Industrial Synthesis of 4,4'-Bis(2,4-diaminophenoxy) Tetramethyldiphenylsulfone for Advanced Polyimide Applications
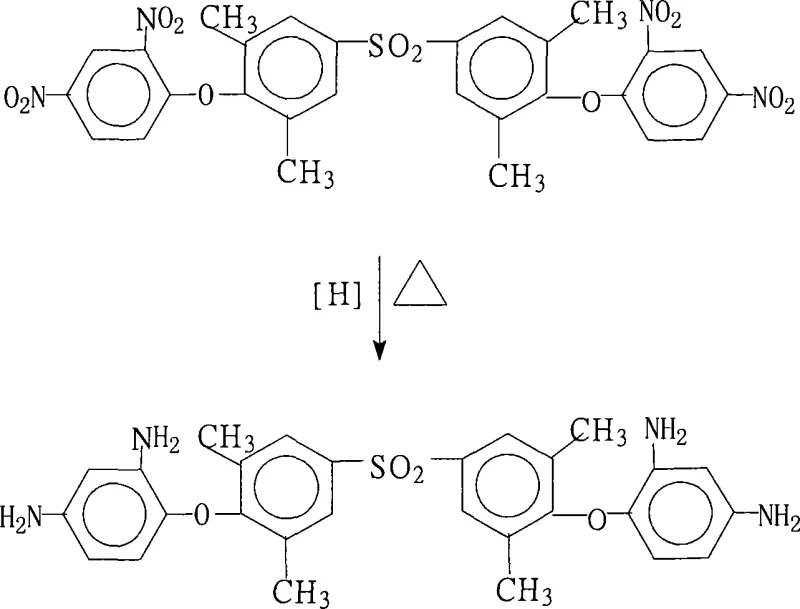
The rapid evolution of the electronics industry, particularly in the sector of flexible printed circuits and high-frequency communication devices, has placed unprecedented demands on the thermal and dielectric properties of polymer substrates. Central to meeting these demands is the development of advanced polyimide precursors that can withstand extreme processing conditions while maintaining electrical integrity. A pivotal breakthrough in this domain is documented in patent CN101250142A, which details a robust and scalable preparation method for 4,4'-bis(2,4-diaminophenoxy)-3,3',5,5'-tetramethyldiphenylsulfone. This specific aromatic polyvalent primary amine serves as a critical building block for high-performance polyimide resins, offering a unique molecular architecture that balances rigidity with processability. The patent outlines a catalytic transfer hydrogenation strategy that circumvents the logistical and safety complexities traditionally associated with high-pressure hydrogenation, presenting a viable pathway for the cost reduction in electronic chemical manufacturing. By leveraging a palladium on carbon (Pd/C) catalyst system coupled with hydrazine hydrate in a saturated fatty alcohol medium, this methodology achieves exceptional conversion rates and product purity, positioning it as a preferred route for industrial-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the reduction of nitro groups to amino groups in complex aromatic systems like diphenylsulfone derivatives has been fraught with significant engineering and economic challenges. Traditional catalytic hydrogenation, while effective, necessitates the use of specialized high-pressure reactors capable of withstanding substantial hydrogen pressures, which introduces severe safety hazards and requires heavy capital investment in explosion-proof infrastructure. Furthermore, the handling of gaseous hydrogen on a large scale involves complex logistics and stringent regulatory compliance, often leading to extended lead times for high-purity intermediates due to supply chain bottlenecks. Alternative chemical reduction methods, such as those employing iron powder in acidic media or sulfide reductions, generate massive quantities of hazardous waste sludge and wastewater, creating an environmental burden that contradicts modern green chemistry principles. These conventional routes often suffer from poor selectivity, leading to the formation of azo or azoxy byproducts that are difficult to separate, thereby compromising the purity required for electronic-grade applications and necessitating energy-intensive purification steps.
