Advanced Synthesis of 4,4'-Bis(2,6-dinitro-4-trifluoromethylphenoxy)diphenylsulfone for High-Performance Polyimides
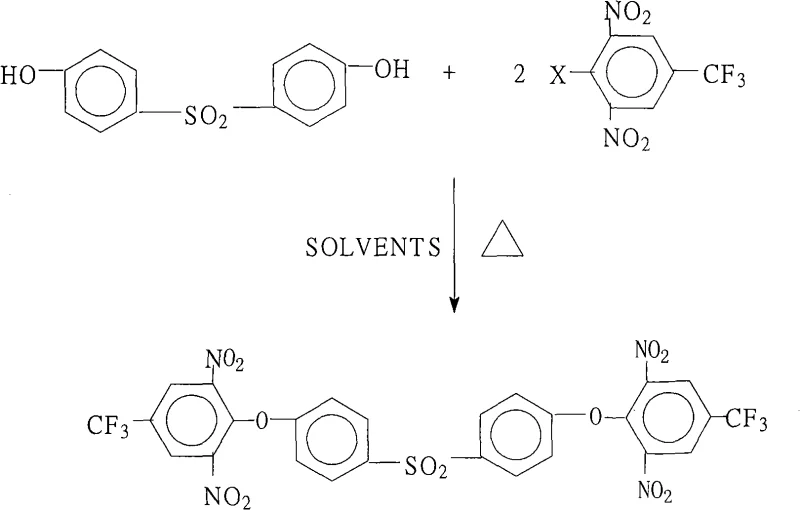
The landscape of advanced electronic materials is constantly evolving, driven by the demand for polymers that can withstand extreme thermal and chemical environments. A pivotal development in this sector is documented in patent CN101265219A, which outlines a robust preparation method for 4,4'-bis(2,6-dinitro-4-trifluoromethylphenoxy)diphenylsulfone. This specific aromatic compound acts as a foundational building block for generating highly branched aromatic polyimides, materials renowned for their exceptional mechanical strength and thermal stability in applications ranging from liquid crystal displays (LCDs) to aerospace components. The patent introduces a streamlined synthetic route that addresses historical challenges in purity and yield, offering a significant technological leap for manufacturers seeking reliable electronic material intermediate suppliers. By leveraging a novel solvent system and precise stoichiometric control, this method ensures the production of high-purity polyimide monomers that meet the rigorous specifications required for next-generation optoelectronic devices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations described in the provided intellectual property, the synthesis of complex sulfone-based intermediates often suffered from inconsistent reaction kinetics and difficult purification protocols. Traditional nucleophilic aromatic substitution reactions involving activated halobenzenes and phenols frequently struggled with incomplete conversion due to the reversible nature of the reaction in certain solvent environments. Furthermore, conventional processes often relied on single-phase solvent systems that made the removal of water, a byproduct of the salt formation, inefficient, thereby limiting the overall yield. The lack of standardized protocols for handling the specific 2,6-dinitro-4-trifluoromethyl halobenzene derivatives meant that manufacturers faced variability in impurity profiles, which is detrimental when producing materials for sensitive electronic applications. These inefficiencies translated into higher operational costs and longer lead times for high-purity electronic material intermediates, creating a bottleneck for downstream polymerization processes.
The Novel Approach
The methodology disclosed in the patent represents a paradigm shift by introducing a biphasic solvent strategy combined with azeotropic water removal. By employing a mixture of water-insoluble organic solvents, such as toluene or benzene, alongside highly polar aprotic solvents like N,N-dimethylformamide (DMF) or N-methyl-2-pyrrolidone (NMP), the reaction environment is optimized for both solubility and equilibrium displacement. This dual-solvent system facilitates the continuous removal of water generated during the formation of the phenoxide salt, effectively driving the reaction to completion. The result is a process that consistently achieves yields exceeding 95% with purity levels reaching 99.6%, significantly outperforming older techniques. This approach not only simplifies the operational workflow but also enhances the economic viability of cost reduction in display material manufacturing by minimizing raw material waste and maximizing throughput.
Mechanistic Insights into Nucleophilic Aromatic Substitution
The core chemical transformation in this synthesis is a classic nucleophilic aromatic substitution (SnAr), facilitated by the strong electron-withdrawing effects of the nitro groups on the halobenzene ring. In this mechanism, the 4,4'-dihydroxydiphenyl sulfone is first deprotonated by a salt-forming agent, such as potassium carbonate or potassium hydroxide, to generate a highly reactive phenoxide anion. This nucleophile then attacks the electron-deficient aromatic ring of the 2,6-dinitro-4-trifluoromethyl halobenzene at the position ortho to the nitro groups, where the electron density is lowest. The presence of two nitro groups significantly activates the ring towards nucleophilic attack, lowering the activation energy required for the substitution of the halogen atom. The trifluoromethyl group further enhances this reactivity through its inductive effect, ensuring that the reaction proceeds efficiently even under moderate thermal conditions. Understanding this electronic interplay is crucial for R&D teams aiming to optimize reaction parameters for commercial scale-up of complex polymer additives.
Impurity control in this reaction is largely dictated by the stoichiometry and the efficiency of the base used. The patent specifies a molar ratio of 1.0:2.0 to 2.2 between the diphenol and the halobenzene, ensuring a slight excess of the electrophile to drive the double substitution to completion and prevent the formation of mono-substituted byproducts. Additionally, the choice of base and the temperature range of 80°C to 200°C are critical; too low a temperature results in sluggish kinetics, while excessive heat could promote side reactions or decomposition of the sensitive nitro groups. The workup procedure, involving concentration and precipitation with water, leverages the differential solubility of the product versus unreacted starting materials and inorganic salts. This physical separation step is vital for achieving the reported purity of >99.5%, ensuring that the final crystalline product is free from metal ions and organic impurities that could compromise the color or electrical properties of the final polyimide film.
