Advanced Synthesis of DNBP-2TF: Technical Breakthroughs for High-Performance Polyimide Manufacturing
Advanced Synthesis of DNBP-2TF: Technical Breakthroughs for High-Performance Polyimide Manufacturing
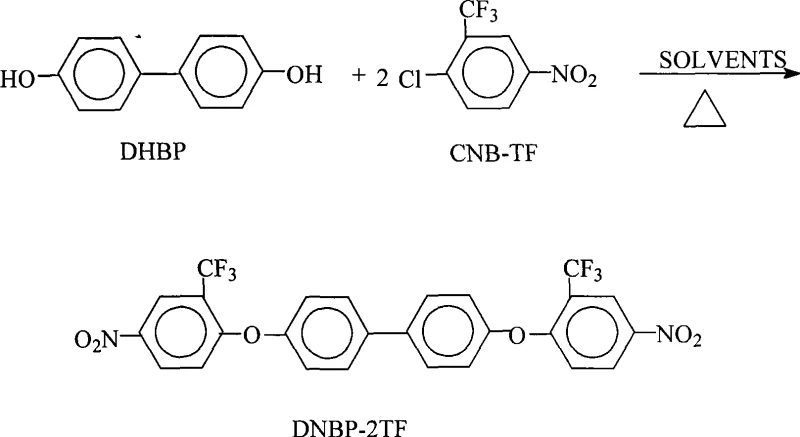
The development of high-performance aromatic polyimides has become a cornerstone in the advancement of modern electronic materials, particularly for applications requiring exceptional thermal stability and dielectric properties. Patent CN101486652A introduces a transformative preparation method for 4,4'-bis(4-nitro-2-trifluoromethylphenoxy)biphenyl, a critical monomer precursor for fluorinated polyimides. This technical disclosure addresses long-standing inefficiencies in the synthesis of complex fluorinated biphenyl derivatives, offering a pathway that significantly enhances both yield and purity while streamlining the operational workflow. For R&D directors and procurement specialists in the electronic chemical sector, understanding the nuances of this azeotropic dehydration technique is essential for optimizing supply chains and reducing the total cost of ownership for high-specification polymer additives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those documented in earlier literature by Yang et al., typically rely on a multi-step purification regime that severely impacts overall process efficiency. In these conventional routes, the crude reaction product is often precipitated by pouring the reaction mixture into vast quantities of water, followed by extensive washing and a mandatory recrystallization step using mixed solvents like DMF and methanol. This approach not only results in substantial material loss, with total yields frequently dropping below 75% due to recrystallization inefficiencies, but also generates excessive volumes of organic-contaminated wastewater. Furthermore, the reliance on multiple solvent systems increases the complexity of solvent recovery, driving up both raw material costs and environmental compliance burdens associated with waste treatment and disposal.
The Novel Approach
The innovative strategy outlined in patent CN101486652A fundamentally reengineers the synthesis workflow by integrating an azeotropic dehydration mechanism directly into the reaction phase. By employing a mixed solvent system comprising a strong polar aprotic solvent and an organic entrainer, the process facilitates the continuous removal of water byproduct as it forms. This thermodynamic shift drives the nucleophilic substitution reaction to near-completion without the need for downstream recrystallization. The result is a direct crystallization of the target molecule from the concentrated mother liquor, achieving yields exceeding 98% and purity levels approaching 99.8%. This elimination of the recrystallization unit operation represents a paradigm shift in manufacturing efficiency, drastically simplifying the process flow and minimizing solvent consumption.
Mechanistic Insights into Azeotropic Nucleophilic Substitution
The core chemical transformation involves a nucleophilic aromatic substitution where the phenoxide anions of 4,4'-dihydroxybiphenyl attack the electron-deficient aromatic ring of 2-chloro-5-nitrotrifluoromethylbenzene. The presence of the nitro group ortho or para to the leaving chlorine atom activates the ring for substitution, while the trifluoromethyl group further enhances electrophilicity through strong inductive effects. However, the generation of water as a byproduct poses a risk of hydrolyzing the reactants or reversing the reaction equilibrium. The patented method mitigates this by utilizing an organic entrainer, such as toluene or xylene, which forms a low-boiling azeotrope with water. As the reaction mixture is heated to temperatures between 115°C and 160°C, the azeotrope distills off, effectively sequestering the water and preventing it from interfering with the reaction kinetics, thus ensuring high conversion rates.
Impurity control in this synthesis is achieved not through post-reaction purification but through precise stoichiometric management and thermal profiling during the reaction itself. By maintaining a molar ratio of the chloronitro compound to the dihydroxybiphenyl between 1.0:2.0 and 1.0:2.6, the process ensures that the dihydroxy species is fully consumed, minimizing the presence of mono-substituted intermediates. Additionally, the choice of base, such as potassium carbonate or sodium carbonate, is optimized to facilitate salt formation without promoting side reactions. The subsequent hot filtration step efficiently removes inorganic salts before the concentration phase, allowing the target product to crystallize in a highly ordered lattice structure upon cooling, which inherently excludes remaining trace impurities without the need for additional solvent washing steps.
