Scalable Photocatalytic Production of Alpha-Azido Ketones for Advanced Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking greener, safer, and more efficient pathways to synthesize complex heterocyclic precursors. A significant breakthrough in this domain is documented in Chinese Patent CN108586283B, which details a novel method for the preparation of α-azido ketone compounds based on visible-light photocatalysis. This technology represents a paradigm shift from traditional hazardous synthetic routes to a sustainable, metal-free protocol that utilizes air as the terminal oxidant. For R&D directors and procurement managers alike, this innovation offers a compelling value proposition by drastically simplifying the operational complexity associated with azide chemistry. By leveraging organic photocatalysts such as Rose Bengal alongside a selenium-based additive, the process eliminates the need for stoichiometric inorganic oxidants and pre-functionalized halogenated starting materials. This not only enhances the safety profile of the manufacturing process but also aligns perfectly with modern environmental compliance standards, making it an ideal candidate for the reliable production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of α-azido ketones has been fraught with significant safety hazards and environmental burdens. The most common conventional route involves the nucleophilic substitution of α-haloketones with sodium azide. As illustrated in the reaction scheme below, this approach necessitates the prior preparation of α-haloketones using aggressive halogenating agents, which generates substantial amounts of halogen-containing waste streams that are difficult and costly to treat. Furthermore, the handling of organic azides and sodium azide on a large scale presents severe explosion risks, requiring specialized equipment and rigorous safety protocols that drive up capital expenditure. Alternative methods employing hypervalent iodine reagents or chromium-based oxidants suffer from similar drawbacks, including the use of expensive, stoichiometric oxidants and the generation of heavy metal waste. These factors collectively result in a high cost of goods sold (COGS) and a complex supply chain for critical API intermediates, often leading to supply disruptions and extended lead times for downstream drug manufacturers.
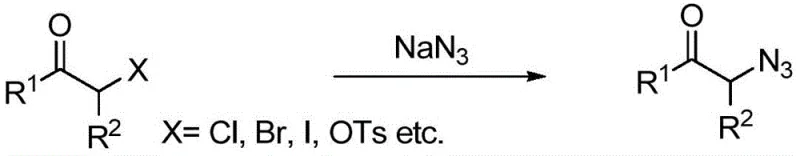
The Novel Approach
In stark contrast to these legacy methods, the photocatalytic strategy outlined in the patent introduces a streamlined, one-step oxidative azidation of simple alkenes. This novel approach bypasses the need for pre-halogenation entirely, utilizing readily available styrene derivatives and azidotrimethylsilane (TMSN3) as direct starting materials. The reaction is driven by visible light irradiation, typically using energy-efficient blue LEDs, and employs atmospheric oxygen as the sole oxidant, thereby eliminating the cost and waste associated with chemical oxidants like chromium trioxide or ceric ammonium nitrate. The use of an organic photocatalyst system ensures that the process remains free from heavy metal contamination, a critical quality attribute for pharmaceutical applications. This methodology not only reduces the number of synthetic steps but also operates under mild room temperature conditions, significantly lowering energy consumption and enhancing the overall safety of the chemical transformation for commercial scale-up of complex pharmaceutical intermediates.

Mechanistic Insights into Visible-Light Photocatalytic Oxidative Azidation
The core of this technological advancement lies in the synergistic interaction between the organic photocatalyst and the selenium additive under visible light irradiation. Upon exposure to blue LED light, the photocatalyst, such as Rose Bengal, enters an excited state capable of engaging in single-electron transfer (SET) processes or energy transfer mechanisms. This excited species activates the molecular oxygen present in the air, generating reactive oxygen species that are crucial for the oxidation of the alkene substrate. Simultaneously, the diphenyl diselenide additive plays a pivotal role in facilitating the azidation step, likely by forming a transient selenonium intermediate that activates the alkene towards nucleophilic attack by the azide source. This dual-catalytic system ensures high regioselectivity and efficiency, converting the carbon-carbon double bond directly into the carbonyl-azide motif without over-oxidation or side reactions. The mechanistic elegance of this system allows for broad substrate tolerance, accommodating various electronic environments on the aromatic ring, which is essential for synthesizing diverse libraries of bioactive molecules.
From an impurity control perspective, this mechanism offers distinct advantages over traditional radical halogenation pathways. Since the reaction does not involve free radical halogen species or harsh acidic conditions, the formation of poly-halogenated byproducts or rearrangement impurities is minimized. The mild nature of the photocatalytic cycle preserves sensitive functional groups that might otherwise be degraded by strong oxidants like IBX or chromium reagents. Furthermore, the use of TMSN3 as the azide source, rather than ionic sodium azide, helps to moderate the reactivity of the azide moiety, reducing the risk of uncontrolled exothermic events. The resulting crude reaction mixtures are generally cleaner, simplifying the downstream purification process and ensuring that the final high-purity pharmaceutical intermediates meet stringent regulatory specifications for residual metals and genotoxic impurities, thereby reducing the burden on quality control laboratories.
