Industrial Scale-Up of 1-Phenyl-5-Mercaptotetrazole via Safe Sulfonylation Pathways
Industrial Scale-Up of 1-Phenyl-5-Mercaptotetrazole via Safe Sulfonylation Pathways
The pharmaceutical industry constantly seeks robust synthetic routes for critical intermediates, particularly those serving the high-volume statin drug market. Patent CN113372287B introduces a transformative methodology for the preparation of 1-phenyl-5-mercaptotetrazole, a pivotal building block in the synthesis of HMG-CoA reductase inhibitors. This innovation addresses long-standing safety and efficiency bottlenecks by utilizing a novel two-step sequence that bypasses hazardous reagents traditionally associated with tetrazole functionalization. The target molecule, characterized by its unique heterocyclic structure containing both sulfur and nitrogen moieties, serves as a nucleophilic center for subsequent coupling reactions in complex drug assembly. 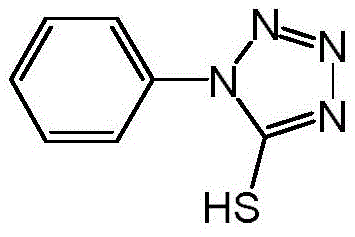 By leveraging a sulfonylation-thiolation strategy, this patent provides a reliable pharmaceutical intermediate supplier with a distinct competitive edge, ensuring consistent quality and supply continuity for downstream API manufacturers who demand rigorous adherence to safety and environmental standards.
By leveraging a sulfonylation-thiolation strategy, this patent provides a reliable pharmaceutical intermediate supplier with a distinct competitive edge, ensuring consistent quality and supply continuity for downstream API manufacturers who demand rigorous adherence to safety and environmental standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-phenyl-5-mercaptotetrazole has been plagued by significant safety hazards and operational complexities that hinder efficient commercial scale-up of complex pharmaceutical intermediates. One prevalent prior art method relies on the reaction of carbon disulfide with aniline, a process notorious for generating toxic sulfide flue gases that pose severe health risks to plant personnel and require extensive scrubbing infrastructure. Another common route involves the use of phenylthioisocyanate reacting with sodium azide; however, phenylthioisocyanate is not only toxic and flammable but also exhibits strong irritant properties, necessitating costly containment measures and specialized handling protocols. Furthermore, these traditional pathways often involve multiple purification steps and harsh reaction conditions that can lead to variable impurity profiles, complicating the regulatory approval process for the final active pharmaceutical ingredient. The reliance on such dangerous precursors creates a fragile supply chain vulnerable to regulatory shutdowns and increases the overall cost burden due to necessary safety mitigations.
The Novel Approach
In stark contrast, the methodology disclosed in CN113372287B offers a streamlined, catalyst-free alternative that fundamentally reshapes the production landscape for this key intermediate. By initiating the synthesis with 1-phenyl-5-hydroxytetrazole, a stable and readily accessible starting material, the process avoids the immediate introduction of volatile sulfur sources. The innovation lies in the strategic activation of the hydroxyl group via sulfonylation, creating a highly reactive sulfonate ester intermediate that is primed for nucleophilic attack. This intermediate is then subjected to a thiolation reaction using benign reagents such as thiourea or sodium hydrosulfide, effectively swapping the oxygen functionality for the desired sulfur group under mild reflux conditions. This approach not only eliminates the need for toxic isocyanates but also simplifies the workup procedure, allowing for high-purity isolation through standard extraction and distillation techniques. The result is a process that delivers yields exceeding 90 percent while drastically reducing the environmental footprint and operational risk profile.
