Advanced Catalytic Reduction for High-Performance Polyimide Monomers and Commercial Scalability
The landscape of advanced electronic materials is constantly evolving, driven by the insatiable demand for flexible, heat-resistant, and high-performance polymers. At the forefront of this innovation is the synthesis of specialized aromatic diamines, which serve as the critical building blocks for next-generation polyimide resins. Specifically, patent CN101260059A introduces a groundbreaking preparation method for 2,6-dimethyl-1,4-bis(2,4-diaminophenoxy)benzene, a molecule of immense value in the fabrication of Flexible Copper Clad Laminates (FCCL) and high-temperature composite materials. This proprietary technique leverages a sophisticated catalytic transfer hydrogenation strategy that fundamentally alters the economic and technical feasibility of producing this complex monomer. By utilizing a palladium on carbon (Pd/C) catalyst system in conjunction with hydrazine hydrate within a saturated fatty alcohol medium, the process achieves exceptional conversion rates under remarkably mild thermal conditions. For R&D directors and procurement specialists alike, this represents a paradigm shift away from hazardous high-pressure hydrogenation towards a safer, more controllable liquid-phase reduction that delivers purity levels exceeding 99% without the need for cumbersome downstream purification steps.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the reduction of polynitro aromatic compounds to their corresponding polyamines has been fraught with significant engineering challenges and safety hazards that hindered widespread industrial adoption. Traditional methodologies often relied on catalytic hydrogenation using molecular hydrogen gas, which necessitated the use of high-pressure autoclaves capable of withstanding extreme stresses, thereby inflating capital expenditure and imposing rigorous safety protocols that slowed down production cycles. Furthermore, alternative chemical reduction methods utilizing iron powder in acidic media generated massive quantities of toxic iron sludge and wastewater, creating an environmental liability that modern sustainable manufacturing simply cannot tolerate. These conventional routes frequently suffered from poor selectivity, leading to the formation of azo or azoxy byproducts that were notoriously difficult to separate from the desired diamine, ultimately compromising the thermal and electrical performance of the final polyimide material. The inability to consistently achieve high purity without multiple recrystallization steps resulted in substantial yield losses and extended lead times, making the reliable sourcing of high-quality 2,6-dimethyl-1,4-bis(2,4-diaminophenoxy)benzene a persistent bottleneck for supply chain managers in the electronic materials sector.
The Novel Approach
In stark contrast to these legacy techniques, the novel approach detailed in the patent data utilizes a catalytic transfer hydrogenation mechanism that elegantly bypasses the need for gaseous hydrogen and harsh acidic conditions. By employing hydrazine hydrate as a liquid hydrogen donor in the presence of a heterogeneous Pd/C catalyst, the reaction proceeds smoothly at atmospheric pressure within a moderate temperature window of 70°C to 85°C. This methodological advancement not only drastically reduces the energy consumption associated with high-pressure compression but also inherently improves the safety profile of the manufacturing facility by eliminating explosive gas hazards. The strategic selection of saturated fatty alcohols as the reaction medium plays a dual role, acting both as a solvent to ensure homogeneous mixing of the bulky dinitro precursor and as a crystallization aid that facilitates the precipitation of the product upon cooling. As illustrated in the reaction scheme below, this streamlined process converts the dinitro starting material directly into the target diamine with high fidelity, minimizing the formation of colored impurities and ensuring that the crude product often meets stringent purity specifications immediately after filtration.
