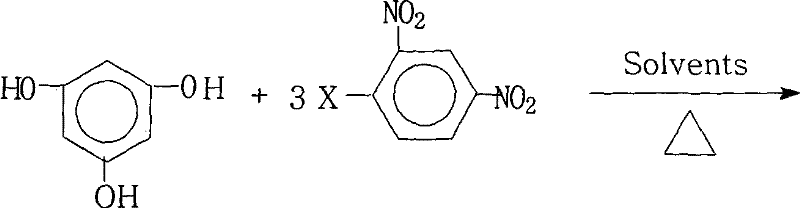
Advanced Synthesis of 1,3,5-Tris(2,4-Dinitrophenoxy)benzene for High-Performance Polyimide Manufacturing
The development of advanced functional materials for the electronics and aerospace sectors relies heavily on the availability of high-purity aromatic intermediates. Patent CN101230005B introduces a robust and scalable method for preparing 1,3,5-tris(2,4-dinitrophenoxy)benzene, a critical precursor for highly branched aromatic polyimides. This specific chemical architecture is indispensable for producing polyimide systems that exhibit superior thermal stability, mechanical strength, and electrical insulation properties required for next-generation liquid crystal displays (LCDs) and microelectronic components. The patented process addresses the historical lack of reported preparation methods for this specific tri-substituted benzene derivative, offering a pathway that combines operational simplicity with industrial viability. By utilizing a nucleophilic aromatic substitution strategy under controlled reflux conditions, the invention ensures high conversion rates while maintaining a clean reaction profile suitable for large-scale manufacturing environments.
For R&D directors and process chemists, the significance of this technology lies in its ability to deliver consistent quality without the need for exotic catalysts or extreme pressure conditions. The synthesis leverages the reactivity of 2,4-dinitrohalobenzenes against phloroglucinol, facilitated by a carefully selected base and solvent system. This approach not only simplifies the purification workflow but also enhances the overall atom economy of the process. As the demand for high-performance polymers grows in the display and semiconductor industries, having a reliable synthetic route for such complex intermediates becomes a strategic asset. The following analysis details the mechanistic advantages and commercial implications of adopting this patented methodology for the production of electronic grade chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of multi-substituted aromatic ethers like 1,3,5-tris(2,4-dinitrophenoxy)benzene has faced significant challenges regarding regioselectivity and reaction completeness. Conventional nucleophilic substitution reactions often suffer from incomplete conversion when attempting to substitute all three hydroxyl groups on the phloroglucinol core, leading to a complex mixture of mono-, di-, and tri-substituted byproducts. Separating these structurally similar impurities is notoriously difficult and costly, often requiring multiple recrystallization steps or preparative chromatography, which drastically reduces the overall yield and increases solvent consumption. Furthermore, traditional methods might employ harsh conditions or stoichiometric amounts of expensive reagents that generate substantial hazardous waste, conflicting with modern green chemistry principles and increasing the environmental compliance burden for manufacturers.
The Novel Approach
The method disclosed in patent CN101230005B overcomes these hurdles through a sophisticated optimization of the reaction medium and stoichiometry. By employing a mixed solvent system comprising both water-insoluble organic solvents and strong polar aprotic solvents, the process creates an ideal environment for the nucleophilic attack while facilitating the removal of water generated during the salt formation. The use of a slight excess of the halogenated reactant (molar ratio 1.0:3.0 to 3.2) ensures that the equilibrium is driven towards the fully substituted tri-ether product, effectively minimizing the formation of partially substituted intermediates. Additionally, the implementation of a heating reflux with water diversion allows for the continuous removal of reaction byproducts, pushing the reaction to completion without the need for excessive temperatures or pressures. This results in a crude product of such high purity that downstream processing is significantly simplified, directly translating to lower operational costs and higher throughput.
Mechanistic Insights into Nucleophilic Aromatic Substitution
The core chemical transformation in this synthesis is a classic nucleophilic aromatic substitution (SnAr), driven by the strong electron-withdrawing nature of the nitro groups on the benzene ring. In the presence of a base such as potassium carbonate or sodium hydroxide, the hydroxyl groups of phloroglucinol are deprotonated to form phenoxide anions, which act as potent nucleophiles. These anions attack the carbon atoms bearing the halogen substituents on the 2,4-dinitrohalobenzene rings. The presence of two nitro groups ortho and para to the leaving group significantly activates the ring towards nucleophilic attack by stabilizing the negatively charged Meisenheimer complex intermediate. This electronic activation is crucial for allowing the reaction to proceed efficiently at moderate temperatures ranging from 80°C to 200°C, avoiding the thermal degradation that might occur under more forcing conditions.