How to Synthesize Alpha-Azido Ketones Efficiently
The practical implementation of this photocatalytic method is designed to be straightforward and adaptable to standard laboratory and pilot plant equipment. The process begins by combining the alkene substrate and azidotrimethylsilane in a suitable organic solvent such as acetonitrile, followed by the addition of catalytic amounts of Rose Bengal and diphenyl diselenide. The reaction vessel is then subjected to visible light irradiation under an air atmosphere, allowing the oxidative azidation to proceed to completion within a few hours at ambient temperature. This operational simplicity makes it an attractive option for process chemists looking to optimize synthetic routes. For a detailed breakdown of the specific reagent ratios, lighting conditions, and workup procedures validated in the patent, please refer to the standardized synthesis guide below.
- Mix alkene substrate, azidotrimethylsilane (TMSN3), Rose Bengal photocatalyst, and diphenyl diselenide additive in acetonitrile solvent.
- Irradiate the reaction mixture with a 3W blue LED lamp under air atmosphere at room temperature for approximately 2 hours.
- Concentrate the reaction mixture under vacuum and purify the crude product via silica gel column chromatography to obtain the alpha-azido ketone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology translates into tangible strategic benefits regarding cost stability and supply security. By shifting away from stoichiometric oxidants and pre-functionalized halogenated intermediates, manufacturers can significantly reduce the raw material costs associated with producing α-azido ketones. The elimination of expensive metal reagents and the ability to use air as a free oxidant directly contributes to cost reduction in pharmaceutical intermediates manufacturing. Moreover, the simplified workflow reduces the dependency on complex multi-step supply chains for specialized starting materials, thereby enhancing supply chain reliability and mitigating the risk of bottlenecks caused by the scarcity of specific halogenated precursors. This resilience is crucial for maintaining continuous production schedules in the face of global market fluctuations.
- Cost Reduction in Manufacturing: The transition to a catalytic system driven by visible light and air eliminates the recurring expense of purchasing stoichiometric quantities of high-valent iodine reagents, chromium oxidants, or halogenating agents. This fundamental shift in reagent consumption leads to substantial cost savings per kilogram of product. Additionally, the removal of heavy metal catalysts negates the need for expensive metal scavenging steps and rigorous testing for residual metals, further streamlining the production budget. The energy efficiency of using low-power LED lamps compared to high-temperature reflux conditions also contributes to lower utility costs, making the overall process economically superior to conventional thermal methods.
- Enhanced Supply Chain Reliability: Utilizing simple alkenes and TMSN3 as starting materials diversifies the supply base, as these commodities are widely available from multiple global suppliers compared to specialized alpha-haloketones. This abundance reduces the lead time for high-purity pharmaceutical intermediates and minimizes the risk of production stoppages due to raw material shortages. The robustness of the reaction conditions, which tolerate air and moisture better than sensitive organometallic protocols, ensures consistent batch-to-batch quality and yield. Consequently, manufacturers can offer more reliable delivery schedules to their clients, strengthening long-term partnerships and contractual agreements in the competitive fine chemical market.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this method, such as atom economy and the use of benign solvents, facilitate easier regulatory approval and environmental compliance. The absence of toxic heavy metal waste simplifies wastewater treatment protocols and reduces the environmental footprint of the manufacturing facility. This alignment with sustainability goals is increasingly becoming a prerequisite for partnerships with major multinational pharmaceutical companies. Furthermore, the mild reaction conditions and lack of explosive hazards associated with handling large quantities of ionic azides make the process inherently safer to scale from kilogram to tonnage levels, ensuring a secure and compliant supply of critical chemical building blocks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic technology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on process feasibility and product quality. Understanding these details is essential for stakeholders evaluating the integration of this method into their existing manufacturing portfolios.
Q: What are the safety advantages of this photocatalytic method compared to traditional alpha-haloketone substitution?
A: Traditional methods require pre-synthesis of hazardous alpha-haloketones and use stoichiometric sodium azide, posing explosion risks. This photocatalytic method uses air as the oxidant and avoids heavy metal reagents, significantly improving process safety and reducing toxic waste.
Q: Can this method be scaled for commercial production of pharmaceutical intermediates?
A: Yes, the reaction operates at room temperature using visible light and common solvents like acetonitrile. The absence of expensive stoichiometric oxidants and the use of air make it highly suitable for cost-effective commercial scale-up.
Q: What is the role of diphenyl diselenide in this reaction system?
A: Diphenyl diselenide acts as a crucial additive that works synergistically with the Rose Bengal photocatalyst to facilitate the oxidation and azidation of the alkene substrate under mild visible light conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Azido Ketone Supplier
The technological potential of visible-light photocatalysis for synthesizing α-azido ketones is immense, offering a pathway to safer and more sustainable chemical manufacturing. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced photocatalytic reactors and stringent purity specifications are maintained through our rigorous QC labs, guaranteeing that every batch of Alpha-Azido Ketone meets the highest international standards for pharmaceutical applications. We are committed to delivering high-purity pharmaceutical intermediates that empower our clients to accelerate their drug development pipelines with confidence.
We invite you to explore how this cutting-edge synthesis method can optimize your supply chain and reduce production costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your project goals. Let us partner with you to bring safer, greener, and more efficient chemical solutions to the global market.
