Advanced Catalytic Hydrogenation for High-Purity Fluorinated Diamines in Electronic Material Manufacturing
Advanced Catalytic Hydrogenation for High-Purity Fluorinated Diamines in Electronic Material Manufacturing
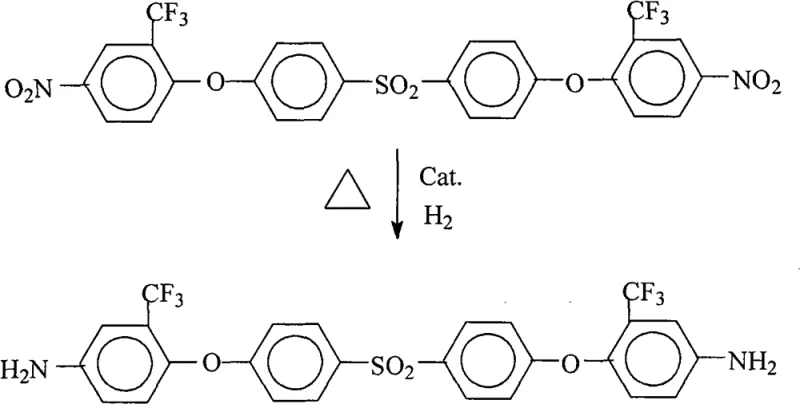
The rapid evolution of the microelectronics and aerospace sectors demands polymer materials with exceptional thermal stability, low dielectric constants, and superior mechanical strength. Central to meeting these rigorous specifications is the development of advanced fluorinated polyimides, which rely heavily on high-quality monomers such as 4,4'-bis(4-amino-2-trifluoromethylphenoxy)diphenylsulfone. A pivotal advancement in the synthesis of this critical intermediate is detailed in patent CN101591277B, which introduces a robust catalytic hydrogenation methodology. This technological breakthrough addresses long-standing challenges in the production of fluorinated aromatic diamines, specifically targeting the elimination of hazardous reagents and the enhancement of process efficiency. By shifting from traditional stoichiometric reduction methods to a catalytic system utilizing hydrogen gas and supported metal catalysts, manufacturers can achieve yields exceeding 97% with purities reaching 99.8%. This report analyzes the technical merits of this approach, offering strategic insights for R&D directors seeking process optimization and procurement leaders focused on sustainable supply chain solutions for high-performance electronic chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aromatic diamines containing sensitive functional groups like trifluoromethyl moieties has relied heavily on reduction protocols involving hydrazine hydrate. While chemically effective in converting nitro groups to amines, the use of hydrazine presents severe drawbacks for modern industrial manufacturing. Firstly, hydrazine hydrate is classified as a highly toxic and potentially carcinogenic substance, imposing stringent regulatory burdens and necessitating expensive containment and waste treatment infrastructure to ensure operator safety. Secondly, conventional hydrazine reduction processes often suffer from moderate yields, typically hovering around 90%, which translates to significant material loss when scaling to metric ton quantities. Furthermore, these older methods frequently require subsequent recrystallization steps to remove byproducts and unreacted intermediates, thereby increasing energy consumption, extending production cycles, and generating substantial volumes of organic solvent waste. For a reliable fluorinated polyimide monomer supplier, relying on such inefficient and hazardous chemistry creates unacceptable risks regarding environmental compliance and cost competitiveness in the global market.
The Novel Approach
The methodology disclosed in the referenced patent represents a paradigm shift towards green chemistry principles by employing catalytic hydrogenation under controlled pressure and temperature conditions. Instead of using stoichiometric amounts of toxic reducing agents, this novel approach utilizes molecular hydrogen in the presence of heterogeneous catalysts such as palladium on carbon (Pd/C) or Raney nickel. This transition not only mitigates the safety hazards associated with hydrazine but also streamlines the downstream processing workflow. The reaction proceeds smoothly at temperatures between 95°C and 110°C under a hydrogen pressure of 0.8 MPa to 1.2 MPa, ensuring complete conversion of the dinitro precursor to the desired diamine. Crucially, the workup procedure is remarkably simple; the solid catalyst is filtered off for potential reuse, and the product is precipitated directly from the mother liquor by the addition of deionized water. This eliminates the need for complex purification sequences like recrystallization, thereby facilitating cost reduction in electronic chemical manufacturing through reduced utility usage and simplified waste management protocols.
