Industrial Scale Synthesis of High-Purity Polyimide Intermediates for Advanced Electronics
The rapid advancement of the optoelectronics and aerospace sectors has created an insatiable demand for high-performance polymers capable of withstanding extreme thermal and mechanical stress. At the forefront of this material revolution is the development of highly branched aromatic polyimides, which offer superior insulation, chemical resistance, and film-forming properties compared to their linear counterparts. Central to the production of these advanced materials is the availability of specialized monomers, specifically 2,6-bis(2,6-dinitro-4-trifluoromethylphenoxy)toluene, a complex fluorinated intermediate that introduces critical structural rigidity and thermal stability. Patent CN101284784A discloses a groundbreaking preparation method for this key intermediate, addressing a significant gap in the prior art where no public patents or literature had previously reported a viable industrial synthesis. This technical insight report analyzes the proprietary chemistry behind this innovation, highlighting its potential to redefine the supply chain for reliable electronic chemical suppliers seeking to optimize cost structures and ensure material consistency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the disclosure of patent CN101284784A, the synthesis of sterically hindered diaryl ethers containing multiple electron-withdrawing groups was fraught with significant technical challenges that hindered commercial scalability. Traditional nucleophilic aromatic substitution reactions often suffered from incomplete conversion due to the deactivation of the nucleophile or the formation of intractable tars when harsh conditions were employed to overcome steric barriers. Furthermore, existing methods for similar fluorinated intermediates frequently relied on expensive phase transfer catalysts or anhydrous conditions that were difficult to maintain on a large scale, leading to batch-to-batch variability. The lack of a standardized protocol meant that procurement managers faced unpredictable lead times and inflated costs associated with low-yielding pilot runs. Additionally, the disposal of heavy metal catalysts or corrosive reagents commonly used in older etherification protocols posed severe environmental compliance risks, complicating the waste management logistics for supply chain heads. Without a robust, literature-backed method, R&D teams were forced to dedicate excessive resources to process development rather than application testing, delaying time-to-market for new polyimide formulations.
The Novel Approach
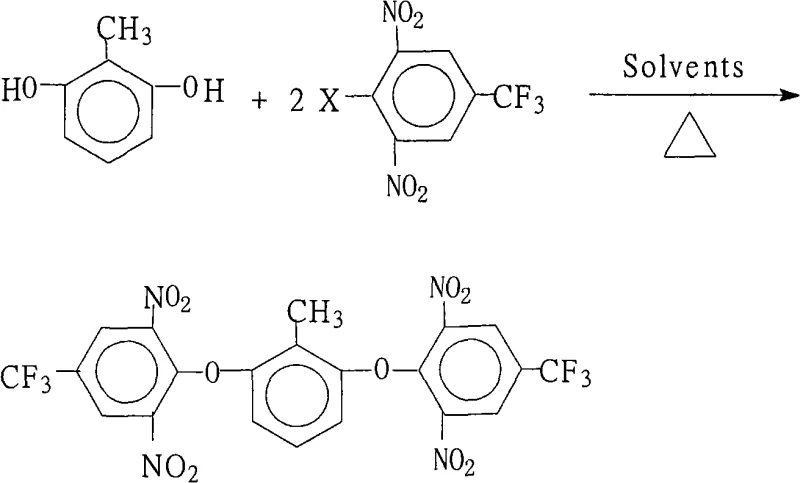
The methodology outlined in the patent represents a paradigm shift by utilizing a straightforward nucleophilic substitution strategy that leverages the inherent reactivity of the poly-nitro and trifluoromethyl substituted benzene ring. By employing a specific molar ratio of 2,6-dihydroxytoluene to the halobenzene derivative (1.0:2.0 to 2.2), the process ensures complete consumption of the dihydroxy precursor while minimizing the formation of mono-substituted byproducts. The innovation lies in the sophisticated solvent system, which combines a water-insoluble organic solvent like benzene or toluene with a strong polar aprotic solvent such as N,N-dimethylformamide (DMF) or N-methyl-2-pyrrolidone (NMP). This dual-solvent approach facilitates the formation of the phenoxide salt in situ while allowing for the continuous removal of water via azeotropic distillation during the reflux period of 6 to 18 hours. Operating at atmospheric pressure between 80°C and 150°C eliminates the need for expensive high-pressure reactors, drastically simplifying the equipment requirements for commercial scale-up of complex fluorinated intermediates. The result is a streamlined process that delivers high-purity crystals with yields consistently exceeding 80%, providing a reliable foundation for industrial manufacturing.
