Advanced Synthesis of Fluorinated Polyimide Monomers for High-Performance Electronic Applications
The rapid evolution of the optoelectronics and aerospace sectors has intensified the demand for high-performance polymers capable of withstanding extreme thermal and chemical environments. Central to this advancement is the development of highly branched aromatic fluorine-containing polyimides, which offer superior dielectric properties and thermal stability compared to their non-fluorinated counterparts. A pivotal breakthrough in this domain is documented in patent CN101302163A, which discloses a robust and scalable preparation method for 2,4-bis(2,6-dinitro-4-trifluoromethylphenoxy)benzotrifluoride. This specific intermediate serves as a critical building block for synthesizing aromatic fluorine-containing polybasic primary amines, which are subsequently polymerized into advanced polyimide materials. For R&D directors and procurement strategists alike, understanding the nuances of this synthesis is essential, as it represents a significant leap forward in manufacturing efficiency and product quality for next-generation LCD and microelectronic applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex fluorinated ether intermediates has been plagued by significant technical hurdles that impede commercial viability. Traditional nucleophilic aromatic substitution reactions often require harsh conditions, including the use of expensive phase transfer catalysts or extremely high temperatures that risk decomposing sensitive nitro-functionalized starting materials. Furthermore, conventional protocols frequently suffer from incomplete conversion due to the poor solubility of phenolic salts in non-polar media, leading to heterogeneous reaction mixtures that are difficult to control. This often results in a broad impurity profile, necessitating costly and time-consuming purification steps such as column chromatography or multiple recrystallizations, which drastically reduce overall throughput. Additionally, many legacy methods generate substantial amounts of saline waste and utilize solvents that are difficult to recover, creating environmental liabilities and inflating the total cost of ownership for the final electronic chemical intermediate.
The Novel Approach
The methodology outlined in the referenced patent introduces a sophisticated yet operationally simple strategy that effectively circumvents these historical bottlenecks. By employing a specific binary solvent system that combines a water-insoluble organic solvent with a strong polar aprotic solvent, the process ensures excellent solubility for both the phenolic substrate and the resulting phenoxide anion. This homogeneity facilitates efficient molecular collisions and drives the reaction kinetics favorably without the need for exotic catalysts. Crucially, the integration of a reflux water-separation technique continuously removes the byproduct water from the equilibrium, pushing the reaction towards completion and ensuring consistently high yields. This novel approach not only streamlines the workflow by operating under atmospheric pressure but also guarantees a product purity exceeding 99%, thereby minimizing downstream processing burdens and establishing a new benchmark for reliability in electronic chemical manufacturing.
Mechanistic Insights into Nucleophilic Aromatic Substitution
The core chemical transformation driving this synthesis is a classic nucleophilic aromatic substitution (SnAr), meticulously optimized to accommodate the steric and electronic demands of the trifluoromethyl and nitro substituents. In this mechanism, the 2,4-dihydroxybenzotrifluoride acts as the nucleophile after deprotonation by the salt-forming agent, generating a highly reactive phenoxide anion. This anion attacks the electron-deficient aromatic ring of the 2,6-dinitro-4-trifluoromethyl halobenzene, where the presence of two strongly electron-withdrawing nitro groups ortho to the leaving halogen atom significantly activates the ring towards nucleophilic attack. The reaction proceeds through a Meisenheimer complex intermediate, which is stabilized by the resonance delocalization of the negative charge onto the nitro oxygen atoms, before the expulsion of the halide leaving group restores aromaticity. The choice of halogen (fluorine, chlorine, bromine, or iodine) allows for fine-tuning of the reaction rate, with fluorine often providing superior activation due to its strong inductive effect despite being a poorer leaving group in aliphatic systems.
