Scalable Synthesis of 2,6-Bis(2,4-Dinitrophenoxy)Toluene for High-Performance Polyimide Applications
The global demand for high-performance polyimide materials in the aerospace and electronic display sectors has necessitated the development of robust synthetic routes for key monomers. Patent CN101234982B introduces a highly efficient method for preparing 2,6-bis(2,4-dinitrophenoxy)toluene, a critical precursor for highly branched aromatic polyimides. This technology addresses the longstanding challenges of achieving both high purity and high yield in the synthesis of complex aromatic ethers. By leveraging a specific nucleophilic aromatic substitution strategy, the process ensures the production of materials that meet the stringent thermal and mechanical requirements of liquid crystal displays (LCDs) and advanced composite materials. For R&D directors and procurement specialists, understanding this pathway is essential for securing a reliable polyimide intermediate supplier capable of delivering consistent quality at scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for aromatic polyimide monomers often suffer from significant drawbacks that hinder large-scale commercial viability. Many conventional methods rely on harsh reaction conditions that require specialized high-pressure equipment, leading to increased capital expenditure and safety risks. Furthermore, older protocols frequently utilize stoichiometric amounts of expensive phase-transfer catalysts or transition metals, which not only inflate the raw material costs but also introduce difficult-to-remove metal impurities into the final product. These metallic residues can be detrimental to the electrical properties of the resulting polyimide films, particularly in microelectronics applications where dielectric integrity is paramount. Additionally, traditional workup procedures often involve complex extraction sequences that generate substantial volumes of hazardous waste, creating environmental compliance burdens and increasing disposal costs for manufacturing facilities.
The Novel Approach
The methodology described in CN101234982B represents a paradigm shift towards greener and more economical manufacturing. This novel approach eliminates the need for exotic catalysts by optimizing the basicity of the reaction medium through the careful selection of inorganic salt-forming agents such as potassium carbonate or sodium hydroxide. The process operates under atmospheric pressure, significantly reducing equipment complexity and maintenance requirements. A key innovation lies in the solvent system design, which combines water-insoluble organic solvents with polar aprotic co-solvents to facilitate azeotropic water removal. This allows the reaction to proceed to completion with high conversion rates while enabling the straightforward recovery and recycling of solvents. Consequently, this method offers substantial cost savings in electronic chemical manufacturing by minimizing waste generation and maximizing resource efficiency.
Mechanistic Insights into Nucleophilic Aromatic Substitution
The core chemical transformation in this synthesis is a classic nucleophilic aromatic substitution (SnAr) reaction, driven by the strong electron-withdrawing nature of the nitro groups on the benzene ring. In this mechanism, the phenoxide anion, generated in situ by the deprotonation of 2,6-dihydroxytoluene by the base, acts as a potent nucleophile. This anion attacks the electron-deficient aromatic ring of the 2,4-dinitrohalobenzene at the position ortho or para to the nitro groups, where the electron density is lowest. The presence of two nitro groups significantly activates the ring towards nucleophilic attack, stabilizing the negatively charged Meisenheimer complex intermediate. The subsequent elimination of the halide leaving group (fluorine, chlorine, bromine, or iodine) restores aromaticity and forms the stable ether linkage. The reaction kinetics are further enhanced by the use of polar aprotic solvents, which solvate the cation of the base but leave the phenoxide anion relatively naked and highly reactive.
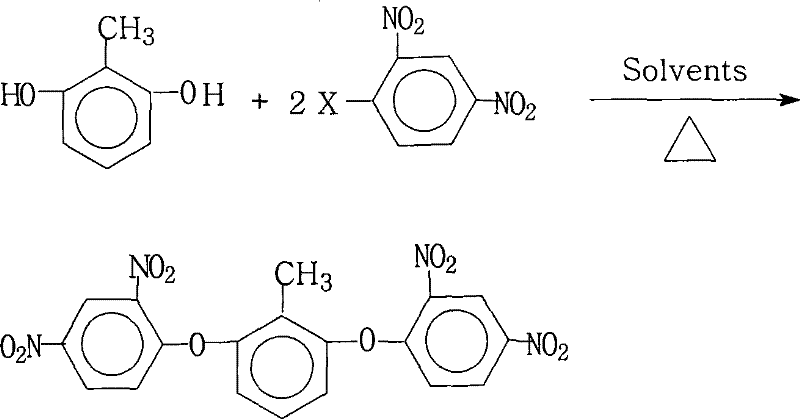
Controlling the impurity profile is critical for ensuring the downstream performance of the polyimide resin. The patent specifies a molar ratio of 2,6-dihydroxytoluene to 2,4-dinitrohalobenzene between 1.0:2.0 and 1.0:2.2. This slight excess of the nitro-compensated electrophile ensures that both hydroxyl groups on the toluene core are fully substituted, preventing the formation of mono-substituted byproducts which could act as chain terminators in the subsequent polymerization step. Furthermore, the reaction temperature is maintained between 80°C and 200°C, a range that provides sufficient thermal energy to overcome the activation barrier for the substitution without promoting thermal decomposition or side reactions such as nucleophilic attack on the nitro groups themselves. The rigorous washing and drying steps post-reaction effectively remove residual inorganic salts and unreacted starting materials, yielding a product with purity levels exceeding 99%, which is essential for high-purity aromatic monomer specifications.
