Advanced Catalytic Reduction for High-Purity 4,4'-Bis(2,4-diaminophenoxy)diphenyl Ether: Commercial Scale-Up and Process Optimization
Advanced Catalytic Reduction for High-Purity 4,4'-Bis(2,4-diaminophenoxy)diphenyl Ether: Commercial Scale-Up and Process Optimization
The rapid evolution of the flexible electronics and aerospace sectors has driven an unprecedented demand for high-performance polyimide materials that exhibit superior thermal stability and mechanical strength. At the heart of synthesizing these advanced polymers lies the critical need for high-purity aromatic diamine monomers, specifically 4,4'-bis(2,4-diaminophenoxy)diphenyl ether. Patent CN101260058A introduces a groundbreaking industrial methodology for preparing this key intermediate through a highly efficient catalytic transfer hydrogenation process. This technical disclosure represents a significant leap forward from traditional reduction techniques, offering a pathway that eliminates the need for complex post-reaction purification while achieving exceptional product purity levels exceeding 99%. For R&D directors and process engineers, this patent provides a robust framework for scaling the production of branched polyimide resins and polymaleimide systems without compromising on quality or environmental standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the reduction of polynitro aromatic compounds to their corresponding polyamines has been plagued by significant operational challenges and environmental liabilities. Traditional methods often rely on stoichiometric reductions using iron powder in acidic media or high-pressure catalytic hydrogenation with molecular hydrogen. The iron-acid reduction route, while inexpensive in terms of raw materials, generates massive quantities of iron sludge and acidic wastewater, creating a severe burden on waste treatment facilities and violating modern green chemistry principles. Furthermore, the separation of the amine product from the iron oxide byproducts is notoriously difficult, often requiring multiple extraction and distillation steps that degrade overall yield and introduce metallic impurities detrimental to electronic-grade applications. On the other hand, high-pressure hydrogenation requires specialized, capital-intensive reactor vessels capable of withstanding extreme pressures, posing safety risks and limiting the agility of manufacturing operations to respond to market fluctuations.
The Novel Approach
The methodology outlined in CN101260058A circumvents these historical bottlenecks by employing a mild yet highly effective hydrazine hydrate reduction system mediated by a palladium on carbon (Pd/C) catalyst. This approach operates at atmospheric pressure and moderate temperatures ranging from 80°C to 90°C, drastically reducing the energy consumption and safety hazards associated with high-pressure hydrogen gas. The use of hydrazine hydrate as the hydrogen donor ensures a clean reaction profile where the only byproduct is nitrogen gas and water, effectively eliminating the generation of heavy metal sludge. Moreover, the process is designed with a built-in purification mechanism; by filtering the reaction mixture while hot and subsequently inducing crystallization through the addition of water, the protocol achieves high-purity crystals directly from the mother liquor. This "reaction-crystallization" integration simplifies the downstream processing train, removing the need for column chromatography or vacuum distillation, which are often costly and time-consuming at an industrial scale.
Mechanistic Insights into Pd/C-Catalyzed Transfer Hydrogenation
The core of this synthetic breakthrough lies in the efficient transfer of hydrogen atoms from hydrazine to the nitro groups of the substrate, facilitated by the heterogeneous Pd/C catalyst. In this mechanistic cycle, hydrazine adsorbs onto the palladium surface where it undergoes dehydrogenation to release active hydrogen species and nitrogen gas. These activated hydrogen atoms are then transferred to the electron-deficient nitrogen atoms of the nitro groups on the 4,4'-bis(2,4-dinitrophenoxy)diphenyl ether molecule. The reaction proceeds through several intermediate stages, potentially involving nitroso and hydroxylamine intermediates, before fully reducing to the stable primary amine functionality. The choice of saturated fatty alcohols as the solvent system is critical, as these solvents not only dissolve the organic substrate effectively but also stabilize the catalyst surface, preventing agglomeration and ensuring consistent catalytic activity throughout the 2 to 8-hour reaction window.
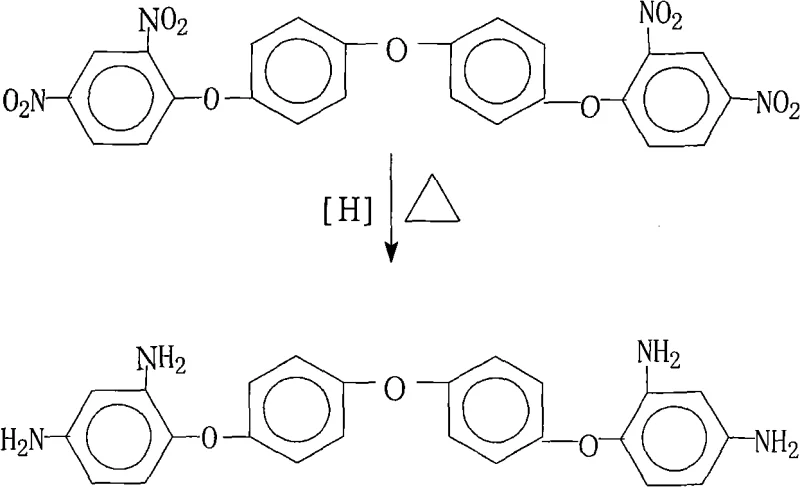
From an impurity control perspective, this mechanism offers superior selectivity compared to non-catalytic reductions. The mild conditions prevent the cleavage of the sensitive ether linkages that connect the aromatic rings, a common side reaction in harsh acidic or high-temperature environments. The patent data indicates that by optimizing the catalyst loading (between 1% and 15% Pd content) and the molar ratio of hydrazine (1:20 to 1:50), the formation of azo or azoxy coupling byproducts is minimized. The subsequent hot filtration step effectively removes the solid Pd/C catalyst, ensuring that the final product is free from palladium contamination, which is a critical specification for electronic grade materials used in flexible copper clad laminates (FCCL). This precise control over the reaction environment allows manufacturers to consistently achieve purity levels above 99%, meeting the rigorous demands of high-end polymer synthesis.
