Advanced Catalytic Hydrogenation Technology For High-Purity Fluorinated Diamine Commercial Production
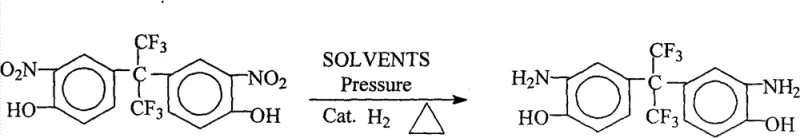
The landscape of advanced material synthesis is undergoing a significant transformation, driven by the urgent need for environmentally sustainable and cost-effective manufacturing processes. Patent CN101492381B introduces a groundbreaking methodology for the preparation of 2,2-bis(3-amino-4-hydroxyphenyl)hexafluoropropane, a critical monomer for high-performance fluorinated polyimides. This technology represents a paradigm shift from traditional, hazardous reduction methods to a cleaner, catalytic hydrogenation approach that aligns with modern green chemistry principles. By leveraging specific pressure and temperature conditions within a high-pressure reactor, this process achieves exceptional yields and purity levels that were previously difficult to attain on an industrial scale. For R&D directors and procurement strategists, understanding the nuances of this patent is essential for securing a reliable fluorinated polyimide monomer supplier capable of meeting the rigorous demands of the electronics and aerospace sectors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluorinated diamines relied heavily on reduction techniques involving hydrazine hydrate or transfer hydrogenation with ammonium formate, both of which present severe operational and environmental challenges. These conventional pathways are plagued by the generation of toxic byproducts, including ammonia emissions and nitrogen-rich wastewater, which necessitate complex and expensive waste treatment infrastructure. Furthermore, the crude products obtained from these methods often exhibit poor color and low purity, typically requiring multiple recrystallization steps using volatile organic solvents like toluene or ethyl acetate. This multi-step purification not only drastically increases production costs but also results in significant material loss, with total yields often stagnating below 60%. The reliance on hazardous reagents and the inability to directly obtain high-purity products make these legacy methods unsuitable for the scalable, continuous manufacturing required by the modern supply chain.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes direct catalytic hydrogenation in an alcoholic solvent system, fundamentally simplifying the production workflow. By employing robust catalysts such as Pd/C or Raney Nickel under controlled hydrogen pressure, the nitro groups are efficiently reduced to amino groups without affecting the sensitive hydroxyl or hexafluoroisopropylidene moieties. A key innovation lies in the isolation strategy: instead of energy-intensive recrystallization, the product is precipitated directly from the mother liquor by the addition of deionized water. This "anti-solvent" precipitation technique not only yields a white, high-purity solid in a single step but also allows for the easy recovery and recycling of the alcohol solvent. This streamlined process eliminates the need for hazardous hydrazine, reduces the three-waste burden, and significantly enhances the overall economic viability of producing high-purity fluorinated diamine for commercial applications.
Mechanistic Insights into Catalytic Hydrogenation of Nitro-Fluorinated Compounds
The core of this technological advancement lies in the precise mechanistic control of the catalytic hydrogenation reaction, where the interaction between the substrate, the catalyst surface, and hydrogen gas is optimized for maximum selectivity. The reaction proceeds via the adsorption of the nitro-fluorinated precursor onto the active sites of the palladium or nickel catalyst, followed by the sequential addition of hydrogen atoms to reduce the nitro group (-NO2) to an amino group (-NH2). Crucially, the reaction conditions—specifically maintaining temperatures between 60°C and 100°C and pressures ranging from 0.3MPa to 3.6MPa—are calibrated to prevent side reactions such as hydrodehalogenation or the reduction of the aromatic ring, which could compromise the structural integrity of the final monomer. The presence of the electron-withdrawing hexafluoroisopropylidene group adds complexity to the electronic environment of the molecule, requiring a catalyst with sufficient activity to overcome steric and electronic barriers without promoting decomposition.
Impurity control is inherently built into this mechanism through the choice of solvent and the precipitation method. In traditional recrystallization, impurities with similar solubility profiles can co-crystallize, leading to persistent contamination. However, in this aqueous precipitation method, the solubility differential between the highly polar amino-product and non-polar or intermediate polarity impurities in the water-alcohol mixture is exploited. As deionized water is added to the alcoholic mother liquor, the dielectric constant of the medium changes, causing the target diamine to crash out of solution while leaving soluble byproducts and residual catalyst fines behind. This physical separation mechanism ensures that the resulting solid possesses a purity of up to 99.8%, as verified by FT-IR and DSC analysis, making it immediately suitable for polymerization into high-performance polyimides without further purification steps that could introduce new contaminants.
How to Synthesize 2,2-bis(3-amino-4-hydroxyphenyl)hexafluoropropane Efficiently
The synthesis protocol outlined in the patent offers a robust framework for laboratory and pilot-scale production, emphasizing safety and reproducibility. The process begins with the careful charging of the nitro-precursor and alcohol solvent into a high-pressure autoclave, followed by the addition of the catalyst. It is imperative to thoroughly purge the system with hydrogen to remove all residual oxygen, preventing the formation of explosive mixtures and ensuring the catalyst remains in its active reduced state. Once the reaction parameters are stabilized, the system is heated and pressurized, allowing the hydrogenation to proceed to completion over a defined period. Following the reaction, the cooling and depressurization phases must be managed slowly to maintain system integrity, after which the catalyst is filtered off for regeneration.
