Advanced Purification Technology for 2,5-Diarylamine Terephthalic Acid Enabling Commercial Scale Production
Advanced Purification Technology for 2,5-Diarylamine Terephthalic Acid Enabling Commercial Scale Production
The global demand for high-performance organic pigments, particularly quinacridone derivatives, necessitates intermediates of exceptional purity and consistency. Patent CN101823977A introduces a transformative separating and refining method for 2,5-diarylamine terephthalic acid (DXTA), a critical precursor in this value chain. This technology addresses the longstanding challenges of impurity control and solvent waste associated with traditional purification routes. By shifting the paradigm from organic solvent recrystallization to an aqueous-based salt formation and acidification process, the patent outlines a pathway to achieve mass content greater than 99.5%. For R&D directors and procurement strategists, this represents a pivotal opportunity to optimize the supply chain for high-grade pigment intermediates, ensuring both economic efficiency and environmental compliance in modern chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of complex aromatic amines like 2,5-diarylamine terephthalic acid has relied heavily on recrystallization using volatile organic solvents. These conventional methods often suffer from significant drawbacks, including high energy consumption for solvent recovery and the generation of hazardous waste streams that require costly treatment. Furthermore, organic solvents may not always provide the selectivity needed to remove specific colored impurities or isomeric by-products, leading to variability in the final product quality. This inconsistency is particularly detrimental in the pigment industry, where even trace impurities can drastically alter the hue, lightfastness, and thermal stability of the final quinacridone pigment. Consequently, manufacturers face increased operational risks and higher compliance costs due to stringent environmental regulations regarding VOC emissions.
The Novel Approach
The methodology described in patent CN101823977A offers a robust alternative by utilizing the distinct solubility properties of DXTA salts in aqueous media. Instead of relying on organic solvents, the process dissolves the crude product in an inorganic base solution, converting the acid into a highly soluble salt. This phase transfer allows for effective separation of insoluble impurities and facilitates the use of adsorption agents to capture dissolved contaminants. Subsequent acidification precipitates the pure product, leaving inorganic salts in the solution. This approach not only simplifies the operational workflow but also drastically reduces the environmental footprint by minimizing organic waste. The ability to recycle the mother liquor and regenerate adsorption agents further enhances the economic viability, making it a superior choice for sustainable industrial production.
Mechanistic Insights into Aqueous Salt-Based Recrystallization
The core of this purification strategy lies in the reversible acid-base chemistry of the terephthalic acid derivative. Under alkaline conditions, typically achieved with sodium hydroxide, potassium hydroxide, or ammonia, the carboxylic acid groups of the DXTA molecule are deprotonated to form water-soluble carboxylate salts. This transformation is critical because it allows the target molecule to enter the aqueous phase while leaving many non-polar organic impurities behind or rendering them susceptible to adsorption. The process operates at elevated temperatures, ranging from 40°C to 120°C, which further enhances the solubility of the salt and ensures complete dissolution of the crude material. This thermal energy input is carefully balanced to maximize yield without promoting hydrolysis or degradation of the sensitive amine linkages.
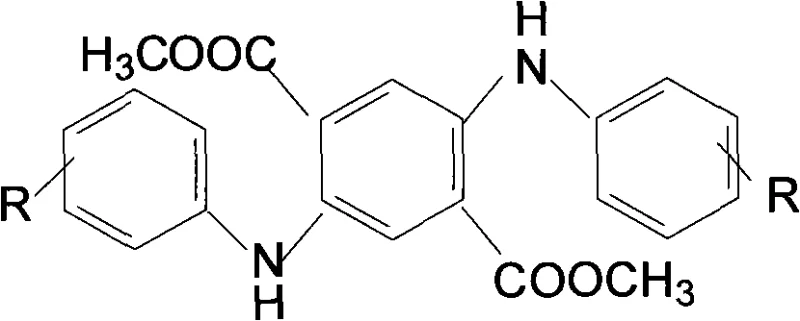
Following dissolution, the introduction of adsorption agents such as activated carbon, molecular sieves, or diatomite plays a pivotal role in impurity profiling. These agents selectively bind to colored by-products and trace organic contaminants that could otherwise compromise the chromatic properties of the final pigment. Once the solution is clarified through solid-liquid separation, the pH is meticulously adjusted to between 2 and 6 using inorganic acids like sulfuric or hydrochloric acid. This acidification step reprotonates the carboxylate groups, causing the DXTA to precipitate out of the solution as a high-purity crystalline solid. The differential solubility of the inorganic salt by-products ensures they remain in the mother liquor, allowing for their easy removal and potential recycling, thereby closing the loop on material usage.