Impurity control in this system is inherently managed by the reaction kinetics and the solubility profiles of the intermediates. The choice of a mixed solvent system, typically involving toluene or xylene combined with dimethylformamide (DMF) or dimethyl sulfoxide (DMSO), plays a dual role. Firstly, the polar aprotic component solvates the ionic intermediates and the base, enhancing the nucleophilicity of the phenoxide. Secondly, the water-insoluble component facilitates the azeotropic removal of water formed during the neutralization of the phenol, preventing the hydrolysis of the halogenated reactant which is a common side reaction in aqueous environments. By strictly controlling the water content through reflux diversion, the process minimizes the formation of 2,4-dinitrophenol byproducts, ensuring that the final crystalline product achieves purity levels exceeding 99 percent, which is critical for electronic applications where trace ionic contaminants can compromise device performance.
How to Synthesize 1,3,5-Tris(2,4-Dinitrophenoxy)benzene Efficiently
The practical execution of this synthesis involves a straightforward sequence of mixing, heating, and isolation steps that are easily adaptable to standard reactor setups found in fine chemical plants. The process begins with the charging of phloroglucinol and the selected 2,4-dinitrohalobenzene into a reactor equipped with a water separator, followed by the addition of the base and the solvent mixture. The reaction is then heated to reflux, allowing the system to self-regulate the temperature while water is continuously removed.
- Mix phloroglucinol and 2,4-dinitrohalobenzene in a molar ratio of 1.0: 3.0-3.2 with a salt-forming agent in a mixed organic solvent system.
- Heat the mixture to reflux (80-200°C) with water separation for 6 to 18 hours to drive the nucleophilic substitution reaction to completion.
- Concentrate the reaction solution, cool, add water to precipitate the solid product, then filter, wash, and dry to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis route offers tangible benefits in terms of cost structure and supply reliability. The elimination of complex catalytic systems and the reliance on commodity chemicals like phloroglucinol and dinitrochlorobenzene means that raw material sourcing is stable and less susceptible to geopolitical fluctuations associated with rare metals or specialized reagents. The simplicity of the workup procedure, which involves concentration and precipitation rather than complex distillation or extraction trains, reduces the energy footprint of the manufacturing process. This efficiency gain allows for a more competitive pricing model for the final intermediate, providing a distinct advantage in cost reduction in electronic chemical manufacturing where margin pressures are constant.
- Cost Reduction in Manufacturing: The process utilizes inexpensive and readily available inorganic bases such as potassium carbonate or sodium carbonate, avoiding the need for costly organic bases or transition metal catalysts. Furthermore, the ability to recover and recycle the organic solvents repeatedly significantly lowers the variable cost per kilogram of product. The high yield achieved in the reaction minimizes the loss of valuable starting materials, ensuring that the theoretical mass balance is closely approached in practice, which is a key driver for optimizing the cost of goods sold (COGS) in high-volume production scenarios.
- Enhanced Supply Chain Reliability: Since the reaction operates under atmospheric pressure and does not involve hazardous gases or highly corrosive reagents, the safety profile of the manufacturing facility is greatly improved. This reduces the risk of unplanned shutdowns due to safety incidents or regulatory inspections, thereby ensuring a consistent and reliable supply of the intermediate to downstream polymer manufacturers. The robustness of the process also means that it can be easily transferred between different manufacturing sites without significant re-engineering, adding flexibility to the global supply network.
- Scalability and Environmental Compliance: The generation of waste is minimized through solvent recycling and the absence of heavy metal contaminants, aligning with increasingly stringent environmental regulations globally. The solid byproduct (inorganic salts) is easy to handle and dispose of, and the aqueous waste stream is relatively clean compared to processes involving strong acids or oxidizers. This environmental compatibility facilitates faster permitting for capacity expansion and reduces the long-term liability associated with waste management, making it a sustainable choice for long-term commercial scale-up of complex electronic materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized intermediate. Understanding these details helps stakeholders evaluate the feasibility of integrating this material into their supply chains.
Q: What are the critical reaction conditions for synthesizing 1,3,5-tris(2,4-dinitrophenoxy)benzene?
A: The process requires a mixed solvent system of water-insoluble solvents (like toluene) and polar aprotic solvents (like DMF), along with a base such as potassium carbonate, under reflux with water removal.
Q: Why is this synthesis method considered environmentally friendly?
A: The method allows for the convenient recycling and repeated use of organic solvents, generates minimal waste, and operates under atmospheric pressure without corrosive byproducts.
Q: What is the primary application of 1,3,5-tris(2,4-dinitrophenoxy)benzene?
A: It serves as a key intermediate for synthesizing highly branched aromatic polyimides, which are essential for high-temperature resistant materials in LCDs and aerospace applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3,5-Tris(2,4-Dinitrophenoxy)benzene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the performance of advanced polyimide materials. Our technical team has extensively studied the pathway described in patent CN101230005B and possesses the expertise to optimize this synthesis for commercial requirements. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of material that meets stringent purity specifications. Our rigorous QC labs are equipped to verify the absence of critical impurities, guaranteeing that every batch supports the high reliability standards of the electronics industry.
We invite you to collaborate with us to secure your supply chain for high-performance polymer precursors. By leveraging our process engineering capabilities, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact our technical procurement team to request specific COA data and route feasibility assessments, and let us demonstrate how our optimized manufacturing processes can enhance your product competitiveness in the global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