Mechanistic Insights into Sulfonylation-Thiolation Cascade
The core of this synthetic breakthrough relies on a precise two-stage mechanistic sequence that ensures high conversion rates without the need for transition metal catalysis. The first stage involves the activation of the tetrazole ring, where 1-phenyl-5-hydroxytetrazole reacts with a sulfonyl chloride derivative in the presence of an acid-binding agent like triethylamine. 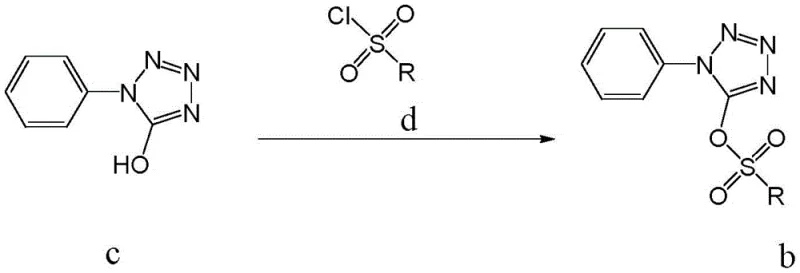 This nucleophilic substitution transforms the poor leaving hydroxyl group into an excellent leaving group, specifically a sulfonate ester, which stabilizes the tetrazole ring against degradation while increasing its electrophilicity at the 5-position. The choice of sulfonyl chloride, whether it be p-toluenesulfonyl chloride, methanesulfonyl chloride, or others, allows for fine-tuning of the intermediate's reactivity, ensuring that the subsequent step proceeds smoothly. The absence of metal catalysts in this activation step is crucial for pharmaceutical applications, as it prevents the introduction of heavy metal impurities that would otherwise require expensive scavenging processes later in the manufacturing line.
This nucleophilic substitution transforms the poor leaving hydroxyl group into an excellent leaving group, specifically a sulfonate ester, which stabilizes the tetrazole ring against degradation while increasing its electrophilicity at the 5-position. The choice of sulfonyl chloride, whether it be p-toluenesulfonyl chloride, methanesulfonyl chloride, or others, allows for fine-tuning of the intermediate's reactivity, ensuring that the subsequent step proceeds smoothly. The absence of metal catalysts in this activation step is crucial for pharmaceutical applications, as it prevents the introduction of heavy metal impurities that would otherwise require expensive scavenging processes later in the manufacturing line.
Following the formation of the activated intermediate, the second stage executes the critical sulfur insertion via a thiolation mechanism.  In this step, the sulfonate intermediate undergoes nucleophilic attack by a sulfur source, such as thiourea, which displaces the sulfonate group to form an isothiouronium salt transiently. Subsequent alkaline hydrolysis cleaves this salt to release the free thiol group, yielding the final 1-phenyl-5-mercaptotetrazole product. The patent specifies that this transformation can be conducted in an ethanol-water solvent system under nitrogen atmosphere, which prevents oxidation of the sensitive thiol group to disulfides. This mechanistic pathway is inherently robust, as the leaving group ability of the sulfonate ensures rapid reaction kinetics even at moderate temperatures, thereby minimizing side reactions and preserving the integrity of the tetrazole ring. The result is a clean reaction profile that facilitates straightforward purification and ensures the final product meets stringent purity specifications required for statin synthesis.
In this step, the sulfonate intermediate undergoes nucleophilic attack by a sulfur source, such as thiourea, which displaces the sulfonate group to form an isothiouronium salt transiently. Subsequent alkaline hydrolysis cleaves this salt to release the free thiol group, yielding the final 1-phenyl-5-mercaptotetrazole product. The patent specifies that this transformation can be conducted in an ethanol-water solvent system under nitrogen atmosphere, which prevents oxidation of the sensitive thiol group to disulfides. This mechanistic pathway is inherently robust, as the leaving group ability of the sulfonate ensures rapid reaction kinetics even at moderate temperatures, thereby minimizing side reactions and preserving the integrity of the tetrazole ring. The result is a clean reaction profile that facilitates straightforward purification and ensures the final product meets stringent purity specifications required for statin synthesis.
How to Synthesize 1-Phenyl-5-Mercaptotetrazole Efficiently
Implementing this synthesis requires careful attention to stoichiometry and reaction conditions to maximize the benefits of the catalyst-free design. The process begins with the preparation of the sulfonate intermediate by mixing the hydroxytetrazole precursor with a sulfonyl chloride in a dichloromethane solvent system, maintaining temperatures between 20°C and 50°C to control exotherms. Once the activation is complete, the crude intermediate can be carried forward directly or purified via recrystallization to ensure maximum purity before the thiolation step. The subsequent conversion to the mercapto derivative involves refluxing the intermediate with thiourea in an ethanol-water mixture, followed by a base-mediated hydrolysis step using sodium hydroxide. Detailed standardized synthesis steps see the guide below.
- Activate 1-phenyl-5-hydroxytetrazole by reacting it with a sulfonyl chloride (such as p-toluenesulfonyl chloride) in the presence of an acid-binding agent like triethylamine to form the sulfonate intermediate.
