Advanced Synthesis of 4,4'-Bis(2,6-Dinitro-4-Trifluoromethylphenoxy)Diphenyl Ether for High-Performance Polyimides
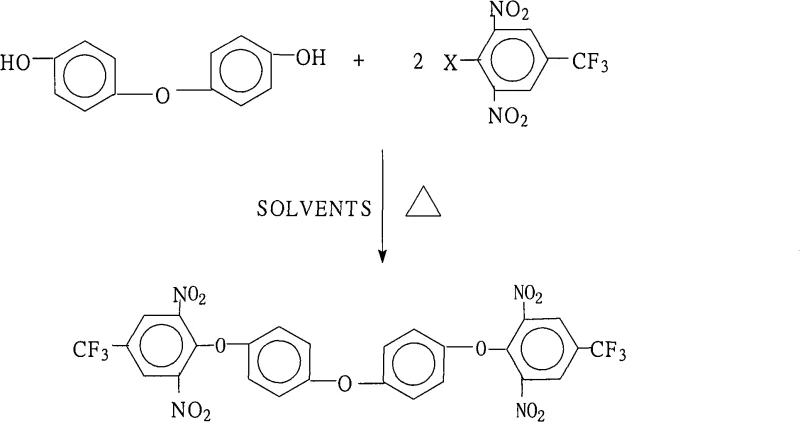
The landscape of advanced electronic materials is constantly evolving, driven by the demand for polymers with superior thermal stability and mechanical strength. A pivotal development in this sector is documented in patent CN101289397A, which discloses a novel preparation method for 4,4'-bis(2,6-dinitro-4-trifluoromethylphenoxy)diphenyl ether. This specific aromatic ether serves as a foundational building block for generating highly branched aromatic polyimides, materials that are indispensable in the fabrication of liquid crystal displays (LCDs) and aerospace components. Unlike traditional synthetic routes that often struggle with low conversion rates or difficult purification, this patented methodology introduces a refined solvent engineering approach that maximizes efficiency. By leveraging a specific molar ratio of reactants and a unique dual-solvent system, the process achieves exceptional purity levels exceeding 99.5%, addressing the rigorous quality standards required by R&D directors in the optoelectronics industry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations detailed in CN101289397A, the synthesis of complex fluorinated aromatic ethers often faced significant hurdles regarding reaction equilibrium and byproduct management. Conventional nucleophilic aromatic substitution reactions typically rely on single-phase solvent systems which can trap water generated during the reaction, thereby reversing the equilibrium and limiting overall yield. Furthermore, many existing protocols require harsh conditions or expensive phase-transfer catalysts that complicate downstream processing and increase the impurity profile. For procurement managers, these inefficiencies translate into higher raw material consumption and increased waste disposal costs. The lack of a standardized, high-yield industrial process for this specific bis-ether intermediate has historically created supply bottlenecks, forcing manufacturers to rely on small-batch laboratory syntheses that are neither cost-effective nor scalable for mass production of high-performance polyimides.
The Novel Approach
The patented method overcomes these historical limitations through a sophisticated manipulation of reaction thermodynamics using a mixed solvent system. By combining a water-insoluble organic solvent, such as toluene or chlorobenzene, with a strong polar aprotic solvent like N-methyl-2-pyrrolidone (NMP) or DMF, the process creates an environment conducive to continuous water removal via azeotropic distillation. This strategic setup ensures that the condensation reaction between 4,4'-dihydroxydiphenyl ether and the halobenzene derivative proceeds to near-completion without the need for excessive reagent loading. The result is a streamlined operation that operates under atmospheric pressure, eliminating the need for costly high-pressure reactors. For supply chain heads, this translates to a robust manufacturing protocol that utilizes readily available commodity chemicals while minimizing the complexity of the reaction setup, thereby enhancing overall process reliability and reducing the risk of production delays.
Mechanistic Insights into Nucleophilic Aromatic Substitution
The core chemical transformation driving this synthesis is a classic nucleophilic aromatic substitution (SnAr), facilitated by the powerful electron-withdrawing effects of the substituents on the benzene ring. In the reactant 2,6-dinitro-4-trifluoromethyl halobenzene, the presence of two nitro groups ortho to the leaving halogen atom, combined with the trifluoromethyl group at the para position, significantly activates the ring towards nucleophilic attack. This electronic activation lowers the energy barrier for the formation of the Meisenheimer complex, allowing the phenoxide anion derived from 4,4'-dihydroxydiphenyl ether to displace the halogen atom efficiently. Understanding this mechanism is crucial for R&D teams aiming to optimize reaction parameters, as the choice of the leaving group (fluorine, chlorine, bromine, or iodine) can subtly influence reaction kinetics, although the patent demonstrates high yields across various halogenated precursors.
Beyond the electronic activation, the control of impurities is meticulously managed through the reaction conditions and workup procedure. The use of inorganic bases such as potassium carbonate or sodium hydroxide ensures the quantitative generation of the phenoxide nucleophile while minimizing side reactions like hydrolysis of the nitro groups. The subsequent workup, involving concentration and precipitation with water, exploits the differential solubility of the product versus unreacted starting materials and inorganic salts. This physical separation step is vital for achieving the reported purity of >99.5%, as it effectively removes residual halobenzene and base salts without requiring energy-intensive chromatographic purification. For quality assurance professionals, this inherent purity built into the process design reduces the analytical burden and ensures consistent batch-to-batch reproducibility essential for electronic grade materials.
