Scalable Production of High-Purity Polyimide Monomers for Advanced Electronics
The rapid evolution of the flexible electronics sector has placed unprecedented demands on the thermal and mechanical properties of substrate materials, specifically Flexible Copper Clad Laminates (FCCL). At the heart of these advanced materials lies the critical need for high-performance polyimide resins, which derive their exceptional stability from specialized aromatic diamine monomers. Patent CN101289406A introduces a groundbreaking preparation method for 1,4-bis(2,4-diaminophenoxy)-2-tert-butylbenzene, a key building block that imparts superior heat resistance and low dielectric loss to polymer matrices. This technical insight report analyzes the proprietary synthesis route detailed in the patent, highlighting its potential to revolutionize the supply chain for reliable electronic chemical suppliers. By leveraging a catalytic transfer hydrogenation strategy, this method addresses the longstanding challenges of purity and scalability that have historically bottlenecked the production of high-grade polyimide precursors.
For R&D directors and procurement specialists alike, understanding the nuances of this synthesis is vital for securing a competitive edge in the manufacturing of next-generation electronic components. The transition from laboratory-scale curiosity to industrial viability often hinges on the robustness of the reduction step, where nitro groups are converted into amines. The methodology outlined in CN101289406A offers a compelling alternative to traditional high-pressure hydrogenation, utilizing a palladium on carbon (Pd/C) catalyst system in conjunction with hydrazine hydrate. This approach not only simplifies the operational workflow but also significantly enhances the safety profile of the manufacturing process, making it an attractive option for cost reduction in electronic chemical manufacturing where safety and efficiency are paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the reduction of polynitro aromatic compounds to their corresponding polyamines has been fraught with significant engineering and economic challenges. Traditional methods often rely on catalytic hydrogenation using molecular hydrogen gas, which necessitates the use of expensive high-pressure reactors and rigorous safety protocols to mitigate explosion risks. Alternatively, chemical reduction using metals like iron in acidic media generates vast quantities of toxic sludge and wastewater, creating a severe environmental burden and escalating waste disposal costs. Furthermore, these conventional routes frequently struggle with selectivity issues, leading to the formation of partially reduced intermediates or azo-byproducts that compromise the purity of the final monomer. For a reliable polyimide monomer supplier, these impurities are unacceptable as they can drastically alter the molecular weight and thermal properties of the resulting polymer, leading to batch failures in downstream applications.
The Novel Approach
The innovative process described in the patent data circumvents these pitfalls by employing a hydrazine hydrate reduction system mediated by a Pd/C catalyst under atmospheric pressure. This method operates within a温和 temperature range of 70°C to 85°C, eliminating the need for energy-intensive high-pressure infrastructure. The use of saturated fatty alcohols as solvents provides a stable medium that facilitates efficient mass transfer while remaining compatible with the catalyst system. Crucially, the reaction byproducts are primarily nitrogen gas and water, which are environmentally benign and easily separated from the reaction mixture. This clean reaction profile means that the crude product often possesses sufficient purity (>99%) to be used directly after simple crystallization, bypassing the need for complex chromatographic purification or multiple recrystallization steps that typically erode profit margins.
Mechanistic Insights into Pd/C-Catalyzed Transfer Hydrogenation
The core of this synthesis lies in the efficient catalytic transfer of hydrogen from hydrazine to the nitro groups of the substrate. In this mechanism, the palladium nanoparticles dispersed on the carbon support act as active sites for the decomposition of hydrazine (N2H4), generating reactive hydrogen species in situ. These surface-bound hydrogen atoms are then transferred to the oxygen atoms of the nitro groups (-NO2) attached to the aromatic rings of the 1,4-bis(2,4-dinitrophenoxy)-2-tert-butylbenzene precursor. The reduction proceeds through several intermediate stages, likely involving nitroso and hydroxylamine species, before finally yielding the stable primary amine groups (-NH2). The presence of the tert-butyl group on the central benzene ring plays a subtle yet important role by influencing the solubility of the intermediate species and preventing unwanted intermolecular cross-linking during the reaction, thereby ensuring the linearity and processability of the final polyimide resin.
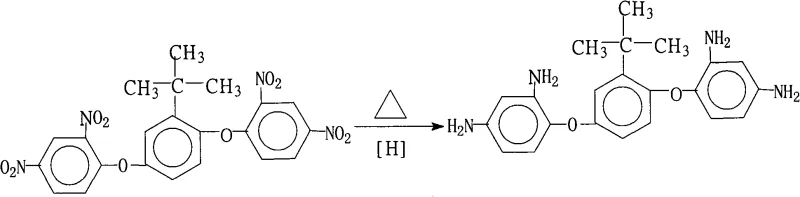
From an impurity control perspective, the choice of hydrazine hydrate concentration (60%-85%) and the specific loading of the Pd/C catalyst (1%-15%) are critical parameters that dictate the reaction kinetics and selectivity. If the catalyst loading is too low, the reaction may stall at the hydroxylamine stage, leading to colored impurities that are difficult to remove. Conversely, excessive catalyst loading does not proportionally increase the rate but increases the cost and the risk of metal contamination in the final product. The patent specifies a hot filtration step immediately following the reaction, which is a strategic move to remove the spent catalyst before the product crystallizes upon cooling. This prevents the catalyst from acting on the newly formed amines or facilitating side reactions during the cooling phase, ensuring that the final crystal lattice is free from metallic inclusions and organic byproducts.