How to Synthesize 4,4'-Bis(2,6-dinitro-4-trifluoromethylphenoxy)diphenylsulfone Efficiently
Executing this synthesis requires careful attention to the solvent composition and the water removal process to replicate the high yields reported in the patent literature. The process begins with the precise weighing of 4,4'-dihydroxydiphenyl sulfone and the selected halobenzene derivative, followed by their suspension in the optimized biphasic solvent mixture. The reaction vessel must be equipped with a Dean-Stark trap or similar apparatus to facilitate the continuous azeotropic removal of water, which is the key driver for high conversion rates. Maintaining the reflux temperature within the specified 80°C to 200°C window is essential to balance reaction rate with product stability. For a comprehensive understanding of the exact operational parameters, including specific solvent volumes and agitation speeds, the detailed standardized synthesis steps are provided in the guide below.
- Mix 4,4'-dihydroxydiphenyl sulfone and 2,6-dinitro-4-trifluoromethyl halobenzene in a molar ratio of 1.0: 2.0-2.2 with a salt-forming agent.
- Heat the mixture in a biphasic solvent system (water-insoluble + polar aprotic) to reflux at 80°C-200°C for 6-18 hours with water separation.
- Concentrate the reaction, cool, add water to precipitate the solid, then filter, wash, and dry to obtain crystals with >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this patented synthesis route offers substantial strategic benefits, primarily centered around cost efficiency and supply chain resilience. The process eliminates the need for exotic or prohibitively expensive catalysts, relying instead on commodity chemicals like carbonates and common organic solvents that are readily available in the global market. This reliance on accessible raw materials drastically reduces the risk of supply disruptions and stabilizes pricing structures, which is a critical factor for long-term contract negotiations. Furthermore, the ability to recover and recycle the organic solvents multiple times without significant loss of performance contributes to a leaner manufacturing model. This solvent recovery capability not only lowers the direct material costs but also aligns with modern sustainability goals by reducing the volume of hazardous waste requiring disposal, thereby minimizing environmental compliance costs.
- Cost Reduction in Manufacturing: The economic advantage of this process is derived from its high atom economy and the elimination of complex purification steps such as column chromatography. By achieving high conversion rates through simple reflux and water separation, the need for extensive downstream processing is removed, leading to significant operational expenditure savings. The use of atmospheric pressure conditions means that specialized high-pressure reactors are not required, allowing for production in standard glass-lined or stainless steel vessels, which lowers capital investment barriers. Additionally, the high purity of the crude product reduces the load on recrystallization steps, saving energy and time. These factors combine to create a highly cost-competitive manufacturing profile that allows for aggressive pricing strategies in the marketplace.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch quality, which is paramount for maintaining trust with downstream polymer manufacturers. Since the process tolerates a range of halobenzene substrates (fluoro, chloro, bromo, iodo), supply chain managers have the flexibility to switch raw material sources based on availability and price fluctuations without altering the core process. This adaptability mitigates the risk of production stoppages due to raw material shortages. Moreover, the simplicity of the workup procedure—precipitation with water and filtration—allows for rapid turnover of batches, effectively reducing lead times for high-purity electronic material intermediates and enabling a more responsive just-in-time delivery model.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial tonnage is straightforward due to the absence of exothermic runaways or hazardous gas evolution. The reaction is thermally manageable and does not generate corrosive byproducts that would degrade equipment, extending the lifespan of production assets. From an environmental standpoint, the closed-loop solvent recovery system significantly minimizes volatile organic compound (VOC) emissions. The aqueous waste stream is primarily composed of inorganic salts, which are easier to treat than complex organic effluents. This environmental friendliness simplifies the permitting process for new production lines and ensures compliance with increasingly stringent global environmental regulations, securing the long-term viability of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized sulfone derivative. These answers are derived directly from the experimental data and claims within the patent documentation to ensure accuracy and reliability for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this intermediate into your specific polymer formulation pipeline.
Q: What is the primary application of 4,4'-bis(2,6-dinitro-4-trifluoromethylphenoxy)diphenylsulfone?
A: This compound serves as a critical intermediate for synthesizing highly branched aromatic polyimides, which are essential for high-temperature resistant materials in LCD displays and aerospace applications.
Q: How does the patented process improve yield compared to traditional methods?
A: The process utilizes a specific biphasic solvent system and water-separation reflux technique, driving the equilibrium forward and achieving yields up to 95.2% with purity exceeding 99.5%.
Q: Is the synthesis scalable for industrial production?
A: Yes, the method operates under atmospheric pressure without corrosive byproducts, uses recoverable solvents, and requires standard equipment, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Bis(2,6-dinitro-4-trifluoromethylphenoxy)diphenylsulfone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance intermediates play in the advancement of electronic materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. Our expertise in handling complex aromatic substitutions allows us to optimize the process described in CN101265219A for maximum yield and minimal environmental impact, providing our clients with a secure and high-quality supply source.
We invite you to collaborate with us to optimize your supply chain for polyimide monomers. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. By partnering with us, you gain access to specific COA data and route feasibility assessments that can help you streamline your production planning. Contact us today to discuss how we can support your project with reliable delivery and technical excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