How to Synthesize 4,4'-Bis(2,6-Dinitro-4-Trifluoromethylphenoxy)Diphenyl Ether Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from laboratory discovery to pilot-scale production. The process begins with the precise charging of reactants into a reactor equipped with a water separator, followed by a controlled heating ramp to maintain reflux conditions. The critical success factor lies in the maintenance of the water separation phase for a duration of 6 to 18 hours, ensuring complete conversion. Detailed standardized operating procedures regarding temperature gradients, stirring speeds, and addition rates are essential to replicate the high yields observed in the patent examples. For technical teams looking to implement this route, adhering to the specified molar ratios and solvent volumes is paramount to avoiding viscosity issues and ensuring efficient heat transfer throughout the reaction mass.
- Charge reactor with 4,4'-dihydroxydiphenyl ether, 2,6-dinitro-4-trifluoromethyl halobenzene, base, and mixed solvent system.
- Heat to reflux (80°C-180°C) with continuous water separation for 6 to 18 hours to drive equilibrium.
- Concentrate reaction mixture, cool, precipitate product with water, filter, wash, and dry to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers substantial strategic benefits for organizations focused on cost optimization and supply security. The process is designed with industrial scalability in mind, utilizing equipment and conditions that are standard in fine chemical manufacturing facilities. By avoiding exotic catalysts or extreme pressure requirements, the capital expenditure (CAPEX) needed to establish production lines is significantly reduced. Furthermore, the ability to recover and recycle the organic solvents used in the reaction mixture directly impacts the operational expenditure (OPEX), lowering the per-kilogram cost of the final intermediate. This economic efficiency makes the material more accessible for downstream polymer manufacturers who are constantly seeking to reduce the cost basis of high-performance electronic chemicals without compromising on material properties.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the implementation of a solvent recovery loop drastically simplify the cost structure of the manufacturing process. Since the reaction proceeds efficiently under atmospheric pressure, there is no need for specialized high-pressure autoclaves, which reduces both equipment investment and maintenance costs. Additionally, the high conversion rate minimizes the loss of valuable fluorinated starting materials, ensuring that raw material utilization is maximized. These factors collectively contribute to a leaner production model that can withstand market fluctuations in raw material pricing, offering a stable cost basis for long-term supply contracts.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as carbonates, toluene, and DMF ensures that the supply chain is not vulnerable to the bottlenecks often associated with specialty reagents. The robustness of the reaction conditions allows for flexible scheduling and rapid scale-up in response to market demand surges. Because the workup involves simple filtration and washing steps rather than complex distillations or extractions, the cycle time for each batch is optimized, leading to improved throughput. This operational agility enables suppliers to maintain consistent inventory levels and meet tight delivery windows, which is critical for just-in-time manufacturing environments in the electronics sector.
- Scalability and Environmental Compliance: The process generates minimal waste due to the high atom economy of the substitution reaction and the recyclability of the solvent system. The absence of corrosive byproducts simplifies waste treatment protocols and reduces the environmental footprint of the manufacturing site. This alignment with green chemistry principles not only ensures compliance with increasingly stringent environmental regulations but also enhances the corporate sustainability profile of the manufacturer. For global supply chains, this environmental compatibility facilitates smoother regulatory approvals and logistics, ensuring uninterrupted flow of materials across international borders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized intermediate. The answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this material into their specific product formulations or supply networks. Clear communication regarding process capabilities and material specifications is essential for fostering trust between suppliers and technical buyers in the high-stakes electronic materials market.
Q: What is the primary application of 4,4'-bis(2,6-dinitro-4-trifluoromethylphenoxy)diphenyl ether?
A: This compound serves as a critical precursor for synthesizing highly branched aromatic polyimides, which are essential for high-temperature resistant films in LCD displays and aerospace applications.
Q: How does the dual-solvent system improve the reaction yield?
A: The combination of a water-insoluble solvent and a polar aprotic solvent facilitates azeotropic water removal during reflux, shifting the equilibrium towards product formation and preventing hydrolysis.
Q: Is this synthesis scalable for industrial production?
A: Yes, the process operates at atmospheric pressure with simple workup steps like filtration and washing, making it highly suitable for large-scale commercial manufacturing without specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Bis(2,6-Dinitro-4-Trifluoromethylphenoxy)Diphenyl Ether Supplier
As the demand for high-performance polyimides continues to grow in the semiconductor and display industries, securing a reliable source of key intermediates is more critical than ever. NINGBO INNO PHARMCHEM stands ready to support your development goals with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is well-versed in the intricacies of nucleophilic aromatic substitutions and solvent engineering, ensuring that we can deliver this complex ether with stringent purity specifications tailored to your application needs. With our rigorous QC labs and state-of-the-art manufacturing facilities, we guarantee batch-to-batch consistency that meets the exacting standards of the global electronics supply chain.
We invite you to collaborate with us to optimize your material sourcing strategy and achieve significant efficiencies in your production processes. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that evaluates the potential economic benefits of switching to our optimized synthesis route. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that will demonstrate how our capabilities align with your project timelines and quality requirements. Let us help you accelerate your path to market with a supply partner dedicated to excellence and innovation.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