- Mix 2,2-bis(3-nitro-4-hydroxyphenyl)hexafluoropropane with organic alcohol solvent (methanol or ethanol) and add Pd/C or Raney Nickel catalyst in a high-pressure reactor.
- Replace air with hydrogen, heat to 60°C–100°C, and maintain pressure between 0.3MPa and 3.6MPa for 2 to 5 hours to complete the reduction.
- Cool the system, filter off the catalyst, and add deionized water to the mother liquor to precipitate the white solid product directly.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology translates into tangible strategic advantages regarding cost structure and supply continuity. The elimination of toxic hydrazine hydrate removes a major regulatory and safety bottleneck, simplifying the logistics of raw material handling and storage. Furthermore, the ability to recover and reuse the alcohol solvent significantly reduces the volume of fresh solvent required per batch, directly lowering variable production costs. The high yield and single-step purification mean that less raw material is wasted, and the throughput of the manufacturing facility is maximized, as reactors are not tied up in lengthy recrystallization cycles. This efficiency is critical for maintaining a competitive edge in the fast-paced market for electronic chemicals, where demand for high-performance materials is constantly rising.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the downstream processing units. By removing the need for recrystallization, manufacturers save substantially on energy consumption associated with heating and cooling large volumes of solvent, as well as the capital expenditure for crystallization tanks and drying equipment. Additionally, the recovery of the precious metal catalyst (Pd/C or Raney Ni) allows for the amortization of catalyst costs over multiple batches, rather than treating it as a single-use consumable. This circular approach to catalyst management, combined with the high atom economy of hydrogenation, results in a significantly lower cost of goods sold (COGS) compared to legacy methods, enabling more aggressive pricing strategies for the final fluorinated polyimide products.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity-grade solvents such as methanol and ethanol, which are readily available globally and less susceptible to the supply shocks that often affect specialized reagents like hydrazine. The robustness of the reaction conditions also means that the process is less sensitive to minor fluctuations in raw material quality, reducing the rate of batch failures and ensuring consistent output. For a reliable fluorinated polyimide monomer supplier, this reliability is paramount; it guarantees that customers in the semiconductor and display industries receive their materials on schedule, preventing costly downtime in their own production lines. The simplified workflow also shortens the overall manufacturing lead time, allowing for faster response to market demand spikes.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method is ideally suited for expansion from pilot plants to multi-ton commercial production. The absence of severe toxicity and corrosive byproducts simplifies the permitting process for new manufacturing facilities, as the environmental impact assessment is far more favorable than for hydrazine-based processes. The "three wastes" (wastewater, waste gas, solid waste) are minimized, particularly the wastewater which is primarily alcohol and water, making it easier to treat and recycle. This alignment with green chemistry standards not only future-proofs the manufacturing operation against tightening environmental regulations but also enhances the brand value for end-users who are increasingly prioritizing sustainable supply chains in their procurement decisions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this monomer into their existing polymer formulations or supply networks. The answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a factual basis for decision-making.
Q: What are the primary advantages of this catalytic hydrogenation method over traditional hydrazine reduction?
A: Unlike traditional methods using toxic hydrazine hydrate which generate severe three-waste issues and require complex recrystallization, this patent utilizes catalytic hydrogenation with water precipitation. This eliminates the need for hazardous reagents, significantly reduces wastewater treatment costs, and achieves higher purity (up to 99.8%) in a single step without additional purification cycles.
Q: Can the catalyst be recovered and reused in this industrial process?
A: Yes, the process explicitly includes a filtration step to recover the filter residue, which contains the valuable Pd/C or Raney Nickel catalyst. This recovery capability is critical for cost reduction in electronic chemical manufacturing, as it minimizes the consumption of precious metals and allows for iterative cycles of catalyst use, thereby lowering the overall raw material expenditure per batch.
Q: How does this method ensure high purity suitable for polyimide applications?
A: The method achieves high purity through precise control of reaction parameters (temperature 60°C–100°C, pressure 0.3MPa–3.6MPa) and a unique isolation technique. By adding deionized water to the alcohol mother liquor, the product precipitates as a white solid while impurities remain in solution. This avoids the thermal stress and potential degradation associated with high-temperature recrystallization, ensuring the structural integrity required for high-performance fluorinated polyimides.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2-bis(3-amino-4-hydroxyphenyl)hexafluoropropane Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced fluorinated materials requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature are successfully translated into consistent, industrial-grade output. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify every batch against the highest industry standards. Our commitment to quality assurance means that every gram of 2,2-bis(3-amino-4-hydroxyphenyl)hexafluoropropane we deliver meets the exacting requirements for high-performance polyimide applications, providing our clients with the confidence they need to innovate.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific application needs. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the potential economic efficiencies available through our manufacturing processes. We encourage you to reach out for specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a trusted partner in your supply chain. Together, we can accelerate the development of next-generation electronic materials while achieving superior cost and performance metrics.