How to Synthesize 2,6-Bis(2,4-Dinitrophenoxy)Toluene Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and minimize cycle time. The process begins with the charging of reactants into a reactor equipped with a water separator, followed by a controlled heating ramp to establish reflux conditions. The removal of water generated during the salt formation and substitution steps is crucial for driving the equilibrium towards the product. Detailed operational parameters, including specific solvent ratios and agitation speeds, are critical for reproducibility. For a comprehensive breakdown of the standardized operating procedures and safety protocols required for this synthesis, please refer to the technical guide below.
- Charge the reactor with 2,6-dihydroxytoluene and 2,4-dinitrohalobenzene in a molar ratio of 1.0: 2.0 to 2.2, along with a salt-forming agent and a mixed solvent system.
- Heat the mixture to reflux between 80°C and 200°C for 3 to 18 hours to facilitate water separation and complete the nucleophilic substitution reaction.
- Concentrate the reaction solution, cool the system, add water to precipitate the solid product, then filter, wash, and dry to obtain the final crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process translates directly into improved bottom-line performance and supply security. The simplicity of the operation means that production can be scaled up rapidly without the need for bespoke reactor designs or extensive operator training. The ability to recycle solvents repeatedly reduces the recurring cost of raw materials and mitigates the volatility associated with solvent pricing markets. Moreover, the use of readily available starting materials like 2,6-dihydroxytoluene and common nitrohalobenzenes ensures that the supply chain is not dependent on single-source or geographically constrained vendors. This diversification of raw material sources significantly enhances supply chain reliability and reduces the risk of production stoppages due to material shortages.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and phase-transfer agents results in a drastic simplification of the bill of materials. By avoiding the use of precious metals, the process removes the need for costly metal scavenging steps and complex purification protocols typically required to meet ppm-level metal specifications. The high atom economy of the reaction, combined with the recovery of solvents, leads to substantial cost savings in polyimide intermediate manufacturing. Furthermore, the high yield reported in the patent examples minimizes the loss of valuable raw materials, ensuring that every kilogram of input generates maximum output value.
- Enhanced Supply Chain Reliability: The robustness of this chemical process allows for consistent batch-to-batch quality, which is vital for maintaining long-term contracts with downstream polymer manufacturers. Since the reaction does not involve sensitive reagents that degrade rapidly upon exposure to air or moisture, inventory management is simplified, and raw materials can be stored for extended periods without significant quality degradation. This stability facilitates better planning and reduces the need for expedited shipping or emergency sourcing, thereby reducing lead time for high-purity polyimide precursors and ensuring a steady flow of materials to production lines.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex aromatic monomers, operating under normal pressure and utilizing standard glass-lined or stainless steel reactors. The reduction in three wastes (waste water, waste gas, and solid waste) aligns with increasingly strict global environmental regulations, reducing the liability and permitting hurdles associated with chemical manufacturing. The environmentally friendly nature of the process also supports corporate sustainability goals, making the final product more attractive to eco-conscious customers in the electronics and automotive sectors who prioritize green supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2,6-bis(2,4-dinitrophenoxy)toluene. These insights are derived directly from the experimental data and beneficial effects outlined in the patent literature, providing a clear picture of the technology's capabilities. Understanding these details helps stakeholders make informed decisions regarding material selection and process integration.
Q: What is the typical yield and purity of this synthesis method?
A: According to patent CN101234982B, the process achieves yields exceeding 90% (e.g., 92.7% in Example 1) with product purity reaching up to 99.7%, making it highly suitable for electronic grade applications.
Q: Which solvents are recommended for this reaction?
A: The process utilizes a mixture of water-insoluble organic solvents (like toluene or xylene) and polar aprotic solvents (such as DMF, DMAc, or NMP) to optimize solubility and facilitate water removal during reflux.
Q: Is this process scalable for industrial production?
A: Yes, the method is designed for industrialization, featuring simple atmospheric pressure operations, recyclable solvents, and minimal waste generation, ensuring robust supply chain continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,6-Bis(2,4-Dinitrophenoxy)Toluene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality monomers play in the performance of next-generation polyimide materials. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements regardless of your project stage. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ advanced analytical techniques to verify identity and assay. Our facility is equipped to handle the specific solvent systems and reaction conditions required for this synthesis, guaranteeing a consistent supply of this vital electronic chemical.
We invite you to collaborate with us to optimize your supply chain for polyimide production. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your R&D and production goals efficiently.