Mechanistic Insights into Pd/C Catalyzed Nitro Reduction
The core of this synthetic strategy lies in the heterogeneous catalytic reduction of the nitro functional groups (-NO2) to primary amines (-NH2) while preserving the integrity of the trifluoromethyl (-CF3) and sulfone (-SO2-) linkages. In this mechanism, hydrogen molecules adsorb onto the surface of the palladium or nickel catalyst, dissociating into reactive atomic hydrogen species. These active hydrogen atoms are then transferred to the nitrogen atoms of the nitro groups attached to the aromatic rings. The presence of electron-withdrawing groups, such as the trifluoromethyl substituent at the ortho position, can influence the electron density of the aromatic ring, potentially affecting the reduction kinetics. However, the optimized conditions described in the patent ensure that the reduction is selective and exhaustive, avoiding the formation of partially reduced intermediates like hydroxylamines or azo-compounds which could act as chromophoric impurities. Maintaining the reaction temperature within the 95°C-110°C window is critical; it provides sufficient thermal energy to overcome the activation barrier for the reduction without promoting side reactions such as hydrodehalogenation, although the C-F bonds in the CF3 group are generally robust under these specific hydrogenation conditions.
Impurity control is a paramount concern for R&D directors aiming to produce high-purity fluorinated diamine suitable for optical applications. The patented process achieves exceptional purity levels (99.8%) primarily due to the cleanliness of the catalytic hydrogenation reaction and the efficacy of the precipitation workup. Unlike liquid-phase reductions that generate stoichiometric amounts of salt byproducts (e.g., from iron/acids or sulfide reductions), catalytic hydrogenation produces water as the only byproduct. This absence of inorganic salts simplifies the isolation process significantly. Furthermore, the precipitation step, where deionized water is added to the alcoholic mother liquor, exploits the solubility differences between the highly polar diamine product and non-polar organic impurities. The resulting white solid precipitate is inherently pure, as evidenced by the sharp melting point range of 218.3°C-218.9°C. This high degree of chemical homogeneity is essential for the subsequent polymerization into polyimides, as even trace impurities can disrupt the molecular weight distribution and degrade the thermal and mechanical properties of the final polymer film used in flexible displays or aerospace components.
How to Synthesize 4,4'-bis(4-amino-2-trifluoromethylphenoxy)diphenylsulfone Efficiently
The implementation of this synthesis route requires precise control over reaction parameters to maximize yield and safety. The process begins with the charging of the dinitro precursor, an alcohol-based solvent system (such as methanol, ethanol, or glycols), and the selected catalyst into a high-pressure autoclave. It is imperative to purge the system with inert gas followed by hydrogen to eliminate oxygen, preventing the formation of explosive mixtures. Once the reaction conditions of 0.8-1.2 MPa and 95-110°C are established, the mixture is agitated for 4 to 5 hours to ensure complete conversion. Following the reaction, the system is cooled and depressurized safely. The catalyst is removed via filtration, allowing for its recovery and regeneration. The filtrate, containing the dissolved diamine, is then treated with a specific volume of deionized water to induce crystallization. This straightforward protocol minimizes unit operations and maximizes throughput, making it ideal for the commercial scale-up of complex aromatic sulfones. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below.
- Load the dinitro precursor, organic solvent (methanol/ethanol), and Pd/C or Raney Ni catalyst into a high-pressure reactor.
- Pressurize with hydrogen to 0.8-1.2 MPa and heat to 95-110°C for 4-5 hours to effect complete nitro reduction.
