Scalable Synthesis of 2,4-Bis(2,6-Dinitro-4-Trifluoromethylphenoxy)Toluene for High-Performance Polyimides
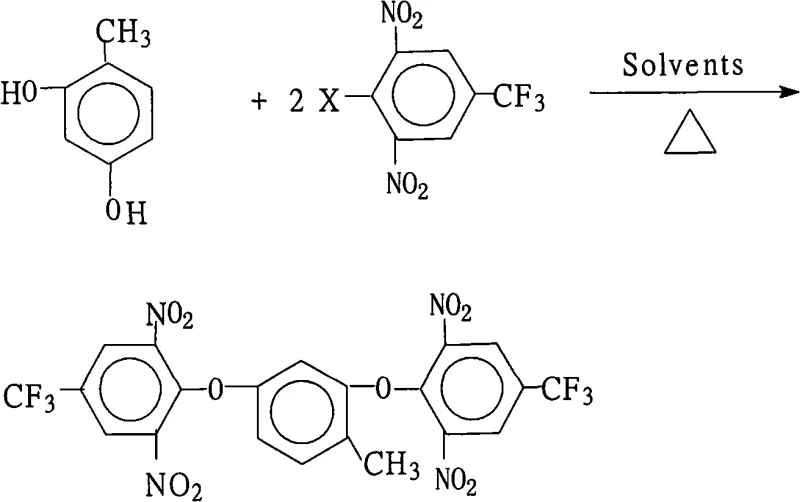
The global demand for high-performance electronic materials, particularly those capable of withstanding extreme thermal and chemical environments, has necessitated the development of advanced fluorinated polyimide precursors. Patent CN101302164A introduces a groundbreaking preparation method for 2,4-bis(2,6-dinitro-4-trifluoromethylphenoxy)toluene, a pivotal intermediate in the synthesis of highly branched fluorine-containing aromatic polyimides. This specific chemical architecture is indispensable for next-generation liquid crystal displays (LCDs) and microelectronics, where traditional materials fail to meet rigorous insulation and thermal stability standards. The disclosed technology represents a significant leap forward in fine chemical synthesis, offering a robust pathway to produce this complex aromatic ether with exceptional purity and yield. By leveraging a sophisticated nucleophilic aromatic substitution strategy, the process addresses historical challenges associated with poly-fluorinated compound synthesis, providing a reliable foundation for the commercial scale-up of complex electronic chemical intermediates. For R&D directors and procurement specialists alike, understanding the nuances of this patented route is essential for securing a stable supply chain of high-value electronic materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of multi-substituted aromatic ethers containing sensitive nitro and trifluoromethyl groups has been plagued by significant technical hurdles that impede efficient cost reduction in electronic chemical manufacturing. Conventional nucleophilic substitution reactions often suffer from incomplete conversion due to the steric hindrance imposed by the ortho-nitro groups and the electron-withdrawing nature of the trifluoromethyl moiety, which can paradoxically deactivate certain positions or lead to side reactions. Furthermore, traditional methods frequently rely on stoichiometric amounts of expensive phase transfer catalysts or harsh conditions that degrade the sensitive nitro functionality, resulting in a complex impurity profile that is difficult to purify. The removal of residual solvents and byproducts in these older methodologies typically requires energy-intensive distillation or chromatographic separation, which drastically increases production costs and environmental waste. Additionally, the lack of effective water removal mechanisms in standard reflux setups often limits the reaction equilibrium, preventing the high yields necessary for viable commercial production. These inefficiencies create bottlenecks in the supply chain, leading to inconsistent quality and extended lead times for downstream polymer manufacturers who require high-purity OLED material precursors and polyimide monomers.
The Novel Approach
The methodology outlined in patent CN101302164A fundamentally transforms the synthesis landscape by introducing a dual-solvent system coupled with azeotropic water separation, effectively overcoming the thermodynamic limitations of prior art. By employing a mixture of a water-insoluble organic solvent, such as toluene or benzene, and a strong polar aprotic solvent like N,N-dimethylformamide (DMF) or N-methyl-2-pyrrolidone (NMP), the process creates an optimal environment for the nucleophilic attack while facilitating the continuous removal of water generated during the reaction. This strategic setup drives the equilibrium towards the product side according to Le Chatelier's principle, ensuring near-quantitative conversion of the starting materials without the need for excessive reagent loading. The use of inorganic bases like potassium carbonate not only activates the phenolic hydroxyl groups but also acts as an acid scavenger, neutralizing the hydrogen halide byproduct and preventing acid-catalyzed decomposition of the nitro groups. This innovative approach simplifies the post-reaction workup significantly, as the product can be precipitated simply by adding water to the concentrated reaction mixture, eliminating the need for complex purification steps. Consequently, this method offers a streamlined, environmentally friendly route that aligns perfectly with the goals of a reliable agrochemical intermediate supplier or electronic chemical provider seeking to minimize waste and maximize efficiency.
