Advanced Synthesis of 1,2-Bis(2,4-Dinitrophenoxy)benzene for High-Performance Polyimide Applications
The global demand for high-performance polymeric materials in the aerospace and microelectronics sectors has necessitated the development of robust synthetic routes for specialized aromatic monomers. Patent CN101234988B introduces a highly efficient preparation method for 1,2-bis(2,4-dinitrophenoxy)benzene, a critical intermediate in the synthesis of highly branched aromatic polyimides. This specific chemical architecture is pivotal for creating materials that withstand extreme thermal and mechanical stress, such as those required for liquid crystal display (LCD) substrates and advanced insulation systems. The disclosed methodology represents a significant leap forward in process chemistry, moving away from cumbersome multi-step sequences toward a streamlined, one-pot nucleophilic aromatic substitution strategy. By optimizing the molar ratios of reactants and employing a sophisticated dual-solvent system, the inventors have achieved a balance between reaction kinetics and thermodynamic equilibrium that was previously difficult to maintain on an industrial scale. This technical breakthrough not only enhances the purity profile of the final crystalline product but also addresses the growing environmental mandates facing modern chemical manufacturing facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex aromatic ethers like 1,2-bis(2,4-dinitrophenoxy)benzene has been plagued by significant operational challenges that hindered widespread commercial adoption. Traditional pathways often relied on harsh reaction conditions, including the use of strong, corrosive bases that demanded specialized alloy reactors capable of withstanding aggressive chemical environments. Furthermore, conventional processes frequently suffered from poor atom economy and generated substantial quantities of hazardous waste streams, complicating downstream purification and disposal logistics. The inability to effectively remove water generated during the condensation reaction often led to reversible hydrolysis, capping yields and resulting in a product laden with unreacted starting materials and side products. These inefficiencies translated directly into higher production costs and inconsistent batch-to-batch quality, creating supply chain vulnerabilities for downstream polymer manufacturers who require stringent specification adherence. Additionally, the reliance on single-solvent systems often made solvent recovery energy-intensive and economically unviable, further eroding the profit margins associated with these high-value intermediates.
The Novel Approach
In stark contrast to these legacy issues, the novel approach detailed in the patent leverages a synergistic mixed-solvent strategy to overcome kinetic barriers and drive the reaction to near-completion. By utilizing a specific blend of water-insoluble organic solvents, such as toluene or xylene, combined with highly polar aprotic solvents like N,N-dimethylformamide (DMF), the process facilitates continuous azeotropic removal of water during the reflux stage. This engineering solution effectively shifts the chemical equilibrium toward the product side without requiring excessive reagent loading or extreme pressures. The method operates under atmospheric pressure at moderate temperatures ranging from 80°C to 200°C, significantly reducing the energy footprint and safety risks associated with high-pressure autoclaves. Moreover, the choice of mild inorganic bases, such as potassium carbonate or sodium bicarbonate, minimizes equipment corrosion and simplifies the workup procedure, allowing for the direct precipitation of high-purity crystals upon cooling and water addition. This holistic optimization of reaction parameters ensures that the process is not only chemically superior but also inherently safer and more environmentally sustainable for large-scale operations.
Mechanistic Insights into Nucleophilic Aromatic Substitution
The core chemical transformation driving this synthesis is a classic nucleophilic aromatic substitution ($S_NAr$), where the electron-rich phenoxide anions derived from catechol attack the electron-deficient aromatic ring of the 2,4-dinitrohalobenzene. The presence of two strongly electron-withdrawing nitro groups at the ortho and para positions relative to the halogen leaving group dramatically activates the benzene ring, lowering the activation energy required for the formation of the Meisenheimer complex intermediate. In this mechanism, the initial step involves the deprotonation of the catechol hydroxyl groups by the added base to generate the reactive nucleophile. This nucleophile then attacks the carbon bearing the halogen atom, forming a resonance-stabilized anionic sigma complex. The subsequent elimination of the halide ion restores aromaticity, yielding the desired diaryl ether linkage. The patent specifies that the halogen leaving group can be fluorine, chlorine, bromine, or iodine, with reactivity generally following the order of F > Cl > Br > I due to the inductive effect enhancing the electrophilicity of the ipso-carbon, despite fluoride typically being a poorer leaving group in aliphatic systems.