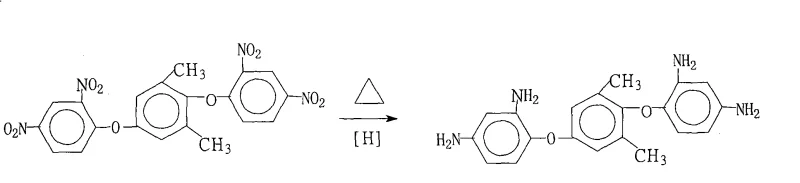
Mechanistic Insights into Pd/C-Catalyzed Transfer Hydrogenation
The core of this technological breakthrough lies in the intricate interplay between the palladium catalyst surface and the hydrazine molecule, which facilitates a highly efficient transfer of hydrogen atoms to the nitro groups. In this catalytic cycle, the hydrazine hydrate adsorbs onto the active sites of the Pd/C catalyst, where it undergoes decomposition to release nascent hydrogen species that are immediately available for reduction reactions. This in-situ generation of reactive hydrogen ensures that the concentration of reducing agents at the catalyst surface remains optimal, preventing the accumulation of partially reduced intermediates such as hydroxylamines which can often couple to form stable azo impurities. The mild thermal conditions of 70°C to 85°C are critical, as they provide sufficient activation energy for the reduction while suppressing thermal degradation pathways that could compromise the integrity of the ether linkages in the molecular backbone. For the R&D team, understanding this mechanism is vital for process optimization, as it highlights the importance of maintaining precise stoichiometric ratios between the hydrazine donor and the nitro substrate to drive the reaction to completion without excess reagent waste.
Furthermore, the choice of solvent system exerts a profound influence on the impurity profile and the physical form of the final product, acting as a crucial parameter for quality control. The saturated fatty alcohols, ranging from methanol to higher glycols, interact with the growing amine molecules through hydrogen bonding, which stabilizes the transition states and prevents the aggregation of byproduct species. Upon completion of the reaction, the solubility characteristics of the diamine product in these alcoholic media change dramatically with temperature, allowing for a 'hot filtration' step that effectively removes the spent Pd/C catalyst and any insoluble particulate matter before crystallization occurs. This thermal crystallization process serves as an intrinsic purification step, where the lattice energy of the forming crystals excludes impurity molecules, resulting in a product with purity levels consistently above 99%. This mechanistic understanding assures technical buyers that the process is robust against minor variations in feedstock quality, providing a reliable pathway to consistent batch-to-batch reproducibility essential for commercial scale-up.
How to Synthesize 2,6-dimethyl-1,4-bis(2,4-diaminophenoxy)benzene Efficiently
Implementing this synthesis route requires careful attention to the sequential addition of reagents and thermal management to maximize yield and safety. The process begins with the suspension of the dinitro precursor and the Pd/C catalyst in the chosen alcoholic solvent system, followed by a controlled heating phase to initiate the reaction environment. Once the mixture reaches the initial activation temperature, the hydrazine hydrate is introduced gradually to manage the exothermic nature of the reduction, ensuring that the reaction temperature remains strictly within the 70°C to 85°C window for a duration of 1 to 5 hours depending on the specific solvent blend used. The detailed standardized operating procedures, including specific weight ratios and stirring speeds required for industrial replication, are outlined in the technical guide below.
- Charge the reactor with 2,6-dimethyl-1,4-bis(2,4-dinitrophenoxy)benzene, Pd/C catalyst, and saturated fatty alcohol solvents.
- Heat the mixture to 60°C and slowly add hydrazine hydrate solution while maintaining strict temperature control.
- Maintain reaction temperature between 70°C and 85°C for 1 to 5 hours, then filter hot and crystallize the product from the mother liquor.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers transformative advantages that directly address the pain points of cost volatility and supply chain fragility often encountered in the fine chemical industry. By shifting from high-pressure gas hydrogenation to a liquid-phase transfer hydrogenation process, manufacturers can significantly reduce the capital intensity of their production facilities, as the requirement for specialized high-pressure vessels and complex gas handling infrastructure is completely eliminated. This reduction in equipment complexity translates directly into lower depreciation costs and reduced maintenance overheads, allowing for a more competitive pricing structure for the final diamine product without sacrificing quality margins. Moreover, the reliance on commodity chemicals such as hydrazine hydrate and common fatty alcohols ensures that the raw material supply chain is resilient and less susceptible to the geopolitical disruptions that often plague the availability of exotic catalysts or specialized gases.
