Advanced Synthesis of Tetra-Amine Monomers for High-Performance Polyimide Manufacturing
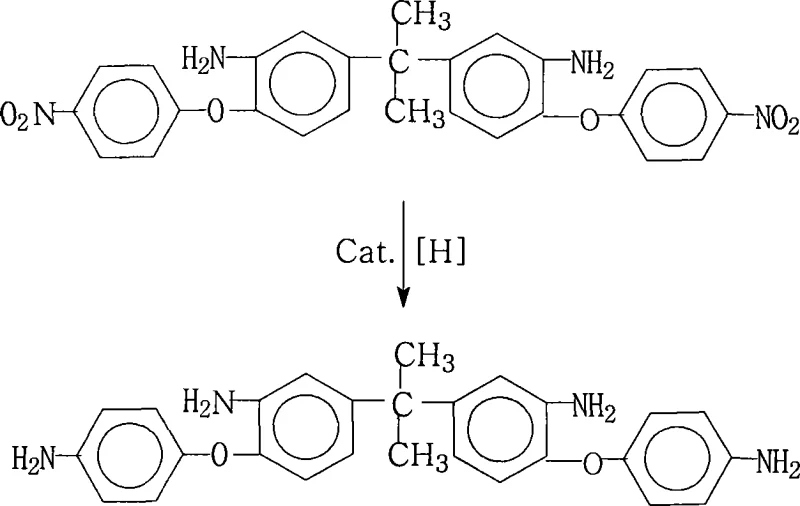
The rapid evolution of the flexible electronics sector demands materials with exceptional thermal stability and dielectric performance, driving the need for advanced polyimide precursors. Patent CN101245027A introduces a groundbreaking preparation method for 2,2-bis[3-amino-4-(4-aminophenoxy)phenyl]propane, a critical tetra-amine monomer used in synthesizing highly branched polyimide resins. This specific molecular architecture is pivotal for developing Flexible Copper Clad Laminates (FCCL) and advanced composite materials that must withstand rigorous thermal cycling and mechanical stress. The disclosed technology represents a significant leap forward in aromatic polyvalent primary amine synthesis, offering a robust pathway that bypasses the complexities often associated with multi-step functional group transformations. By leveraging a catalytic transfer hydrogenation strategy, this process achieves remarkable efficiency, delivering product purity exceeding 99% without the need for cumbersome downstream purification steps. For R&D directors and procurement specialists in the electronic materials space, understanding the nuances of this patented route is essential for securing a competitive edge in the supply of high-performance polymer intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of complex aromatic tetra-amines has been plagued by significant operational hurdles that impact both cost and scalability. Conventional reduction methods often rely on high-pressure catalytic hydrogenation using molecular hydrogen, which necessitates specialized, expensive reactor infrastructure and stringent safety protocols to manage explosion risks. Furthermore, alternative chemical reduction pathways frequently employ stoichiometric amounts of metal powders, such as iron or zinc, in acidic media, generating massive quantities of hazardous sludge that complicate waste treatment and environmental compliance. These legacy processes often suffer from poor selectivity, leading to the formation of azo-byproducts or incomplete reduction species that are difficult to separate from the desired amine. The resulting crude products typically require multiple recrystallization cycles or column chromatography to reach the purity standards required for electronic-grade polyimides, drastically increasing processing time and solvent consumption. Such inefficiencies create bottlenecks in the supply chain, making it challenging to meet the growing demand for high-purity electronic chemical intermediates in a cost-effective manner.