- Filter the catalyst, retain the mother liquor, and precipitate the pure diamine product by adding deionized water.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this catalytic hydrogenation technology offers transformative benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the EHS (Environment, Health, and Safety) profile. By eliminating hydrazine hydrate, a substance subject to strict global transport and storage regulations, companies can significantly reduce the administrative burden and insurance costs associated with hazardous material handling. This shift also enhances supply chain resilience, as the reliance on specialized toxic reagents is replaced by commodity chemicals like hydrogen and alcohols, which are readily available from multiple sources. Furthermore, the ability to recover and recycle the precious metal catalyst (Pd/C) contributes to substantial long-term cost savings, insulating the manufacturing process from volatility in precious metal markets. The simplified workup, which avoids energy-intensive distillation or recrystallization, further drives down the operational expenditure per kilogram of product, ensuring a competitive pricing structure for downstream polymer manufacturers.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive and hazardous stoichiometric reagents. Traditional methods using hydrazine require careful disposal of toxic waste, which incurs high treatment fees. In contrast, the catalytic method generates water as the main byproduct, drastically lowering waste disposal costs. Additionally, the recovery of the catalyst allows for the reuse of palladium or nickel, reducing the raw material cost per batch. The omission of recrystallization steps saves significant amounts of energy and solvent, leading to a leaner, more cost-effective production model that improves overall margin potential without compromising product quality.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of robust and widely available raw materials. Hydrogen gas and alcohol solvents are commodity chemicals with stable supply networks, unlike specialized reducing agents that may face supply disruptions. The simplified process flow also reduces the risk of production bottlenecks caused by complex purification stages. Faster cycle times, achieved by removing recrystallization and streamlining the workup, mean that production capacity can be increased without additional capital investment in new reactors. This agility allows suppliers to respond more rapidly to fluctuations in market demand for high-performance polyimide precursors, ensuring consistent delivery schedules for key clients in the electronics sector.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial production is straightforward due to the well-understood nature of high-pressure hydrogenation in the fine chemical industry. The equipment required—standard high-pressure autoclaves—is readily available and easily integrated into existing facilities. Environmentally, the process aligns with green chemistry principles by minimizing waste generation and avoiding toxic reagents. The solvent system can be designed for closed-loop recycling, further reducing the environmental footprint. This compliance with increasingly stringent environmental regulations future-proofs the manufacturing asset, reducing the risk of regulatory shutdowns and enhancing the corporate sustainability profile, which is increasingly important for multinational customers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of fluorinated diamines using this advanced hydrogenation technique. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the technology's viability. Understanding these details is crucial for stakeholders assessing the feasibility of integrating this monomer into their supply chains or R&D pipelines. The answers highlight the balance between high performance and operational safety that defines this modern synthetic approach.
Q: Why is catalytic hydrogenation preferred over hydrazine reduction for this fluorinated diamine?
A: Catalytic hydrogenation eliminates the use of highly toxic hydrazine hydrate, significantly improving operator safety and environmental compliance while increasing yield from 90% to over 97%.
Q: What is the purity level achievable with this patented process?
A: The process yields product with a purity as high as 99.8% and a sharp melting point of 218.3°C-218.9°C without requiring energy-intensive recrystallization steps.
Q: Can the solvents and catalysts be recovered in this manufacturing route?
A: Yes, the solid catalyst (Pd/C or Raney Ni) is recovered via filtration for reuse, and the organic solvent mother liquor can be recycled, drastically reducing raw material consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-bis(4-amino-2-trifluoromethylphenoxy)diphenylsulfone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced fluorinated polyimides requires a partner who understands both the intricacies of organic synthesis and the demands of industrial scale-up. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high purity and yield demonstrated in laboratory patents are faithfully reproduced in our manufacturing facilities. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch of 4,4'-bis(4-amino-2-trifluoromethylphenoxy)diphenylsulfone. Our commitment to quality ensures that the monomers we supply meet the exacting standards required for high-end electronic applications, including flexible printed circuit boards and liquid crystal display alignment films.
We invite procurement managers and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized catalytic hydrogenation processes, we can offer competitive pricing structures that reflect the efficiencies of modern green chemistry. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your projects. Whether you are developing next-generation optical films or high-temperature insulation materials, our expertise in fluorine chemistry positions us as the ideal partner to support your innovation goals and secure your supply chain against future disruptions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