How to Synthesize 2,5-Diarylamine Terephthalic Acid Efficiently
Implementing this purification protocol requires precise control over reaction parameters to ensure reproducibility and high yield. The process begins with the preparation of the alkaline dissolution mixture, where the molar ratio of base to crude acid is maintained between 2.05:1 and 2.2:1 to ensure complete conversion to the salt form. Operators must monitor temperature and pressure closely, as these variables influence both the dissolution rate and the efficiency of the subsequent adsorption step. The choice of adsorption agent and its loading ratio are also critical; for instance, activated carbon is highly effective for decolorization, while molecular sieves may be preferred for removing specific polar impurities. Detailed standard operating procedures for each stage, from dissolution to final drying at 80°C to 120°C, are essential for maintaining product specifications.
- Dissolve crude 2,5-diarylamine terephthalic acid in an aqueous inorganic base solution at elevated temperatures (40-120°C) to form a soluble salt.
- Treat the alkaline solution with adsorption agents like activated carbon or molecular sieves to remove colored impurities and by-products.
- Acidify the purified mother liquor with inorganic acid to pH 2-6 to precipitate the high-purity crystalline product, followed by filtration and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this aqueous purification technology translates into tangible strategic benefits beyond mere technical performance. The shift away from expensive and regulated organic solvents towards water and common inorganic reagents significantly lowers the raw material cost base. Moreover, the simplicity of the equipment requirements—standard stirred reactors, centrifuges, and filter presses—means that the barrier to entry for scaling production is low. This accessibility ensures a more resilient supply chain, as the technology can be deployed across multiple manufacturing sites without the need for specialized, high-cost infrastructure. The result is a more stable supply of critical intermediates, mitigating the risk of production bottlenecks.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the elimination of solvent recovery units and the reduction of waste disposal fees. By utilizing water as the primary medium and enabling the recycling of mother liquor, the operational expenditure is substantially decreased. The regeneration of adsorption agents further contributes to long-term cost savings, as consumable materials do not need to be constantly replenished. Additionally, the high purity of the final product reduces the need for downstream reprocessing, streamlining the overall manufacturing workflow and enhancing profit margins for pigment producers.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as sodium hydroxide, sulfuric acid, and water ensures that the supply chain is not vulnerable to the volatility of specialty solvent markets. This stability is crucial for maintaining continuous production schedules, especially in times of global supply chain disruption. The robustness of the process also means that quality deviations are minimized, leading to fewer batch rejections and a more predictable delivery timeline for customers. This reliability fosters stronger partnerships between intermediate suppliers and downstream pigment manufacturers.
- Scalability and Environmental Compliance: From an environmental perspective, the process aligns perfectly with green chemistry principles by minimizing hazardous waste generation. The aqueous nature of the effluent simplifies wastewater treatment, reducing the burden on environmental compliance teams. Furthermore, the scalability of the process is proven by its compatibility with standard industrial unit operations, allowing for seamless expansion from pilot scale to multi-ton annual production. This scalability ensures that suppliers can meet growing market demand for high-performance pigments without compromising on sustainability goals.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this purification method is essential for stakeholders evaluating its integration into their supply chains. The following questions address common inquiries regarding the process mechanics, product quality, and industrial feasibility. These answers are derived directly from the technical disclosures in patent CN101823977A, providing a reliable foundation for decision-making. By clarifying these points, we aim to facilitate a smoother transition to this advanced manufacturing technique.
Q: What is the primary advantage of the aqueous salt-based purification method for DXTA?
A: The primary advantage is the elimination of large volumes of organic solvents, significantly reducing environmental impact and operational costs while allowing for the recycling of mother liquor and adsorption agents.
Q: How does this process ensure high purity suitable for quinacridone pigments?
A: By leveraging the solubility difference between the alkaline salt and the acidic form, combined with specific adsorption steps, the process effectively removes trace impurities and colored by-products that degrade pigment quality.
Q: Is this purification method scalable for industrial production?
A: Yes, the process utilizes standard industrial equipment such as stirred reactors, centrifuges, and filter presses, making it highly adaptable for commercial scale-up from pilot plants to multi-ton annual production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,5-Diarylamine Terephthalic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the success of advanced material applications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results of patent CN101823977A can be effectively translated into industrial reality. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2,5-diarylamine terephthalic acid supports the superior performance of your final quinacridone pigments. Our expertise in process optimization allows us to navigate the complexities of scale-up while maintaining the highest standards of quality and safety.
We invite you to collaborate with us to explore how this innovative purification technology can enhance your product portfolio and reduce your manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. By partnering with us, you gain access to specific COA data and route feasibility assessments that will empower you to make informed strategic decisions. Contact us today to discuss how we can support your supply chain with reliable, high-quality chemical solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