- Cost Reduction in Manufacturing: The elimination of high-pressure equipment and the associated safety systems results in a drastic simplification of the plant layout and operational expenditure. Additionally, the ability to filter the catalyst while hot and crystallize the product directly from the mother liquor removes the need for energy-intensive distillation or chromatographic purification steps, leading to substantial savings in utility costs and solvent consumption. The high atom economy of the hydrazine reduction further ensures that reagent costs are minimized, as the stoichiometric efficiency prevents the wasteful use of expensive reducing agents.
- Enhanced Supply Chain Reliability: The use of widely available saturated fatty alcohols and standard Pd/C catalysts means that procurement teams are not locked into single-source suppliers for critical inputs, thereby mitigating the risk of production stoppages due to raw material shortages. The robustness of the reaction conditions allows for flexible scheduling and rapid turnaround times, enabling suppliers to respond quickly to fluctuating market demands for polyimide precursors. This flexibility is crucial for maintaining continuity in the downstream production of FCCL and advanced composites, where delays in monomer supply can halt entire assembly lines.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste compared to traditional iron-acid reductions, as the primary byproduct is nitrogen gas, which vents harmlessly, and the spent catalyst can often be regenerated or sent for precious metal recovery. This alignment with green chemistry principles simplifies regulatory compliance and reduces the costs associated with wastewater treatment and hazardous waste disposal. The straightforward scale-up from laboratory to multi-ton production is facilitated by the absence of mass transfer limitations typical of gas-liquid reactions, ensuring that process performance remains consistent regardless of batch size.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this specific synthesis route for aromatic diamines. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a transparent view of the technology's capabilities for potential partners and licensees. Understanding these nuances is essential for evaluating the feasibility of integrating this monomer into your existing polymer formulation workflows.
Q: What are the critical purity specifications for FCCL grade diamines?
A: For high-performance Flexible Copper Clad Laminates (FCCL), the diamine monomer typically requires purity exceeding 99% to ensure optimal dielectric properties and thermal stability in the final polyimide resin.
Q: Why is hydrazine hydrate preferred over high-pressure hydrogen for this reduction?
A: Hydrazine hydrate acts as a liquid hydrogen donor in the presence of Pd/C, eliminating the need for expensive high-pressure autoclaves and specialized gas handling infrastructure, thereby simplifying the process safety profile.
Q: How does the solvent system impact crystal formation?
A: The use of saturated fatty alcohols or their mixtures facilitates the dissolution of the bulky dinitro precursor at elevated temperatures while allowing for controlled crystallization upon cooling, which inherently purifies the product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,6-dimethyl-1,4-bis(2,4-diaminophenoxy)benzene Supplier
As the global demand for high-performance electronic materials continues to surge, securing a stable supply of critical monomers like 2,6-dimethyl-1,4-bis(2,4-diaminophenoxy)benzene is paramount for maintaining competitive advantage in the polyimide market. NINGBO INNO PHARMCHEM stands ready to support your development and production needs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the precise thermal and safety requirements of catalytic transfer hydrogenation, ensuring that every batch meets stringent purity specifications and rigorous QC labs standards. We understand that consistency is key in the electronics industry, and our dedicated process engineering team works tirelessly to optimize yield and minimize variability, delivering a product that performs reliably in your most demanding applications.
We invite you to engage with our technical procurement team to discuss how we can tailor our manufacturing capabilities to your specific volume and quality requirements. By partnering with us, you gain access to a Customized Cost-Saving Analysis that evaluates the total landed cost of switching to our optimized supply chain. We encourage you to request specific COA data and route feasibility assessments to verify how our advanced synthesis method can enhance the performance and profitability of your final polyimide products. Let us collaborate to engineer a supply solution that drives innovation and efficiency in your manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