Mechanistic Insights into Nucleophilic Aromatic Substitution (SNAr)
The core chemical transformation driving this synthesis is a classic Nucleophilic Aromatic Substitution (SNAr), but it is finely tuned through electronic activation and steric management to achieve exceptional efficiency. The presence of two strongly electron-withdrawing nitro groups at the ortho positions and a trifluoromethyl group at the para position of the halobenzene ring creates a highly electron-deficient aromatic system. This electronic configuration significantly lowers the energy of the Lowest Unoccupied Molecular Orbital (LUMO), making the carbon atom bearing the halogen leaving group extremely susceptible to nucleophilic attack by the phenoxide anion generated from 2,6-dihydroxytoluene. The reaction proceeds through a Meisenheimer complex intermediate, where the negative charge is delocalized onto the nitro oxygen atoms, stabilizing the transition state and lowering the activation energy barrier. The choice of base, ranging from potassium carbonate to potassium hydroxide, is critical for generating the reactive phenoxide species without causing hydrolysis of the sensitive nitro or trifluoromethyl functionalities. Furthermore, the use of a mixed solvent system enhances the solubility of both the ionic salt intermediate and the organic reactants, ensuring a homogeneous reaction environment that promotes kinetic control over thermodynamic side reactions.
Impurity control is meticulously managed through stoichiometric precision and thermal regulation, which are vital for producing high-purity OLED material or polyimide precursors. By maintaining a slight excess of the halobenzene reactant (up to 2.2 equivalents), the process drives the equilibrium towards the desired bis-ether product, effectively suppressing the accumulation of the mono-ether intermediate which could act as a chain terminator in subsequent polymerization steps. The reflux temperature range of 80°C to 150°C is optimized to provide sufficient thermal energy for the substitution to occur at the sterically crowded 2,6-positions without inducing decomposition of the thermally sensitive nitro groups. Post-reaction processing involves concentrating the solution and adding water, which exploits the differential solubility of the product versus inorganic salts and unreacted starting materials to induce crystallization. Washing the crude solid with hot water further removes residual inorganic bases and polar impurities, resulting in a final product with purity specifications often reaching 99.8%, thereby reducing lead time for high-purity polyimide precursors by eliminating the need for complex chromatographic purification.
How to Synthesize 2,6-Bis(2,6-Dinitro-4-Trifluoromethylphenoxy)Toluene Efficiently
The synthesis protocol described in the patent offers a reproducible pathway for manufacturing this critical intermediate, balancing reaction kinetics with operational simplicity to ensure consistent quality. The process begins with the precise charging of reactants into a standard reactor equipped with a reflux condenser and a water separator, followed by the addition of the base and the tailored solvent mixture. Heating is applied gradually to reach the reflux temperature, where the system is maintained for a duration determined by the specific halogen substituent, typically ranging from 6 to 18 hours to ensure full conversion.
- Charge 2,6-dihydroxytoluene and 2,6-dinitro-4-trifluoromethyl halobenzene (molar ratio 1.0: 2.0-2.2) with a base like potassium carbonate into a reactor containing a mixed solvent system.
- Heat the mixture to reflux (80°C to 150°C) for 6 to 18 hours to facilitate water separation and drive the nucleophilic aromatic substitution to completion.
- Concentrate the reaction liquid, cool the system, add water to precipitate the solid product, then filter, wash with hot water, and dry to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers transformative benefits that extend far beyond simple chemical yield, impacting the total cost of ownership and operational resilience. The elimination of exotic catalysts and the reliance on commodity chemicals like carbonates and common organic solvents creates a robust supply chain that is less vulnerable to geopolitical disruptions or raw material shortages. The ability to recover and recycle the organic solvent system repeatedly not only aligns with stringent environmental regulations but also generates substantial cost savings by reducing the volume of fresh solvent required for each production batch. Moreover, the operation at atmospheric pressure significantly lowers the capital expenditure required for plant infrastructure, as it negates the need for specialized high-pressure vessels and the associated safety systems, making it an attractive option for cost reduction in electronic chemical manufacturing. The high purity of the final product minimizes downstream processing costs for polymer manufacturers, who can rely on consistent feedstock quality to maintain their own production schedules without unexpected downtime.