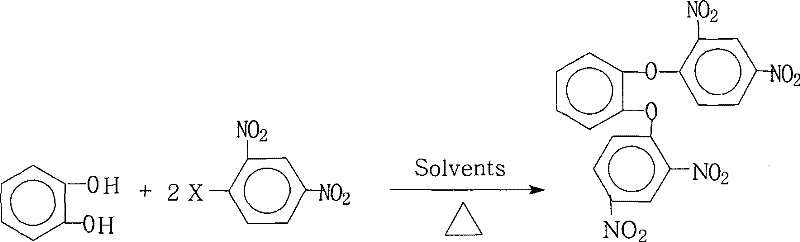
Beyond the fundamental electronic effects, the control of impurities in this reaction is critically dependent on the stoichiometry and the solvent environment. The patent mandates a molar ratio of catechol to dinitrohalobenzene between 1.0:2.0 and 1.0:2.2, ensuring a slight excess of the electrophile to drive the double substitution to completion and prevent the formation of mono-substituted byproducts. The mixed solvent system plays a dual role here: the polar aprotic component solvates the cation of the base, increasing the nucleophilicity of the phenoxide anion (the "naked anion" effect), while the non-polar component aids in the physical separation of water. This precise control over the reaction medium prevents side reactions such as hydrolysis of the nitro groups or oxidative degradation of the catechol, which are common pitfalls in less optimized systems. The result is a crude product with exceptionally high purity, often exceeding 99% prior to recrystallization, which drastically reduces the burden on downstream purification units and ensures the final polymer properties are not compromised by trace ionic contaminants.
How to Synthesize 1,2-Bis(2,4-Dinitrophenoxy)benzene Efficiently
Implementing this synthesis on a production scale requires careful attention to the sequence of addition and temperature profiling to maximize safety and yield. The process begins by charging the reactor with the calculated amounts of catechol, the selected 2,4-dinitrohalobenzene, and the base, followed by the introduction of the pre-mixed solvent system. It is crucial to maintain vigorous stirring to ensure homogeneous suspension of the solid base and efficient heat transfer throughout the viscous reaction mass. As the mixture is heated to reflux, the Dean-Stark apparatus or equivalent water separator must be employed to continuously remove the water of reaction, a step that is non-negotiable for achieving the high conversion rates reported in the patent examples. Following the reaction period, which can range from 3 to 18 hours depending on the specific halogen and temperature used, the solvent is recovered via distillation for reuse, aligning with green chemistry principles. The residue is then cooled, and water is added to induce crystallization, after which the solid is filtered, washed with hot water to remove inorganic salts, and dried to yield the final product. Detailed standardized synthetic steps see the guide below.
- Charge catechol and 2,4-dinitrohalobenzene (molar ratio 1.0: 2.0-2.2) with a base like potassium carbonate into a reactor containing a mixed solvent system.
- Heat the mixture to reflux (80°C-200°C) for 3 to 18 hours to facilitate water separation and drive the reaction to completion.
- Concentrate the reaction solution, cool, add water to precipitate the solid product, then filter, wash, and dry to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers profound strategic advantages that extend far beyond simple chemical yield improvements. The ability to recycle the organic solvent system repeatedly without significant loss of efficiency translates directly into substantial operational expenditure reductions, as solvent purchase and disposal costs are among the largest variable costs in fine chemical manufacturing. Furthermore, the elimination of corrosive reagents and high-pressure requirements means that the process can be executed in standard glass-lined or stainless steel reactors, significantly lowering the barrier to entry for contract manufacturing organizations and reducing the capital intensity of new production lines. The simplicity of the workup procedure, which relies on straightforward filtration and washing rather than complex chromatographic separations, ensures faster batch turnover times and higher throughput capacity. This operational agility allows suppliers to respond more rapidly to fluctuating market demands, thereby enhancing supply chain reliability and reducing the risk of stockouts for critical downstream customers in the electronics and aerospace industries.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of inexpensive, commodity-grade raw materials such as catechol and simple inorganic bases like potassium carbonate, which are readily available in the global market. By avoiding the need for exotic catalysts or specialized reagents, the direct material costs are kept to a minimum, allowing for competitive pricing structures even in volatile market conditions. The high atom economy of the reaction, coupled with the efficient recovery of solvents, ensures that waste generation is minimized, which in turn lowers the costs associated with environmental compliance and waste treatment facilities. Additionally, the high purity of the crude product reduces the need for extensive purification steps, saving both energy and labor costs while maximizing the overall yield of saleable material per batch.