How to Synthesize 4,4'-Bis(2,4-diaminophenoxy)diphenyl Ether Efficiently
Implementing this synthesis route in a pilot or commercial plant requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and safety. The process begins with the careful charging of the reactor with the dinitro precursor, the selected solvent system (such as ethanol or ethylene glycol), and the Pd/C catalyst. Temperature control is paramount during the addition of hydrazine hydrate; the mixture is initially heated to around 60°C before the dropwise addition begins to manage the exothermic nature of the decomposition. Once the addition is complete, the temperature is raised to the 80°C–90°C range and maintained for the requisite duration to drive the conversion to completion.
- Charge the reactor with 4,4'-bis(2,4-dinitrophenoxy)diphenyl ether, Pd/C catalyst (1-15% Pd), and a saturated fatty alcohol solvent system.
- Heat the mixture to 60°C and slowly add hydrazine hydrate solution (60-85% concentration) while maintaining a molar ratio of 1: 20 to 1:50.
- Maintain reaction temperature between 80°C and 90°C for 2 to 8 hours, then filter hot, cool the mother liquor, add water to precipitate crystals, and vacuum dry.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this hydrazine-based reduction technology translates into tangible operational efficiencies and risk mitigation strategies. The elimination of high-pressure hydrogen infrastructure significantly lowers the barrier to entry for manufacturing this intermediate, allowing for more flexible production scheduling and reduced capital expenditure on specialized equipment. Furthermore, the reliance on commodity chemicals like hydrazine hydrate and common alcohols ensures a stable and resilient supply chain, insulating the production process from the volatility often associated with specialty gases or rare metal catalysts. The simplified work-up procedure, which avoids complex distillation columns, reduces the overall cycle time per batch, thereby increasing the throughput capacity of existing facilities without the need for major expansion.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the removal of expensive purification steps and the high atom economy of the catalytic system. By avoiding the use of stoichiometric metal powders like iron or zinc, the process eliminates the costs associated with purchasing, handling, and disposing of tons of solid waste. The ability to recover and recycle the alcohol solvents further drives down the variable cost per kilogram of the final product. Additionally, the high yield reported in the patent examples minimizes raw material wastage, ensuring that the maximum amount of input material is converted into saleable high-value product, directly improving the gross margin for the manufacturer.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more predictable and reliable supply schedule. Unlike processes sensitive to trace impurities or requiring ultra-dry conditions, this method tolerates standard industrial grade solvents, reducing the lead time for raw material procurement. The simplicity of the isolation technique—filtration and crystallization—means that production bottlenecks in the downstream finishing area are virtually eliminated. This reliability is crucial for maintaining continuous feed to polymerization lines, preventing costly downtime in the production of downstream polyimide films and advanced composite materials.
- Scalability and Environmental Compliance: Scaling this process from laboratory to multi-ton production is straightforward due to the absence of hazardous high-pressure steps and the use of standard stirred-tank reactors. The environmental footprint is significantly reduced, as the process generates minimal aqueous waste and no heavy metal sludge, facilitating easier compliance with increasingly stringent global environmental regulations. This "green" profile not only reduces waste disposal costs but also enhances the corporate sustainability image, a growing factor in supplier selection criteria for major multinational electronics and automotive companies seeking eco-friendly supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the experimental data and beneficial effects described in the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this monomer into their existing polymer formulations or supply networks.
Q: What is the achieved purity of the final diamino product using this hydrazine reduction method?
A: According to patent CN101260058A, the process consistently yields 4,4'-bis(2,4-diaminophenoxy)diphenyl ether with a purity exceeding 99%, often reaching up to 99.8% without requiring complex subsequent purification steps.
Q: Which solvents are compatible with this catalytic reduction system?
A: The patent specifies a versatile range of saturated fatty alcohol solvents, including methanol, ethanol, isopropanol, ethylene glycol, and various glycol ethers like 2-methoxyethanol, allowing for flexibility in solvent recovery and cost management.
Q: How does this method improve environmental compliance compared to traditional metal-acid reductions?
A: By utilizing hydrazine hydrate and recoverable Pd/C instead of stoichiometric metal powders and strong acids, the process significantly reduces heavy metal waste and acidic wastewater, aligning with stricter environmental regulations for fine chemical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Bis(2,4-diaminophenoxy)diphenyl Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires a partner with deep technical expertise and scalable infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of CN101260058A are realized in every batch we deliver. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify that every shipment of 4,4'-bis(2,4-diaminophenoxy)diphenyl ether meets the exacting standards required for high-performance polyimide applications. Our commitment to quality assurance means that your R&D and production teams can rely on a consistent supply of material that performs predictably in your polymerization processes.
We invite you to collaborate with us to optimize your supply chain for next-generation electronic materials. By leveraging our process engineering capabilities, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to validate the superiority of our manufacturing approach against your current benchmarks. Let us help you secure a competitive advantage through superior chemistry and reliable supply.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