How to Synthesize 4,4'-bis(4-nitro-2-trifluoromethylphenoxy)biphenyl Efficiently
The execution of this synthesis requires careful attention to solvent ratios and temperature gradients to maximize the efficacy of the azeotropic water removal. The process begins with the suspension of reactants and base in the mixed solvent system, followed by a controlled heating ramp to initiate reflux. Maintaining the azeotropic distillation for a duration of 6 to 10 hours is critical to ensure complete conversion before the temperature is raised to finalize the reaction. Detailed standardized operating procedures regarding specific solvent volumes, stirring rates, and cooling profiles are essential for reproducibility at scale. For a comprehensive guide on the exact step-by-step protocol validated by experimental data, please refer to the technical instructions below.
- Mix 4,4'-dihydroxybiphenyl and 2-chloro-5-nitrotrifluoromethylbenzene with a base in a polar aprotic solvent and entrainer.
- Heat the mixture to 115°C-160°C to facilitate azeotropic reflux, continuously removing water byproduct to drive equilibrium.
- Filter hot to remove salts, concentrate the mother liquor, and cool to crystallize the high-purity product directly.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers profound advantages for procurement managers and supply chain heads focused on cost reduction in electronic chemical manufacturing. The most significant economic driver is the elimination of the recrystallization step, which traditionally consumes large volumes of high-purity solvents and requires dedicated equipment time. By removing this unit operation, manufacturers can realize substantial cost savings in both raw material procurement and utility consumption. Furthermore, the simplified workflow reduces the labor intensity and equipment footprint required for production, allowing facilities to increase throughput without proportional capital expenditure. This efficiency gain translates directly into a more competitive pricing structure for the final polyimide monomer, enhancing the margin potential for downstream polymer producers.
- Cost Reduction in Manufacturing: The streamlined process eliminates the need for expensive recrystallization solvents like methanol and reduces the overall volume of polar aprotic solvents required per kilogram of product. Since the solvents can be recovered and recycled more efficiently due to the absence of aqueous contamination from precipitation steps, the recurring cost of solvent makeup is drastically lowered. Additionally, the high yield minimizes the cost of goods sold by maximizing the output from every batch of raw materials, ensuring that valuable fluorinated starting materials are not lost to purification waste streams.
- Enhanced Supply Chain Reliability: Simplifying the manufacturing process inherently reduces the number of potential failure points and bottlenecks in the production schedule. Without the dependency on a separate recrystallization phase, lead times for batch completion are significantly shortened, allowing for more responsive fulfillment of customer orders. The robustness of the reaction conditions, which operate at atmospheric pressure and utilize stable, commercially available reagents, ensures consistent supply continuity even during periods of raw material volatility, making it a reliable option for long-term strategic sourcing agreements.
- Scalability and Environmental Compliance: The reduction in wastewater generation is a critical factor for modern chemical manufacturing, where environmental regulations are increasingly stringent. By avoiding the water precipitation step, the facility significantly lowers its effluent load, reducing the costs associated with wastewater treatment and disposal. This environmental benefit not only ensures compliance with local regulations but also aligns with the sustainability goals of major multinational corporations. The process is inherently scalable, as the azeotropic distillation principle functions effectively from pilot plant scales up to multi-ton commercial reactors, facilitating seamless technology transfer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on how this method compares to legacy processes. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this monomer into their existing polymer production lines.
Q: What is the primary advantage of the azeotropic dehydration method in DNBP-2TF synthesis?
A: The use of an organic entrainer allows for the continuous removal of water generated during the reaction. This shifts the chemical equilibrium towards the product side, significantly increasing yield without requiring energy-intensive recrystallization steps.
Q: How does this process improve environmental compliance compared to traditional methods?
A: Traditional methods often require pouring reaction mixtures into large volumes of water for precipitation, generating significant wastewater. This patented process eliminates that step, relying on solvent recovery and direct crystallization, thereby drastically reducing liquid waste discharge.
Q: Can this synthesis route be scaled for industrial production of polyimide monomers?
A: Yes, the process operates at atmospheric pressure with standard equipment and avoids corrosive reagents. The simplified workflow, which removes the recrystallization unit operation, makes it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-bis(4-nitro-2-trifluoromethylphenoxy)biphenyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity monomers play in the performance of next-generation electronic materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of DNBP-2TF supports the thermal and electrical requirements of advanced polyimide applications. Our dedication to technical excellence allows us to navigate the complexities of fluorinated chemistry with precision and reliability.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific application needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic impact of switching to this high-efficiency process. We encourage potential partners to contact us for specific COA data and route feasibility assessments, ensuring that our solutions are perfectly aligned with your quality standards and production timelines. Let us collaborate to drive innovation in the electronic materials sector through superior chemical manufacturing.