- Enhanced Supply Chain Reliability: From a logistics perspective, the robustness of this synthetic route mitigates many of the risks associated with the supply of complex intermediates. Since the reaction tolerates a variety of halogenated starting materials (fluoro, chloro, bromo, or iodo derivatives), manufacturers have the flexibility to source the most cost-effective or readily available electrophile at any given time without compromising the final product quality. This raw material flexibility acts as a buffer against supply disruptions, ensuring continuous production even if one specific precursor faces temporary shortages. Moreover, the stability of the intermediates and the final product allows for safer storage and transportation, reducing the regulatory burden and insurance costs associated with handling hazardous chemicals. This reliability makes the supplier a more attractive partner for long-term contracts, fostering stronger relationships with key accounts in the high-tech sector.
- Scalability and Environmental Compliance: The design of this process is inherently scalable, having been validated from laboratory bench scales up to industrial pilot runs without encountering the typical heat transfer or mixing issues that plague batch reactions. The absence of exothermic runaways and the operation at atmospheric pressure simplify the engineering controls required for scale-up, making it easier to transition from kilogram to tonne-scale production. Environmentally, the process aligns with modern sustainability goals by minimizing the use of toxic solvents and generating minimal aqueous waste, primarily consisting of benign inorganic salts. This "green" profile is increasingly important for multinational corporations that are under pressure to reduce their carbon footprint and adhere to strict environmental, social, and governance (ESG) criteria. Consequently, adopting this technology not only improves the bottom line but also enhances the corporate reputation of the manufacturer as a responsible and forward-thinking entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 1,2-bis(2,4-dinitrophenoxy)benzene, based on the specific embodiments and data provided in the patent literature. Understanding these nuances is essential for R&D teams evaluating the feasibility of integrating this intermediate into their own polymer synthesis workflows. The answers provided reflect the optimal conditions and expected outcomes when strictly adhering to the disclosed protocols, ensuring that stakeholders have a realistic expectation of performance metrics.
Q: What is the primary advantage of the solvent system described in CN101234988B?
A: The patent utilizes a mixed solvent system comprising a water-insoluble organic solvent and a polar aprotic solvent. This combination facilitates azeotropic water removal during reflux, driving the equilibrium forward while allowing for easy solvent recovery and recycling, significantly reducing waste.
Q: Why is this intermediate critical for the electronics industry?
A: 1,2-Bis(2,4-dinitrophenoxy)benzene serves as a key precursor for synthesizing highly branched aromatic polyimides. These polymers exhibit exceptional thermal stability and mechanical strength, making them essential for advanced LCD displays and aerospace applications.
Q: Does this process require high-pressure equipment?
A: No, the reaction proceeds under atmospheric pressure at temperatures between 80°C and 200°C. This eliminates the need for expensive high-pressure reactors, lowering capital expenditure and simplifying safety protocols for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2-Bis(2,4-Dinitrophenoxy)benzene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced polyimide materials hinges on the consistent supply of high-quality precursors like 1,2-bis(2,4-dinitrophenoxy)benzene. Our technical team has extensively analyzed the pathway described in CN101234988B and possesses the expertise to optimize this route for maximum efficiency and purity. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a steady stream of material regardless of their volume requirements. Our state-of-the-art rigorous QC labs enforce stringent purity specifications, guaranteeing that every batch meets the exacting standards required for electronic grade applications. By leveraging our deep understanding of nucleophilic aromatic substitution and solvent engineering, we can deliver a product that enables your downstream polymerization processes to achieve superior mechanical and thermal properties.
We invite you to collaborate with us to unlock the full potential of this technology for your specific application needs. Our team is prepared to provide a Customized Cost-Saving Analysis that demonstrates how switching to our optimized supply chain can reduce your overall manufacturing expenses. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project timeline. Whether you are developing next-generation LCD materials or high-temperature aerospace composites, NINGBO INNO PHARMCHEM is committed to being your strategic partner in innovation, delivering both chemical excellence and commercial value.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
