Industrial Synthesis of 2,6-Bis(2,4-Diaminophenoxy)Toluene for High-Performance Polyimides
The rapid evolution of the flexible electronics sector has placed unprecedented demand on the performance metrics of substrate materials, specifically Flexible Copper Clad Laminates (FCCL). At the heart of this technological shift lies the critical need for advanced polyimide precursors that offer superior thermal stability and dielectric properties. Patent CN101260052A introduces a robust and highly efficient preparation method for 2,6-bis(2,4-diaminophenoxy)toluene, a pivotal aromatic diamine monomer used in synthesizing high-branched polyimide resins. This proprietary technology addresses the longstanding challenges of achieving high purity and yield in aromatic amine synthesis without resorting to energy-intensive or hazardous high-pressure hydrogenation protocols. By leveraging a catalytic transfer hydrogenation system utilizing hydrazine hydrate and palladium on carbon, the process delivers a crystalline product with purity levels surpassing 99%, directly meeting the stringent specifications required for next-generation electronic insulation materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the reduction of polynitro aromatic compounds to their corresponding amines has relied heavily on catalytic hydrogenation using molecular hydrogen gas or stoichiometric metal reductions such as iron powder in acidic media. While effective, catalytic hydrogenation necessitates specialized high-pressure reactors and rigorous safety protocols to manage explosive gas mixtures, significantly inflating capital expenditure and operational complexity for manufacturers. Alternatively, metal-based reductions often generate substantial quantities of solid waste sludge, creating severe environmental compliance burdens and complicating the isolation of the final product. Furthermore, conventional methods frequently struggle with selectivity issues, where over-reduction or side reactions can compromise the integrity of sensitive ether linkages present in complex molecules like 2,6-bis(2,4-dinitrophenoxy)toluene, leading to lower yields and requiring costly, multi-step purification procedures to remove trace impurities that could degrade the electrical performance of the final polyimide film.
The Novel Approach
The methodology outlined in CN101260052A represents a paradigm shift towards safer and more sustainable chemical manufacturing by employing hydrazine hydrate as a liquid hydrogen donor in the presence of a heterogeneous Pd/C catalyst. This approach effectively bypasses the need for high-pressure gaseous hydrogen infrastructure, allowing the reaction to proceed under atmospheric pressure within a moderate temperature range of 70°C to 85°C. The use of saturated fatty alcohols, such as ethanol or ethylene glycol, as the reaction medium not only solubilizes the reactants effectively but also facilitates a clean workup procedure where the product can be precipitated simply by the addition of water. This innovation drastically simplifies the post-reaction processing, eliminating the need for complex distillation or chromatographic purification, thereby reducing the overall production cycle time and minimizing the generation of hazardous waste streams associated with traditional reduction techniques.
Mechanistic Insights into Hydrazine-Mediated Catalytic Reduction
The core of this synthetic strategy relies on the efficient transfer of hydrogen atoms from hydrazine to the nitro groups of the substrate, mediated by the palladium surface. In this catalytic cycle, hydrazine adsorbs onto the active sites of the Pd/C catalyst and decomposes to release nitrogen gas and reactive hydrogen species in situ. These activated hydrogen species then attack the electron-deficient nitrogen atoms of the nitro groups (-NO2) on the 2,6-bis(2,4-dinitrophenoxy)toluene molecule. The reaction proceeds through a series of intermediate reduction steps, converting the nitro functionality first to nitroso, then to hydroxylamine, and finally to the primary amine (-NH2). The choice of Pd/C as the catalyst is critical, as it provides high activity at relatively low loadings (with Pd mass percentages ranging from 1% to 15%), ensuring economic viability while maintaining rapid reaction kinetics.
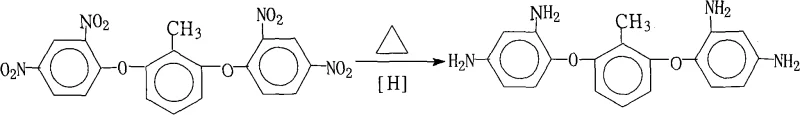
Crucially, the reaction conditions are finely tuned to preserve the structural integrity of the molecule. The ether linkages connecting the aromatic rings are generally stable under these mild reducing conditions, preventing unwanted cleavage that could occur under harsher acidic or basic environments typical of other reduction methods. The patent specifies a molar ratio of substrate to hydrazine hydrate between 1:20 and 1:50, providing a significant excess of the reducing agent to drive the equilibrium towards complete conversion of all four nitro groups. This excess ensures that no partially reduced intermediates remain, which is vital for the subsequent polymerization step, as any residual nitro groups could act as chain terminators or defects in the polyimide backbone, compromising the thermal and mechanical properties of the final electronic material.
