Scalable Synthesis of High-Purity 1,3,5-Tris(2,6-Dinitro-4-Trifluoromethylphenoxy)Benzene for Advanced Polyimide Applications
Introduction to Advanced Polyimide Precursor Technology
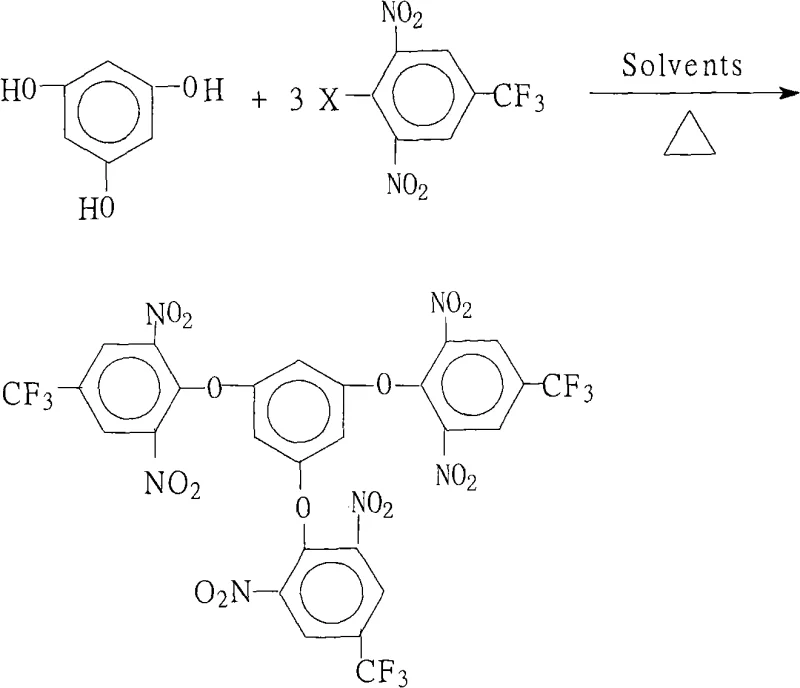
The rapid evolution of the electronic materials sector, particularly in the realm of liquid crystal displays (LCDs) and high-performance polymers, demands precursors that offer exceptional thermal stability and mechanical integrity. Patent CN101279918A introduces a groundbreaking preparation method for 1,3,5-tris(2,6-dinitro-4-trifluoromethylphenoxy)benzene, a critical intermediate for synthesizing highly branched aromatic polyimides. This specific molecular architecture is pivotal for creating next-generation functional materials that must withstand rigorous operating conditions in aerospace and microelectronics. The disclosed technology represents a significant leap forward, transitioning from theoretical possibility to a robust, industrially viable process that ensures high purity and yield. By leveraging a precise nucleophilic aromatic substitution strategy, this method overcomes historical synthetic bottlenecks, providing a reliable foundation for the mass production of advanced optical and electrical materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovation detailed in CN101279918A, the chemical industry faced a conspicuous void in the literature regarding the efficient synthesis of this specific trifluoromethyl-substitated ether. Conventional approaches to constructing highly nitrated, fluorinated ether linkages often suffered from severe limitations, including the requirement for harsh reaction conditions that compromised safety and equipment longevity. Traditional methods frequently struggled with incomplete substitution due to steric hindrance around the benzene ring, leading to complex mixtures of mono- and di-substituted impurities that were difficult to separate. Furthermore, many existing protocols relied on expensive or hazardous reagents that generated substantial toxic waste, rendering them economically unfeasible for large-scale manufacturing. The lack of a standardized, high-yielding pathway meant that supply chains for these specialized polyimide monomers were fragile, inconsistent, and incapable of meeting the growing demand from the display and semiconductor industries.
