Scalable Synthesis of High-Purity 2,6-Dimethyl-1,4-Bis(2,4-Dinitrophenoxy)Benzene for Advanced Polyimides
The rapid advancement of the optoelectronics and aerospace sectors has created an insatiable demand for high-performance polymers capable of withstanding extreme thermal and mechanical stress. At the heart of this material revolution lies the development of specialized aromatic polyimides, which require precise and highly pure monomeric building blocks to achieve their superior properties. Patent CN101234983A introduces a groundbreaking preparation method for 2,6-dimethyl-1,4-bis(2,4-dinitrophenoxy)benzene, a pivotal intermediate in the synthesis of highly branched aromatic polyimides. This novel synthetic route addresses the critical industry need for scalable, environmentally friendly, and cost-effective production of complex ether-linked aromatic compounds. By leveraging a refined nucleophilic aromatic substitution strategy, this technology enables the manufacture of precursors that ultimately yield polyimide systems with enhanced temperature resistance and comprehensive mechanical performance, meeting the rigorous standards required for liquid crystal display (LCD) applications and advanced microelectronics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of multi-substituted aromatic ethers with high steric hindrance and specific substitution patterns has been plagued by significant technical challenges. Traditional etherification routes often suffer from incomplete conversion due to the deactivation of the aromatic ring or the formation of intractable by-products that are difficult to separate. Many conventional processes rely on harsh conditions, such as extremely high temperatures or the use of expensive phase-transfer catalysts that leave behind toxic metallic residues, complicating downstream purification. Furthermore, standard methods frequently struggle with the removal of reaction-generated water, which can hydrolyze sensitive intermediates or shift the equilibrium backward, resulting in suboptimal yields. The inability to efficiently recycle solvents in these older methodologies also leads to excessive waste generation and inflated operational expenditures, making them unsustainable for modern large-scale industrial production where environmental compliance and cost control are paramount.
The Novel Approach
The methodology disclosed in patent CN101234983A represents a paradigm shift in the synthesis of this specific dinitro-functionalized intermediate. By employing a dual-solvent system consisting of a water-insoluble organic solvent and a strongly polar aprotic solvent, the process achieves an optimal balance between reactant solubility and efficient water removal. The reaction utilizes a molar ratio of 1.0:2.0 to 2.2 between 2,6-dimethyl-hydroquinone and 2,4-dinitrohalobenzene, ensuring complete consumption of the valuable dihydroxy component. As illustrated in the reaction scheme below, the process operates under atmospheric pressure with heating reflux, utilizing a salt-forming agent to generate the reactive phenoxide species in situ. This approach not only simplifies the equipment requirements but also allows for the continuous separation of water, driving the equilibrium towards the product side and achieving exceptional yields exceeding 95% in optimized examples.
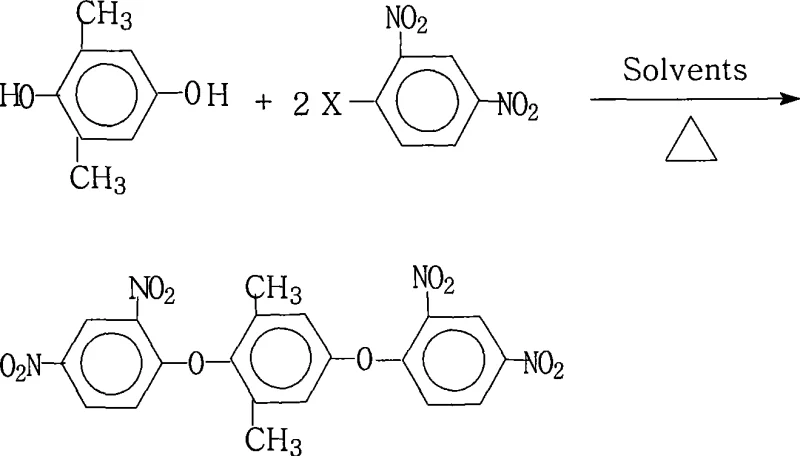
Mechanistic Insights into Nucleophilic Aromatic Substitution
The core chemical transformation driving this synthesis is a classic nucleophilic aromatic substitution (SnAr), facilitated by the powerful electron-withdrawing nature of the nitro groups positioned ortho and para to the leaving halogen atom. In this mechanism, the 2,6-dimethyl-hydroquinone is first deprotonated by the inorganic base, such as potassium carbonate or sodium hydroxide, to form a highly nucleophilic phenoxide anion. This anion then attacks the electron-deficient aromatic ring of the 2,4-dinitrohalobenzene at the carbon bearing the halogen substituent. The presence of two nitro groups significantly stabilizes the negatively charged Meisenheimer complex intermediate through resonance delocalization, thereby lowering the activation energy required for the substitution. The choice of halogen (fluorine, chlorine, bromine, or iodine) offers tunability; while fluorine is typically the best leaving group in SnAr reactions due to its high electronegativity enhancing the ring's electrophilicity, chlorine and bromine offer cost advantages and are effectively utilized in this protocol due to the optimized thermal conditions and solvent environment.
Impurity control is meticulously managed through the specific selection of the solvent system and the water-separation technique. The use of a polar aprotic co-solvent like N,N-dimethylformamide (DMF) or dimethyl sulfoxide (DMSO) ensures that the ionic phenoxide species remains dissolved and reactive, preventing localized high concentrations that could lead to polymerization or side reactions. Simultaneously, the water-insoluble solvent, such as toluene or xylene, forms an azeotrope with the water generated during the etherification. Continuous removal of this water via reflux prevents the hydrolysis of the 2,4-dinitrohalobenzene, a common side reaction that produces 2,4-dinitrophenol and reduces overall yield. This precise control over the reaction environment ensures that the final crystalline product exhibits a purity of greater than 99%, minimizing the need for extensive recrystallization and reducing the risk of color bodies or structural defects in the final polyimide material.