How to Synthesize 2,6-Bis(2,4-Diaminophenoxy)Toluene Efficiently
Implementing this synthesis route requires precise control over reagent addition and thermal management to maximize yield and safety. The process begins by charging the reactor with the dinitro precursor, the Pd/C catalyst, and the selected alcohol solvent system, followed by heating to initiate the reaction before the controlled addition of hydrazine. Maintaining the temperature strictly within the 70°C to 85°C window is essential; temperatures below this range may result in incomplete reaction and accumulation of intermediates, while excessive heat could lead to the decomposition of hydrazine or solvent loss. Detailed standardized operating procedures regarding mixing speeds, addition rates, and filtration techniques are critical for reproducibility on a commercial scale.
- Charge the reactor with 2,6-bis(2,4-dinitrophenoxy)toluene, Pd/C catalyst (1-15% Pd loading), and saturated fatty alcohol solvent.
- Heat the mixture to 60°C and slowly add hydrazine hydrate solution (60-85% concentration) while maintaining stirring.
- Maintain reaction temperature between 70°C and 85°C for 1 to 5 hours, then filter hot, cool, precipitate with water, and dry.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this hydrazine-based reduction protocol offers tangible strategic benefits beyond mere technical feasibility. The elimination of high-pressure hydrogenation equipment translates directly into reduced capital investment and lower maintenance overheads for production facilities. Furthermore, the reliance on commodity chemicals such as hydrazine hydrate, ethanol, and palladium on carbon ensures a stable and resilient supply chain, mitigating the risks associated with sourcing specialized or hazardous gases. The simplicity of the workup procedure, which involves hot filtration and water precipitation, significantly reduces utility consumption related to solvent recovery and waste treatment, aligning with modern green chemistry initiatives and lowering the total cost of ownership for the manufacturing process.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive high-pressure autoclaves and the associated safety systems required for gaseous hydrogen, leading to substantial capital expenditure savings. Additionally, the high selectivity of the reaction minimizes the formation of by-products, reducing the raw material waste and the costs associated with downstream purification and waste disposal. The ability to recover and potentially recycle the alcohol solvents further enhances the economic efficiency of the operation, making it a highly competitive route for large-scale production.
- Enhanced Supply Chain Reliability: By utilizing widely available reagents like hydrazine hydrate and common alcohols, manufacturers can avoid supply bottlenecks often seen with specialized reducing agents. The robustness of the Pd/C catalyst, which can be sourced with varying palladium loadings to suit budget constraints, adds another layer of flexibility to procurement strategies. This reliance on standard industrial chemicals ensures consistent production schedules and reduces the risk of downtime caused by material shortages, thereby securing the continuity of supply for critical electronic material customers.
- Scalability and Environmental Compliance: The atmospheric pressure operation and moderate temperature profile make this process inherently safer and easier to scale from pilot plant to full commercial production without significant engineering redesigns. The reduction in hazardous waste generation, particularly the avoidance of heavy metal sludge from iron powder reductions, simplifies environmental compliance and lowers the regulatory burden. This eco-friendly profile not only meets current environmental standards but also future-proofs the manufacturing site against increasingly stringent global regulations on industrial emissions and waste management.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its integration into existing production lines. The following questions address common concerns regarding process safety, product quality, and scalability, drawing directly from the experimental data and beneficial effects reported in the patent literature. These insights are intended to provide clarity on how this method compares to alternative synthetic routes and what operational parameters are critical for success.
Q: What is the purity level achievable with this hydrazine reduction method?
A: The patented process consistently achieves product purity exceeding 99% without the need for complex downstream purification steps, making it ideal for electronic grade applications.
Q: Why is hydrazine hydrate preferred over catalytic hydrogenation for this intermediate?
A: Hydrazine hydrate acts as a liquid reducing agent, eliminating the safety risks and high-pressure infrastructure costs associated with gaseous hydrogen, while offering excellent selectivity for nitro groups.
Q: Can this process be scaled for large-volume FCCL material production?
A: Yes, the method utilizes common solvents like ethanol and glycols and operates at atmospheric pressure with moderate temperatures (70-85°C), ensuring straightforward scalability and operational safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,6-Bis(2,4-Diaminophenoxy)Toluene Supplier
As the demand for high-performance polyimides continues to surge in the aerospace and flexible electronics industries, securing a dependable source of high-purity monomers is paramount. NINGBO INNO PHARMCHEM stands ready to support your development and production needs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the precise thermal and catalytic requirements of this hydrazine reduction process, ensuring that every batch meets stringent purity specifications through our rigorous QC labs. We understand that consistency is key in electronic material manufacturing, and our dedicated technical team is committed to delivering products that enable your next breakthrough in FCCL and composite technologies.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced synthesis capabilities can enhance your product quality and operational efficiency. Let us be your partner in transforming innovative chemical processes into commercial reality.