The Novel Approach
The novel approach presented in this patent fundamentally reshapes the synthetic landscape by introducing a streamlined, one-pot reaction system that maximizes efficiency while minimizing complexity. By utilizing a specific molar ratio of phloroglucinol to the halogenated nitro compound (1.0:3.0-3.2) in a dual-solvent system, the process ensures complete triple substitution with remarkable selectivity. The integration of a salt-forming agent, such as potassium carbonate, facilitates the generation of the reactive phenoxide nucleophile under mild conditions, avoiding the need for extreme temperatures or pressures. Crucially, the method incorporates an azeotropic water removal technique during reflux, which thermodynamically drives the equilibrium towards the desired product, effectively suppressing reverse reactions and side-product formation. This strategic design not only simplifies the operational workflow but also guarantees a product purity exceeding 99%, directly addressing the stringent quality requirements of high-end electronic applications.
Mechanistic Insights into Nucleophilic Aromatic Substitution
The core chemical transformation driving this synthesis is a classic yet meticulously optimized nucleophilic aromatic substitution (SnAr). In this mechanism, the electron-withdrawing nature of the two nitro groups (-NO2) and the trifluoromethyl group (-CF3) on the halobenzene ring significantly activates the carbon-halogen bond towards nucleophilic attack. When the base deprotonates the hydroxyl groups of the phloroglucinol, the resulting phenoxide anions become potent nucleophiles that attack the electron-deficient aromatic ring at the position ortho to the nitro groups. The presence of the strong electron-withdrawing trifluoromethyl group further stabilizes the Meisenheimer complex intermediate, lowering the activation energy required for the substitution. This electronic activation is critical, as it allows the reaction to proceed smoothly at moderate temperatures (80°C to 150°C) without requiring aggressive catalysts that could introduce metallic impurities detrimental to electronic performance.
Controlling the impurity profile in such a multi-step substitution is paramount for ensuring the final polyimide possesses uniform properties. The patent specifies a carefully balanced solvent system comprising a water-insoluble organic solvent (like toluene or benzene) and a strong polar aprotic solvent (such as DMF or NMP). This binary solvent mixture serves a dual purpose: it solubilizes both the organic reactants and the inorganic base salts, ensuring a homogeneous reaction phase, while simultaneously facilitating the removal of water generated during the salt formation. By continuously removing water via azeotropic distillation, the system prevents the hydrolysis of the sensitive halobenzene starting material and suppresses the formation of phenolic byproducts. This precise control over the reaction environment ensures that the final crystalline product is free from oligomeric impurities, thereby guaranteeing the high thermal and mechanical stability required for advanced polyimide films used in LCD substrates.
How to Synthesize 1,3,5-Tris(2,6-Dinitro-4-Trifluoromethylphenoxy)Benzene Efficiently
Implementing this synthesis route requires strict adherence to the stoichiometric ratios and thermal profiles outlined in the patent to achieve the reported yields of over 90%. The process begins with the careful charging of reactants into a reactor equipped with a water separator, followed by a controlled heating ramp to establish reflux conditions. Operators must monitor the water collection closely, as the cessation of water evolution indicates the completion of the reaction, typically occurring within a 6 to 18-hour window depending on the specific halogen substituent used. The subsequent workup involves concentrating the reaction mixture to recover valuable solvents for reuse, a step that is critical for both economic and environmental reasons, before precipitating the product with water. For a detailed, step-by-step breakdown of the standardized operating procedures and safety protocols, please refer to the technical guide below.
- Charge a reactor with phloroglucinol and 2,6-dinitro-4-trifluoromethyl halobenzene in a molar ratio of 1.0: 3.0-3.2, along with a carbonate or hydroxide base and a mixed solvent system of water-insoluble and polar aprotic solvents.
- Heat the reaction mixture to reflux between 80°C and 150°C for 6 to 18 hours, utilizing azeotropic distillation to continuously remove water and drive the equilibrium towards product formation.