How to Synthesize 2,6-Dimethyl-1,4-Bis(2,4-Dinitrophenoxy)Benzene Efficiently
The operational procedure for this synthesis is designed for robustness and scalability, making it ideal for transfer from laboratory benchtop to kiloton-scale production reactors. The process begins with the precise charging of reactants into a standard stirred tank reactor equipped with a Dean-Stark trap or similar water-separation apparatus. The reaction mixture is heated to a temperature range of 80°C to 200°C, depending on the boiling point of the chosen solvent system, and maintained under reflux for a duration of 3 to 18 hours. This extended reaction time ensures complete conversion even when using less reactive halogenated substrates like chlorobenzene derivatives. Following the reaction completion, the workup involves concentrating the reaction liquor to recover the valuable organic solvents for reuse, followed by cooling and the addition of water to precipitate the solid product. The resulting yellowish-brown crystals are then filtered, washed with hot water to remove inorganic salts, and dried to yield the high-purity intermediate ready for subsequent reduction to the corresponding amine.
- Charge reactor with 2,6-dimethyl-hydroquinone and 2,4-dinitrohalobenzene (molar ratio 1: 2.0-2.2) in a mixed solvent system.
- Add salt-forming agent (e.g., potassium carbonate) and heat to reflux (80-200°C) for 3-18 hours with continuous water separation.
- Concentrate reaction mixture, cool, precipitate with water, filter, wash, and dry to obtain yellowish-brown crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers substantial strategic advantages beyond mere technical feasibility. The process is engineered to minimize operational complexity and maximize resource efficiency, directly translating to a more resilient and cost-effective supply chain for critical electronic chemical intermediates. By eliminating the need for exotic catalysts or high-pressure equipment, the capital expenditure (CapEx) required for setting up production lines is significantly reduced. Furthermore, the ability to recover and recycle the organic solvent system multiple times without loss of efficacy drastically lowers the recurring cost of goods sold (COGS). This sustainability aspect not only reduces the environmental footprint but also insulates the manufacturing process from volatility in raw solvent pricing, ensuring stable long-term supply for downstream polyimide manufacturers.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of readily available and inexpensive raw materials, such as 2,6-dimethyl-hydroquinone and common nitrohalobenzenes. Unlike competing technologies that may require precious metal catalysts or cryogenic conditions, this method operates under atmospheric pressure using standard industrial bases like potassium carbonate. The elimination of expensive transition metals removes the necessity for costly purification steps to meet trace metal specifications, which is a critical requirement for electronic grade materials. Additionally, the high atom economy and the ability to recycle the solvent mixture mean that waste disposal costs are minimized, contributing to a leaner and more competitive cost structure for the final high-purity aromatic monomer.
- Enhanced Supply Chain Reliability: Supply continuity is a paramount concern for global manufacturers of LCDs and aerospace components. This synthesis route enhances reliability by relying on commodity chemicals that are widely produced and available from multiple global sources, reducing the risk of single-source bottlenecks. The robustness of the reaction conditions—tolerating a range of temperatures and halogenated inputs—means that production can be maintained even if there are temporary shortages of a specific halogenated reagent, as the process can be adapted to use fluorine, chlorine, or bromine derivatives interchangeably. This flexibility ensures that production schedules remain uninterrupted, safeguarding the downstream supply of polyimide precursors against market fluctuations.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen safety and environmental challenges, but this method is inherently designed for industrial expansion. The reaction generates minimal hazardous waste, primarily consisting of inorganic salts that are easy to treat, and avoids the use of chlorinated solvents that are increasingly regulated. The simplicity of the workup procedure, involving precipitation and filtration rather than complex distillation or chromatography, facilitates easy scale-up from pilot plants to full commercial production. This alignment with green chemistry principles not only simplifies regulatory compliance but also future-proofs the manufacturing facility against tightening environmental legislation, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized intermediate. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a transparent view of the technology's capabilities. Understanding these details is crucial for R&D teams evaluating process integration and procurement officers assessing supplier qualifications.
Q: What is the primary application of 2,6-dimethyl-1,4-bis(2,4-dinitrophenoxy)benzene?
A: It serves as a critical precursor for synthesizing highly branched aromatic polybasic primary amines, which are essential monomers for high-performance, heat-resistant polyimide materials used in LCDs and aerospace.
Q: Which halogenated starting materials are compatible with this synthesis?
A: The process is versatile and accommodates 2,4-dinitrofluorobenzene, 2,4-dinitrochlorobenzene, 2,4-dinitrobromobenzene, or 2,4-dinitroiodobenzene, allowing flexibility based on raw material availability and cost.
Q: How does the solvent system contribute to yield optimization?
A: The use of a binary solvent system comprising a water-insoluble solvent (like toluene) and a polar aprotic solvent (like DMF) facilitates effective azeotropic water removal while maintaining high solubility for the ionic intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,6-Dimethyl-1,4-Bis(2,4-Dinitrophenoxy)Benzene Supplier
As the global demand for high-performance polyimides continues to surge, securing a dependable source of high-quality intermediates is essential for maintaining competitive advantage. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging deep expertise in aromatic synthesis to deliver 2,6-dimethyl-1,4-bis(2,4-dinitrophenoxy)benzene with unmatched consistency. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the pilot phase or full-scale manufacturing. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch meets the exacting standards required for electronic and aerospace applications, minimizing the risk of failure in your final polymer products.
We invite you to collaborate with us to optimize your supply chain and reduce your overall material costs. Our technical sales team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating how our efficient synthesis route can improve your bottom line. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to validate our capabilities and quality standards firsthand. Let us be your strategic partner in delivering the next generation of advanced materials.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
