: Which halobenzene derivatives are suitable for this nucleophilic substitution?
A: The patent specifies that 2,6-dinitro-4-trifluoromethyl halobenzenes where the halogen is fluorine, chlorine, bromine, or iodine can all be effectively utilized as electrophiles in this reaction.
Q: How does this method improve environmental compliance compared to traditional routes?
A: This method utilizes recoverable organic solvents and avoids corrosive reagents or transition metal catalysts, significantly reducing hazardous waste generation and simplifying the purification workflow.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,6-Bis(2,6-Dinitro-4-Trifluoromethylphenoxy)Naphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of advanced polyimide materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of global electronics and aerospace manufacturers. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of 1,6-bis(2,6-dinitro-4-trifluoromethylphenoxy)naphthalene meets the exacting standards necessary for high-performance applications. Our commitment to quality assurance guarantees that our customers receive materials that perform consistently in their final polymer formulations.
We invite potential partners to contact our technical procurement team to discuss how our optimized synthesis routes can support your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our efficient manufacturing processes can reduce your overall material costs. We encourage you to reach out for specific COA data and route feasibility assessments to ensure that our solutions align perfectly with your R&D and production goals, fostering a long-term partnership built on reliability and technical excellence.