- Concentrate the reaction solution to recover solvents for recycling, cool the residue, add water to precipitate the solid product, then filter, wash with hot water, and dry to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers transformative benefits that extend far beyond simple chemical synthesis. The process is engineered to drastically simplify the manufacturing workflow, eliminating the need for complex purification columns or exotic reagents that often bottleneck production schedules. By utilizing widely available commodity chemicals like phloroglucinol and common carbonates, the supply chain becomes inherently more resilient against raw material volatility. Furthermore, the ability to recover and recycle the organic solvent system multiple times without significant loss of efficiency translates directly into substantial operational expenditure (OPEX) reductions. This closed-loop solvent management not only lowers the cost per kilogram of the final product but also aligns perfectly with modern sustainability mandates, reducing the facility's overall environmental footprint and waste disposal costs.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by its reliance on inexpensive, commercially abundant starting materials and the elimination of costly transition metal catalysts. By avoiding the use of precious metals, manufacturers bypass the expensive and time-consuming heavy metal removal steps typically required in pharmaceutical and electronic grade synthesis. Additionally, the high atom economy of the reaction, combined with the efficient recovery of solvents like toluene and DMF, ensures that raw material utilization is maximized. This holistic approach to cost management results in a significantly lower cost of goods sold (COGS), providing a competitive pricing advantage in the global market for specialty electronic chemicals.
- Enhanced Supply Chain Reliability: Supply chain continuity is fortified by the robustness of the reaction conditions, which operate at atmospheric pressure and do not require specialized high-pressure reactors or cryogenic cooling systems. This accessibility means that production can be scaled across a wider range of manufacturing facilities without necessitating massive capital investment in new infrastructure. The simplicity of the workup procedure, involving basic filtration and drying, minimizes the risk of processing errors and batch failures, ensuring consistent on-time delivery to downstream polymer manufacturers. Consequently, partners can rely on a stable, predictable supply of high-purity intermediates, mitigating the risks associated with production delays or quality deviations.
- Scalability and Environmental Compliance: Scaling this synthesis from laboratory benchtop to multi-ton production is seamless due to the linear nature of the reaction kinetics and the absence of exothermic runaway risks. The process generates minimal hazardous waste, as the primary byproduct is inorganic salt and water, which are easily treated. The capability to recycle solvents repeatedly reduces the volume of volatile organic compounds (VOCs) released into the atmosphere, facilitating easier compliance with stringent environmental regulations. This eco-friendly profile not only future-proofs the manufacturing asset against tightening regulatory frameworks but also enhances the brand reputation of the supplier as a responsible partner in the green chemistry initiative.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 1,3,5-tris(2,6-dinitro-4-trifluoromethylphenoxy)benzene. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these nuances is essential for evaluating the feasibility of integrating this intermediate into your specific polymer formulation or supply chain strategy. We encourage stakeholders to review these insights to fully appreciate the operational efficiencies and quality assurances offered by this novel synthetic route.
Q: What are the primary advantages of this synthesis method compared to traditional approaches?
A: This method addresses the lack of existing industrial protocols by offering a simple, atmospheric pressure process that avoids corrosive reagents. It achieves exceptionally high yields (over 90%) and purity (over 99%) while enabling solvent recovery, significantly reducing environmental waste and operational costs.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is explicitly designed for industrialization. It utilizes readily available raw materials like phloroglucinol and common organic solvents, operates under standard pressure without specialized high-pressure equipment, and features a straightforward workup procedure involving filtration and drying, ensuring excellent scalability.
Q: How does this method handle environmental compliance and waste management?
A: The protocol emphasizes environmental friendliness by using a solvent system that allows for easy separation and repeated recycling. The reaction generates minimal three-waste (waste water, gas, and solids), and the absence of corrosive byproducts simplifies equipment maintenance and disposal compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3,5-Tris(2,6-Dinitro-4-Trifluoromethylphenoxy)Benzene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent to commercial reality requires a partner with deep technical expertise and proven scaling capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and purity specifications demonstrated in the lab are faithfully reproduced at an industrial level. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the most demanding electronic grade standards. Our commitment to quality assurance means that every kilogram of 1,3,5-tris(2,6-dinitro-4-trifluoromethylphenoxy)benzene we supply is backed by comprehensive data packages, giving your R&D and production teams the confidence to proceed with formulation and processing without delay.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your specific material requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic benefits tailored to your volume needs. We encourage you to reach out today to索取 specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing excellence can become a strategic advantage for your supply chain. Let us collaborate to accelerate the development of your next-generation polyimide materials with a reliable, high-quality source.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