The Novel Approach
In stark contrast, the methodology outlined in patent CN101245027A presents a streamlined, industrially viable solution that addresses these historical pain points through elegant process design. This novel approach utilizes hydrazine hydrate as the reducing agent in conjunction with a palladium on carbon (Pd/C) catalyst within a saturated fatty alcohol solvent system. This combination facilitates a mild yet highly effective reduction of the four nitro groups on the precursor molecule to amino groups under atmospheric pressure conditions. The reaction proceeds smoothly at temperatures between 70°C and 85°C, completing within a short timeframe of 1 to 5 hours, which significantly enhances throughput compared to traditional batch processes. A key innovation lies in the workup procedure; the catalyst is removed via simple hot filtration, and the product is isolated by cooling the mother liquor and inducing crystallization with the addition of pure water. This eliminates the need for energy-intensive distillation or complex extraction sequences, directly yielding crystals with purity levels above 99%. This operational simplicity translates directly into lower manufacturing costs and a reduced environmental footprint, positioning this method as a superior choice for the commercial scale-up of complex aromatic amines.
Mechanistic Insights into Pd/C-Catalyzed Transfer Hydrogenation
The core of this synthetic breakthrough relies on the mechanism of catalytic transfer hydrogenation, where hydrazine hydrate serves as the hydrogen donor rather than gaseous hydrogen. In this system, the palladium nanoparticles dispersed on the carbon support act as active sites that facilitate the decomposition of hydrazine into nitrogen gas and reactive hydrogen species adsorbed on the metal surface. These activated hydrogen atoms are then transferred to the nitro groups of the 2,2-bis[3-amino-4-(4-nitrophenoxy)phenyl]propane substrate. The presence of the ether linkages and the central isopropylidene bridge in the molecular structure could potentially pose steric challenges, but the chosen solvent system—comprising saturated fatty alcohols like ethanol or glycols—ensures excellent solubility of the organic substrate while stabilizing the transition states. The reaction conditions are carefully tuned to prevent the reduction of other sensitive functional groups, ensuring chemoselectivity that is crucial for maintaining the integrity of the monomer structure. The evolution of nitrogen gas as the only byproduct from the reducing agent drives the reaction equilibrium forward and simplifies the off-gas handling requirements compared to processes generating ammonia or metal salts.
Controlling impurity profiles is paramount for applications in flexible electronics, where trace metal ions or organic contaminants can degrade the dielectric properties of the final polyimide film. This process inherently minimizes impurity formation through the use of a heterogeneous catalyst that is physically separated from the product stream via hot filtration. By filtering the reaction mixture while hot, the solubility of the product is maintained in the mother liquor while the solid catalyst and any insoluble byproducts are retained on the filter medium. Subsequent cooling and the addition of water reduce the solubility of the target amine, causing it to precipitate as high-purity crystals. This crystallization step acts as a powerful purification mechanism, excluding soluble impurities and residual hydrazine from the crystal lattice. The result is a product with a purity of greater than 99%, as demonstrated in the patent examples, which meets the stringent specifications required for high-performance polymer synthesis without the need for further refining. This level of intrinsic purity control reduces the burden on quality control laboratories and ensures consistent batch-to-batch reproducibility.
How to Synthesize 2,2-bis[3-amino-4-(4-aminophenoxy)phenyl]propane Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and maintain safety standards. The process begins by charging a reactor with the nitro-precursor, a specific loading of Pd/C catalyst (ranging from 1% to 15% palladium content), and a selected saturated fatty alcohol solvent. The mixture is heated to approximately 60°C before the gradual addition of hydrazine hydrate solution, which acts as the reducing agent. Maintaining the reaction temperature within the optimal window of 70°C to 85°C is critical to ensure complete conversion while preventing thermal degradation or runaway reactions. Detailed standardized operating procedures regarding reagent ratios, addition rates, and crystallization protocols are essential for successful technology transfer and scale-up.
- Charge the reactor with the nitro-precursor, Pd/C catalyst, and a saturated fatty alcohol solvent system.
- Heat the mixture to 60°C and slowly add hydrazine hydrate solution while maintaining temperature between 70°C and 85°C.