- Cost Reduction in Manufacturing: The process achieves significant economic efficiency by utilizing inexpensive, widely available inorganic bases such as potassium carbonate or sodium hydroxide instead of costly organometallic catalysts. The solvent system is designed for easy recovery; after the reaction, the solvent can be distilled and reused multiple times without significant loss of performance, which drastically cuts down on raw material expenses. Furthermore, the high yield (up to 98.0% in optimized examples) means that less raw material is wasted per kilogram of finished product, directly improving the gross margin for the manufacturer. The absence of corrosive byproducts also reduces maintenance costs for reactor vessels and piping, extending the lifespan of the production equipment.
- Enhanced Supply Chain Reliability: By relying on a synthetic route that uses stable, non-hazardous starting materials, the risk of supply chain interruptions due to regulatory bans on toxic reagents is minimized. The reaction conditions are mild enough to be performed in standard glass-lined or stainless steel reactors found in most fine chemical facilities, ensuring that production can be scaled up quickly across different geographic locations without technology transfer bottlenecks. The robustness of the crystallization step ensures that the final product is a stable solid that is easy to store and transport, reducing logistics complexities compared to liquid intermediates that may require temperature-controlled shipping. This stability allows for the maintenance of strategic inventory buffers, safeguarding against market volatility.
- Scalability and Environmental Compliance: The process is inherently green, generating minimal waste streams since the primary byproduct is inorganic salt and water, which are easily treated. The solvent recovery loop significantly reduces the emission of Volatile Organic Compounds (VOCs), helping manufacturers meet increasingly strict environmental standards without investing in expensive abatement technologies. The scalability is proven by the linear relationship between lab-scale and pilot-scale results, indicating that the heat and mass transfer characteristics of the reaction are well-suited for large batch reactors. This ease of scale-up allows companies to respond rapidly to surges in demand from the LCD or aerospace sectors, ensuring continuity of supply for critical downstream applications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized intermediate, derived directly from the patent specifications and industry best practices. Understanding these details is crucial for R&D directors evaluating the feasibility of integrating this monomer into new polymer formulations.
Q: What is the primary application of 2,6-bis(2,6-dinitro-4-trifluoromethylphenoxy)toluene?
A: This compound serves as a critical intermediate for synthesizing highly branched aromatic polyimide monomers. These polyimides are essential for high-performance applications in LCD displays, aerospace, and microelectronics due to their superior thermal stability and mechanical strength.
Q: How does this patented method improve upon traditional ether synthesis?
A: Unlike conventional methods that may struggle with steric hindrance or require hazardous conditions, this process operates at atmospheric pressure without corrosive byproducts. It utilizes a specific mixed solvent system that allows for easy solvent recovery and recycling, significantly reducing environmental waste and operational costs.
Q: What purity levels can be achieved with this synthesis route?
A: The patented procedure consistently yields products with purity exceeding 99.0%, with specific examples demonstrating purity up to 99.8%. This high level of purity is crucial for downstream polymerization processes where impurities can degrade the thermal and electrical properties of the final polyimide material.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,6-Bis(2,6-Dinitro-4-Trifluoromethylphenoxy)Toluene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced polyimides depends on the consistent availability of high-quality monomers like 2,6-bis(2,6-dinitro-4-trifluoromethylphenoxy)toluene. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of the global electronics supply chain. We operate stringent purity specifications and utilize rigorous QC labs to verify that every batch meets the exacting standards required for high-performance optical and electrical applications. Our commitment to process optimization allows us to deliver this complex intermediate with the reliability and purity necessary to support your most demanding R&D projects.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs through our Customized Cost-Saving Analysis. Our technical procurement team is ready to provide specific COA data and route feasibility assessments tailored to your unique volume requirements. By partnering with us, you gain access to a secure, scalable source of critical chemical building blocks that will empower your next generation of high-performance materials.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