Mechanistic Insights into Nucleophilic Aromatic Substitution
The core chemical transformation in this synthesis is a classic yet highly optimized nucleophilic aromatic substitution (SnAr) reaction, where the electron-deficient aromatic ring of the 2,6-dinitro-4-trifluoromethyl halobenzene serves as the electrophile. The presence of two strongly electron-withdrawing nitro groups at the ortho positions relative to the leaving halogen atom dramatically activates the ring towards nucleophilic attack by stabilizing the negatively charged Meisenheimer complex intermediate. Simultaneously, the para-trifluoromethyl group further enhances this electrophilicity through its powerful inductive effect, making the carbon-halogen bond highly susceptible to cleavage by the phenoxide nucleophile generated from 2,4-dihydroxytoluene. The reaction proceeds through a concerted addition-elimination mechanism, where the phenoxide oxygen attacks the ipso-carbon, forming a resonance-stabilized anionic sigma complex, followed by the rapid expulsion of the halide leaving group. The choice of solvent is critical here; the polar aprotic component solvates the cation of the base, leaving the phenoxide anion "naked" and highly reactive, while the water-immiscible component allows for the physical separation of the water byproduct. This precise control over the reaction microenvironment ensures that the substitution occurs selectively at the desired positions without affecting the sensitive nitro groups, thereby maintaining the structural integrity required for subsequent polymerization into high-performance polyimides.
Controlling the impurity profile in such a complex multi-step synthesis is paramount for ensuring the final polymer exhibits consistent thermal and mechanical properties. The patented process achieves superior impurity control primarily through the strict regulation of the molar ratio between the dihydroxytoluene and the halobenzene, maintaining a slight excess of the electrophile (1.0:2.0 to 2.2) to ensure complete consumption of the diol and prevent the formation of mono-substituted byproducts. Furthermore, the continuous removal of water via azeotropic distillation prevents the hydrolysis of the halobenzene starting material, a common side reaction that can generate phenolic impurities difficult to separate from the product. The use of specific inorganic bases, such as potassium carbonate or bicarbonate, minimizes the risk of base-catalyzed degradation of the nitro groups, which can occur with stronger hydroxide bases at elevated temperatures. Post-reaction, the precipitation step using water acts as a crude purification stage, where the highly non-polar product crashes out of the polar solvent mixture, leaving behind soluble inorganic salts and polar organic impurities in the mother liquor. This inherent selectivity of the crystallization process, combined with the high conversion rates of the reaction itself, results in a crude product with purity levels often exceeding 99%, reducing the need for further recrystallization and preserving overall yield.
How to Synthesize 2,4-Bis(2,6-Dinitro-4-Trifluoromethylphenoxy)Toluene Efficiently
To achieve the high yields and purity described in the patent, operators must adhere to a precise sequence of mixing and thermal controls that optimize the kinetics of the substitution reaction. The process begins with the careful charging of the reactor with 2,4-dihydroxytoluene and the halobenzene derivative in the specified molar ratio, ensuring homogeneity before the addition of the base and solvent system. The selection of the solvent mixture is critical, with the volume ratio of the water-insoluble solvent to the polar aprotic solvent needing to be balanced to facilitate both solubility of the reactants and efficient water separation during reflux. Heating the mixture to a temperature range of 80°C to 150°C initiates the reaction, and the system must be maintained under reflux with a water separator for a duration of 6 to 18 hours, depending on the specific reactivity of the halogen leaving group employed. Detailed standardized synthesis steps see the guide below.
- Charge the reactor with 2,4-dihydroxytoluene and 2,6-dinitro-4-trifluoromethyl halobenzene in a molar ratio of 1.0: 2.0-2.2, along with a salt-forming agent and a mixed solvent system.
- Heat the reaction mixture to reflux between 80°C and 150°C for 6 to 18 hours, utilizing a water separator to continuously remove generated water and drive the equilibrium forward.