The Novel Approach
The methodology described in the patent represents a paradigm shift by utilizing hydrazine hydrate as a liquid hydrogen donor in the presence of a heterogeneous Pd/C catalyst. This approach operates under atmospheric or near-atmospheric pressure conditions, effectively removing the need for high-pressure equipment and drastically simplifying the reactor design and operational safety protocols. The use of saturated fatty alcohols, such as ethanol or ethylene glycol, as the solvent system provides an optimal balance of solubility for the starting dinitro material and insolubility for the final diamino product upon water addition, facilitating a straightforward crystallization process. This novel route not only streamlines the workflow by integrating reaction and isolation into a cohesive sequence but also ensures that the resulting 4,4'-bis(2,4-diaminophenoxy)-3,3',5,5'-tetramethyldiphenylsulfone is obtained with a purity exceeding 99% without the need for subsequent refining. The elimination of heavy metal waste streams and the reduction of energy consumption through lower temperature operations underscore the sustainability and economic viability of this process for commercial scale-up of complex polymer additives.
Mechanistic Insights into Catalytic Transfer Hydrogenation
The core of this synthesis lies in the efficient transfer of hydrogen from hydrazine to the nitro groups mediated by the palladium surface. In this mechanism, hydrazine adsorbs onto the active sites of the Pd/C catalyst, where it undergoes dehydrogenation to release nitrogen gas and reactive hydrogen species. These surface-bound hydrogen atoms are then transferred to the nitro groups of the 4,4'-bis(2,4-dinitrophenoxy)-3,3',5,5'-tetramethyldiphenylsulfone substrate, sequentially reducing them through nitroso and hydroxylamine intermediates to the final amine functionality. The choice of Pd/C with a palladium mass percentage ranging from 1% to 15% is critical, as it optimizes the surface area available for catalysis while minimizing the leaching of precious metal into the product stream. The reaction kinetics are finely tuned by maintaining the temperature within the 80°C to 90°C range, which is sufficient to drive the dehydrogenation of hydrazine without promoting thermal decomposition of the sensitive diamine product or the solvent system.
Impurity control is inherently built into this mechanistic pathway due to the high chemoselectivity of the Pd/C-hydrazine system. Unlike harsher reducing agents that might attack the ether linkages or the sulfone bridge, this catalytic system specifically targets the nitro functionalities, preserving the structural integrity of the bulky tetramethyldiphenylsulfone backbone. The steric hindrance provided by the four methyl groups on the central rings further protects the molecule from unwanted side reactions, ensuring a clean impurity profile. Additionally, the workup procedure involving hot filtration removes the solid catalyst immediately after reaction completion, preventing any post-reaction degradation or over-reduction. The subsequent addition of pure water to the cooled mother liquor exploits the differential solubility of the product, causing it to precipitate as high-quality crystals while leaving soluble byproducts and residual hydrazine in the aqueous-alcoholic supernatant, thus achieving the stringent purity specifications required for reliable polyimide intermediate supplier standards.
How to Synthesize 4,4'-Bis(2,4-diaminophenoxy) Tetramethyldiphenylsulfone Efficiently
Executing this synthesis requires precise control over stoichiometry and thermal parameters to maximize yield and safety. The process begins with the suspension of the dinitro precursor in a selected saturated fatty alcohol solvent, followed by the addition of the Pd/C catalyst. The reaction is initiated by the controlled dropwise addition of hydrazine hydrate, which must be managed carefully to prevent exothermic runaway, although the moderate temperature range mitigates this risk. Following the reaction period, the separation of the catalyst and the crystallization of the product are critical unit operations that determine the final physical form and purity of the material. For a detailed, step-by-step breakdown of the exact reagent ratios, temperature profiles, and isolation techniques validated by experimental data, please refer to the standardized protocol below.
- Charge the reactor with 4,4'-bis(2,4-dinitrophenoxy)-3,3',5,5'-tetramethyldiphenylsulfone, Pd/C catalyst (1%-15% Pd loading), and a saturated fatty alcohol solvent system.
- Heat the mixture to 60°C and slowly add hydrazine hydrate solution (60%-85% concentration) while maintaining a molar ratio of substrate to hydrazine between 1: 20 and 1:50.