- Filter the hot reaction mixture to remove the catalyst, cool the mother liquor, and induce crystallization by adding pure water.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis route offers tangible strategic benefits that extend beyond mere technical feasibility. The shift from high-pressure hydrogenation to atmospheric hydrazine reduction fundamentally alters the capital expenditure profile of the manufacturing facility, removing the need for costly high-pressure vessels and associated safety systems. This reduction in infrastructure complexity allows for more flexible production scheduling and lowers the barrier to entry for scaling up production capacity. Furthermore, the simplified workup procedure, which avoids complex solvent exchanges or extensive washing steps, results in significant reductions in utility consumption and labor hours per kilogram of product. These operational efficiencies translate into a more resilient supply chain capable of responding rapidly to market fluctuations in the electronics and aerospace sectors.
- Cost Reduction in Manufacturing: The elimination of expensive high-pressure equipment and the reduction in processing steps directly contribute to substantial cost savings in polyimide manufacturing. By utilizing a heterogeneous catalyst that can be filtered and potentially regenerated, and avoiding the generation of heavy metal waste sludge typical of iron/zinc reductions, the overall cost of goods sold is optimized. The high yield reported in the patent examples indicates efficient raw material utilization, minimizing waste and maximizing the output from each batch. Additionally, the use of common, commercially available solvents like ethanol and glycols ensures that raw material costs remain stable and predictable, shielding the supply chain from volatility associated with exotic reagents.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials and reagents significantly enhances the reliability of the supply chain for this critical electronic chemical intermediate. Hydrazine hydrate and Pd/C are commodity chemicals with robust global supply networks, reducing the risk of production stoppages due to raw material shortages. The short reaction time of 1 to 5 hours allows for higher asset turnover, meaning manufacturers can produce more batches in a given timeframe to meet surging demand. This agility is crucial for supporting the fast-paced development cycles of consumer electronics and automotive industries, where delays in material delivery can halt entire production lines.
- Scalability and Environmental Compliance: From an environmental perspective, this process offers a cleaner alternative to traditional methods, aligning with increasingly strict global regulations on industrial emissions and waste disposal. The primary byproduct of the reduction is nitrogen gas, which is harmless and easily vented, avoiding the release of toxic ammonia or volatile organic compounds associated with other reduction techniques. The minimization of aqueous waste streams, particularly those contaminated with heavy metals, simplifies wastewater treatment and reduces the environmental liability of the manufacturing site. This eco-friendly profile not only ensures compliance but also enhances the brand value of the end-products by supporting sustainable manufacturing practices in the high-tech sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these aspects is vital for stakeholders evaluating the feasibility of integrating this monomer into their supply chains. The answers are derived directly from the technical disclosures and beneficial effects described in the patent documentation, providing a reliable basis for decision-making.
Q: What is the primary advantage of using hydrazine hydrate over hydrogen gas for this reduction?
A: Hydrazine hydrate offers superior operational safety and eliminates the need for high-pressure hydrogenation equipment, significantly reducing capital expenditure and safety risks in large-scale manufacturing.
Q: How does this method ensure high purity without recrystallization?
A: The process utilizes a hot filtration step to remove the heterogeneous catalyst followed by controlled water-induced crystallization, which inherently excludes impurities and achieves purity levels exceeding 99%.
Q: Is this synthesis scalable for industrial polyimide production?
A: Yes, the patent explicitly describes the method as suitable for industrial production due to its simple operation, short reaction time (1-5 hours), and minimal waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2-bis[3-amino-4-(4-aminophenoxy)phenyl]propane Supplier
As the demand for high-performance polyimides continues to surge in the flexible electronics and aerospace industries, securing a dependable source of high-purity monomers is more critical than ever. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging deep expertise in process chemistry to deliver complex aromatic amines that meet the most rigorous industry standards. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your needs from pilot trials to full-scale manufacturing. We operate with stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 2,2-bis[3-amino-4-(4-aminophenoxy)phenyl]propane delivers the consistent performance required for next-generation FCCL and composite materials.
We invite you to collaborate with us to optimize your material sourcing strategy and achieve significant efficiencies in your production operations. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out to request specific COA data and route feasibility assessments, allowing you to validate our capabilities against your internal benchmarks. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain partner committed to innovation, quality, and long-term reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