- Concentrate the reaction liquid, cool the system, add water to precipitate the solid product, then filter, wash with hot water, and dry to obtain the final crystalline product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers transformative advantages that directly impact the bottom line and operational resilience of the manufacturing facility. The elimination of expensive transition metal catalysts and the reliance on commodity chemicals like potassium carbonate and common organic solvents drastically reduces the raw material cost base, making the production of this high-value intermediate economically viable on a large scale. The simplicity of the workup procedure, which involves merely concentrating the reaction mixture and precipitating the product with water, significantly lowers labor costs and reduces the turnaround time between batches, thereby enhancing overall plant throughput. Furthermore, the ability to recover and recycle the organic solvents, particularly the expensive polar aprotic solvents like NMP or DMF, contributes to substantial long-term cost savings and aligns with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions. This process stability ensures a consistent supply of material, mitigating the risks associated with batch-to-batch variability that often plague complex fine chemical syntheses.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the strategic elimination of costly purification steps and the maximization of atom economy through high-yield reactions. By avoiding the use of precious metal catalysts, the process removes the need for expensive metal scavenging resins and the associated validation testing required to ensure residual metal levels meet pharmaceutical or electronic grade specifications. The high conversion rates mean that raw material waste is minimized, and the excess reagents can often be recovered or are inexpensive enough to be treated as consumables without impacting margins significantly. Additionally, the energy consumption is optimized by operating at atmospheric pressure and utilizing standard heating mantles or steam jackets, avoiding the need for high-pressure autoclaves or cryogenic cooling systems. These cumulative efficiencies result in a significantly reduced cost of goods sold (COGS), allowing suppliers to offer competitive pricing while maintaining healthy profit margins in the volatile electronic chemicals market.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route translates directly into improved supply chain security for downstream customers who depend on timely deliveries for their own production schedules. Since the starting materials, such as 2,4-dihydroxytoluene and various nitro-halobenzenes, are commercially available bulk chemicals with established supply chains, the risk of raw material shortages is inherently low. The process tolerance to slight variations in reaction conditions means that manufacturing delays due to failed batches are rare, ensuring a steady flow of inventory. Moreover, the scalability of the reaction from gram to kilogram and ton scales has been proven, meaning that suppliers can rapidly ramp up production in response to surges in demand from the LCD or semiconductor sectors without requiring major capital investment in new equipment. This flexibility is crucial for maintaining just-in-time delivery models and building long-term trust with key accounts in the high-tech industry.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this method offers a cleaner alternative to traditional etherification processes, generating minimal hazardous waste and facilitating easier compliance with local environmental protection laws. The solvent system is designed for easy separation and recycling, which not only reduces the volume of chemical waste requiring disposal but also lowers the carbon footprint of the manufacturing process. The absence of corrosive reagents means that standard stainless steel reactors can be used, reducing capital expenditure on specialized Hastelloy equipment and extending the lifespan of the manufacturing assets. The solid product isolation via water precipitation avoids the use of large volumes of anti-solvents that would otherwise need to be distilled and treated, further simplifying the waste management workflow. These factors collectively make the process highly attractive for facilities aiming to achieve green chemistry certifications and sustainable manufacturing status.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized fluorinated intermediate, based on the detailed specifications provided in the patent literature. Understanding these aspects is vital for technical teams evaluating the feasibility of integrating this material into their polymer formulations or electronic device architectures. The answers reflect the consensus on the process capabilities and the material properties derived from the disclosed examples.
Q: What is the primary application of 2,4-bis(2,6-dinitro-4-trifluoromethylphenoxy)toluene?
A: This compound serves as a critical intermediate for synthesizing highly branched fluorine-containing aromatic polyimides, which are essential for high-performance LCDs and electronic materials due to their superior thermal stability and insulation properties.
Q: How does this patented method improve upon conventional etherification processes?
A: The method utilizes a specific dual-solvent system with water separation to drive the reaction to completion, achieving yields over 95% and purity exceeding 99%, while allowing for solvent recycling and minimizing waste generation.
Q: Is the process suitable for large-scale industrial production?
A: Yes, the process operates under atmospheric pressure without corrosive substances, uses readily available raw materials, and features a simple workup procedure involving water precipitation, making it highly scalable and cost-effective for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4-Bis(2,6-Dinitro-4-Trifluoromethylphenoxy)Toluene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the advancement of electronic materials and high-performance polymers. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. Our expertise in handling fluorinated compounds and nitro-aromatics allows us to navigate the complexities of this synthesis with precision, guaranteeing a supply of 2,4-bis(2,6-dinitro-4-trifluoromethylphenoxy)toluene that empowers your innovation in LCD and polyimide technologies.
We invite you to collaborate with us to explore how this advanced synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your project goals, ensuring a partnership that drives mutual growth and technological success in the competitive global market.