How to Synthesize 1,4-bis(2,4-diaminophenoxy)-2-tert-butylbenzene Efficiently
Implementing this synthesis route requires precise control over stoichiometry and thermal profiles to maximize yield and minimize cycle time. The process begins with the suspension of the dinitro precursor in a selected saturated fatty alcohol solvent, such as ethanol or ethylene glycol, followed by the addition of the Pd/C catalyst. The mixture is heated to initiate the reaction, and hydrazine hydrate is added dropwise to control the exotherm and gas evolution. Maintaining the temperature between 70°C and 85°C for a duration of 1 to 5 hours allows for complete conversion, as monitored by HPLC or TLC. The detailed standardized synthetic steps, including specific solvent ratios and work-up procedures, are outlined below to guide process engineers in replicating this high-efficiency protocol.
- Charge the reactor with 1,4-bis(2,4-dinitrophenoxy)-2-tert-butylbenzene, Pd/C catalyst, and a saturated fatty alcohol solvent system.
- Heat the mixture to 60°C and slowly add hydrazine hydrate solution (60%-85% concentration) while maintaining stirring.
- Maintain reaction temperature between 70°C and 85°C for 1 to 5 hours, then filter hot and cool the mother liquor to crystallize the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method translates into tangible operational efficiencies and risk mitigation strategies. The elimination of high-pressure equipment requirements significantly lowers the barrier to entry for manufacturing this monomer, allowing for production in standard glass-lined or stainless steel reactors that are already prevalent in fine chemical facilities. This flexibility enhances supply chain reliability by enabling multi-vendor sourcing strategies, as the technology does not require specialized, proprietary hardware that limits the pool of qualified contract manufacturers. Furthermore, the simplified work-up procedure, which relies on hot filtration and cooling crystallization rather than complex distillation or extraction sequences, drastically reduces the utility consumption and labor hours associated with each batch, driving down the overall cost of goods sold.
- Cost Reduction in Manufacturing: The most significant economic advantage of this process is the removal of downstream purification bottlenecks. Since the patent indicates that the product achieves over 99% purity without subsequent refining, manufacturers can save substantial amounts on solvents, energy, and time that would otherwise be spent on recrystallization or column chromatography. Additionally, the use of hydrazine hydrate as a reducing agent is generally more cost-effective than managing high-pressure hydrogen gas logistics, and the catalyst can potentially be recovered and regenerated, further optimizing the raw material spend. These factors combine to create a leaner manufacturing process that improves margin potential in a competitive market.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including various saturated fatty alcohols and hydrazine hydrate, are commodity chemicals with robust global supply chains. This availability ensures that production schedules are not disrupted by the scarcity of exotic reagents. Moreover, the mild reaction conditions reduce the likelihood of unplanned shutdowns due to equipment stress or safety incidents, ensuring a consistent flow of high-purity polyimide monomers to downstream polymerization units. This stability is crucial for maintaining just-in-time inventory levels for electronics manufacturers who cannot afford delays in their substrate production lines.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial tonnage is straightforward due to the absence of gas-liquid mass transfer limitations typical of hydrogenation reactions. The liquid-phase nature of the hydrazine reduction ensures uniform mixing and heat distribution even in large reactors. From an environmental standpoint, the generation of nitrogen gas as the primary byproduct aligns with increasingly stringent green chemistry regulations, minimizing the facility's carbon footprint and wastewater treatment load. This compliance reduces the regulatory burden and potential fines, making the operation more sustainable and socially responsible in the long term.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized diamine monomer. These answers are derived directly from the technical specifications and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this material into their existing product portfolios and supply networks.
Q: What is the purity level achievable with this synthesis method?
A: The patented process consistently achieves product purity exceeding 99% without the need for complex subsequent purification steps, making it ideal for high-performance electronic applications.
Q: Why is hydrazine hydrate preferred over catalytic hydrogenation for this reduction?
A: Hydrazine hydrate serves as a convenient liquid reducing agent that operates at atmospheric pressure, eliminating the safety risks and capital costs associated with high-pressure hydrogen gas equipment.
Q: Can this process be scaled for industrial production of polyimide resins?
A: Yes, the mild reaction conditions (70°C-85°C) and simple work-up procedure involving hot filtration and crystallization make this method highly suitable for large-scale industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-bis(2,4-diaminophenoxy)-2-tert-butylbenzene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent concept to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 1,4-bis(2,4-diaminophenoxy)-2-tert-butylbenzene meets the exacting standards required for high-performance polyimide applications. Our commitment to quality assurance means that you can rely on us to deliver materials that perform predictably in your final electronic formulations.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to reach out for specific COA data and route feasibility assessments to verify how our advanced synthesis capabilities can support your strategic goals. Let us help you secure a stable supply of critical electronic chemicals that drive innovation in the flexible electronics industry.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