- Maintain reaction temperature between 80°C and 90°C for 1 to 5 hours, then filter hot, cool the mother liquor, add water to precipitate crystals, and vacuum dry.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this hydrazine-based reduction technology offers profound advantages in terms of operational expenditure and supply chain resilience. The transition away from high-pressure hydrogenation infrastructure allows manufacturing facilities to repurpose existing standard glass-lined or stainless steel reactors, significantly lowering the barrier to entry for production and reducing the depreciation costs associated with specialized high-pressure vessels. This flexibility translates directly into cost reduction in manufacturing, as the capital intensity per kilogram of produced intermediate is substantially decreased. Furthermore, the reliance on liquid reagents like hydrazine hydrate and common alcohols simplifies logistics, as these materials are widely available commodities with stable supply chains, unlike gaseous hydrogen which often requires on-site generation or complex tube trailer deliveries. This reliability ensures consistent production schedules and minimizes the risk of downtime due to raw material shortages.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment and the associated safety systems results in a drastic simplification of the plant layout and a reduction in maintenance overheads. By avoiding the use of expensive noble metal catalysts in homogeneous forms and instead utilizing recoverable heterogeneous Pd/C, the process minimizes raw material costs and facilitates catalyst recycling. The high yield and purity achieved in a single step remove the necessity for costly downstream purification processes such as column chromatography or multiple recrystallizations, thereby shortening the overall production cycle time and reducing utility consumption per unit of output.
- Enhanced Supply Chain Reliability: The use of readily available solvents like ethanol and ethylene glycol ensures that production is not held hostage by niche chemical supply disruptions. The robustness of the reaction conditions, which tolerate a range of solvent mixtures and catalyst loadings, provides manufacturing teams with the flexibility to substitute grades or suppliers of raw materials without compromising product quality. This adaptability is crucial for maintaining continuous supply to downstream polymer manufacturers, especially in volatile market conditions where agility is a key competitive advantage for any reliable agrochemical intermediate supplier or electronics material provider.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, primarily consisting of nitrogen gas and aqueous alcoholic solutions that can be easily treated or recycled, aligning with increasingly strict global environmental regulations. The simplicity of the workup, involving basic filtration and precipitation, makes the process highly scalable from pilot plant to multi-ton commercial production without encountering the mixing or heat transfer limitations often seen in high-pressure gas-liquid reactions. This ease of scale-up ensures that supply volumes can be rapidly increased to meet surging demand for high-performance polyimides in the aerospace and electronics sectors.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for partners looking to integrate this intermediate into their own polymer formulations. The following questions address common concerns regarding process safety, product specification, and application suitability, derived directly from the technical disclosures and experimental examples within the patent literature. These insights are intended to provide clarity for R&D and procurement teams evaluating the feasibility of adopting this material for next-generation electronic applications.
Q: What is the primary advantage of using hydrazine hydrate over catalytic hydrogenation for this reduction?
A: The hydrazine hydrate method eliminates the need for high-pressure hydrogen gas equipment, significantly reducing capital expenditure and safety risks associated with high-pressure operations while maintaining high selectivity for the amino group.
Q: What purity levels can be achieved with this crystallization method?
A: The patented process utilizes a specific solvent system and water precipitation technique that yields crystals with purity exceeding 99%, often eliminating the need for further recrystallization or column chromatography.
Q: Is this intermediate suitable for flexible copper clad laminates (FCCL)?
A: Yes, this diamine monomer is specifically designed for synthesizing highly branched polyimide resins that offer the low dielectric constant and high thermal stability required for next-generation FCCL and electronic packaging materials.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Bis(2,4-diaminophenoxy) Tetramethyldiphenylsulfone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics observed in patent literature are faithfully reproduced on a commercial scale. We are committed to delivering 4,4'-bis(2,4-diaminophenoxy)-3,3',5,5'-tetramethyldiphenylsulfone with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch against the highest industry standards. Our facility is equipped to handle the specific solvent systems and catalytic requirements of this synthesis, guaranteeing a steady supply of this critical monomer for your polyimide resin production.
We invite you to collaborate with us to optimize your supply chain and reduce your overall material costs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage potential partners to reach out for specific COA data and route feasibility assessments to understand how our optimized manufacturing process can enhance the performance and economics of your final electronic materials. Let us be your strategic partner in advancing the frontiers of high-performance polymers.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