- Perform the thiolation reaction by refluxing the sulfonate intermediate with a thio-reagent such as thiourea or sodium hydrosulfide in an ethanol-water mixture under nitrogen atmosphere.
- Complete the synthesis through alkaline hydrolysis, followed by acidification, extraction with dichloromethane, drying, and vacuum distillation to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology represents a significant opportunity to optimize cost structures and mitigate supply risks associated with hazardous chemical handling. By shifting away from volatile and toxic precursors like phenylthioisocyanate, manufacturers can substantially reduce the capital expenditure required for specialized safety infrastructure, such as gas scrubbing systems and explosion-proof reactors. This transition directly translates to cost reduction in API manufacturing, as the simplified process flow reduces utility consumption and labor hours associated with complex safety protocols. Furthermore, the use of commodity chemicals like sulfonyl chlorides and thiourea ensures a stable and diversified supply base, reducing the risk of raw material shortages that often plague niche reagent markets. The high yield and purity achieved by this route also minimize waste generation, aligning with increasingly strict environmental regulations and lowering disposal costs.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the avoidance of hazardous reagents significantly lower the direct material costs per kilogram of product. Since the process does not require heavy metal scavengers or complex purification trains to remove trace metals, the downstream processing costs are drastically simplified. Additionally, the high overall yield exceeding 90 percent means less raw material is wasted, improving the atom economy and further driving down the unit cost of production for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Sourcing stable solid reagents like 1-phenyl-5-hydroxytetrazole and various sulfonyl chlorides is far more reliable than managing the supply of toxic liquids like phenylthioisocyanate, which often face strict transportation restrictions. This stability ensures reducing lead time for high-purity pharmaceutical intermediates, as suppliers can maintain larger inventory buffers without the safety liabilities associated with hazardous materials. The robustness of the reaction conditions also means that production schedules are less likely to be disrupted by minor variations in raw material quality or environmental factors.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard unit operations like reflux, extraction, and distillation that are common in multi-purpose pharmaceutical plants. The absence of toxic gas evolution (such as H2S or CS2 byproducts) simplifies the environmental permitting process and reduces the burden on waste treatment facilities. This makes the technology ideal for commercial scale-up of complex pharmaceutical intermediates, allowing manufacturers to increase capacity from pilot scale to multi-ton production without encountering significant engineering bottlenecks related to safety or emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the patent's experimental data and comparative analysis. These insights are intended to clarify the operational benefits and technical feasibility for potential partners evaluating this technology for their supply chains. Understanding these details is crucial for making informed decisions about integrating this method into existing manufacturing portfolios.
Q: How does this new method improve safety compared to traditional phenylthioisocyanate routes?
A: The traditional route utilizes phenylthioisocyanate, which is highly toxic, flammable, and irritating. The patented method replaces this hazardous reagent with stable sulfonyl chlorides and common thio-reagents like thiourea, significantly reducing operational risks and eliminating the need for specialized containment equipment.
Q: What yields can be expected from this catalyst-free synthesis pathway?
A: The patent data indicates that this novel approach achieves total yields exceeding 90 percent. Specifically, the activation step yields over 99 percent, and the subsequent thiolation maintains high efficiency, making it superior to older multi-step methods that often suffer from cumulative yield losses.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is explicitly designed for industrial scalability. It avoids expensive transition metal catalysts, uses standard solvents like dichloromethane and ethanol, and operates under manageable reflux conditions, ensuring that cost reduction in API manufacturing is achievable without compromising on throughput.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Phenyl-5-Mercaptotetrazole Supplier
As a leading CDMO partner, NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 1-phenyl-5-mercaptotetrazole meets the exacting standards required for statin drug synthesis. We understand the critical nature of this intermediate in the global pharmaceutical supply chain and are committed to delivering consistent quality and reliability. Our technical team is well-versed in the nuances of heterocyclic chemistry and sulfur-containing compounds, allowing us to troubleshoot and optimize processes rapidly to meet your specific timeline requirements.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project needs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that demonstrates exactly how adopting this safer, more efficient synthesis route can impact your bottom line. Let us help you secure a sustainable and cost-effective supply of this vital intermediate, enabling you to focus on your core drug development goals while we manage the complexities of chemical manufacturing.