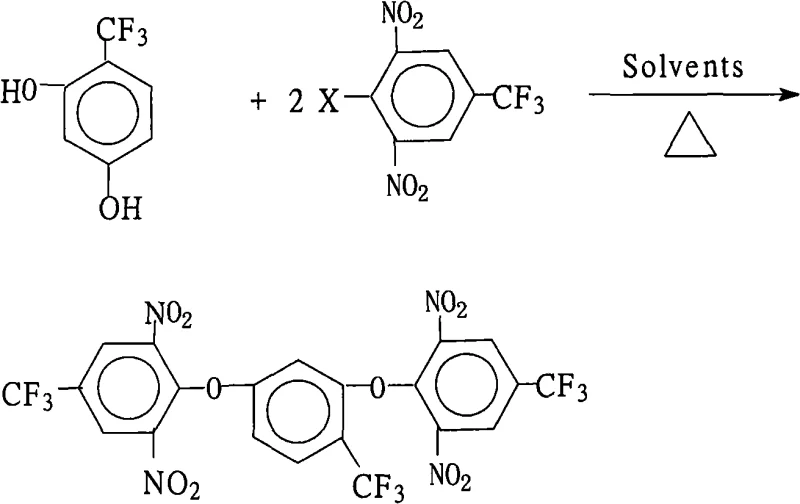
Beyond the primary substitution event, the process design incorporates critical controls to manage potential side reactions and ensure impurity suppression. The use of a mixed solvent system is not merely for solubility; it creates a microenvironment that stabilizes the transition state while preventing the hydrolysis of the sensitive nitro groups, which can occur in purely aqueous or highly protic conditions. Furthermore, the precise control of the molar ratio, maintaining a slight excess of the halobenzene (1.0:2.0-2.2), ensures that both hydroxyl groups on the central ring are fully substituted, preventing the formation of mono-substituted byproducts that could act as chain terminators in subsequent polymerization steps. The rigorous removal of water via azeotropic distillation during reflux is another mechanistic safeguard, as residual water could protonate the phenoxide nucleophile, reverting it to the unreactive phenol and stalling the catalytic cycle. This deep understanding of the reaction dynamics allows for the production of high-purity polyimide monomers with minimal structural defects.
How to Synthesize 2,4-Bis(2,6-Dinitro-4-Trifluoromethylphenoxy)Benzotrifluoride Efficiently
Implementing this synthesis on an industrial scale requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and safety. The process begins with the careful selection of reagents, specifically choosing a halobenzene derivative that balances reactivity with cost-effectiveness, typically the chloro- or bromo- variants. Operators must prepare the binary solvent mixture with precision, ensuring the volume ratio supports both the dissolution of the inorganic base and the organic substrates. The reaction is then initiated by heating the mixture to a controlled reflux temperature, typically ranging between 80°C and 180°C depending on the specific solvent boiling points, while utilizing a Dean-Stark trap or similar apparatus to continuously separate the generated water. Detailed standardized synthetic steps see the guide below.
- Combine 2,4-dihydroxybenzotrifluoride and 2,6-dinitro-4-trifluoromethyl halobenzene in a molar ratio of 1.0: 2.0-2.2 with a salt-forming agent in a mixed organic solvent system.
- Heat the mixture to reflux (80°C-180°C) with continuous water separation for 6 to 18 hours to drive the nucleophilic substitution to completion.
- Concentrate the reaction solution, cool the system, add water to precipitate the solid product, then filter, wash, and dry to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented manufacturing route offers profound strategic benefits that extend far beyond simple chemical yield. The elimination of complex catalytic systems and the reliance on commodity-grade solvents like benzene, toluene, and DMF significantly lowers the raw material entry barrier, ensuring a stable and predictable supply chain even during market fluctuations. Moreover, the ability to recover and recycle the organic solvent system multiple times without degradation of performance translates directly into substantial cost savings and a reduced environmental footprint, aligning with modern green chemistry mandates. This process efficiency allows manufacturers to offer more competitive pricing structures while maintaining healthy margins, making it an attractive option for long-term contracts in the volatile electronic materials sector.
- Cost Reduction in Manufacturing: The economic model of this synthesis is heavily favored by the simplicity of the workup procedure, which avoids expensive purification technologies like preparative HPLC or extensive chromatography. Since the product precipitates directly upon the addition of water to the concentrated reaction mass, the isolation step is reduced to simple filtration and washing, drastically cutting down on labor hours and energy consumption associated with solvent evaporation. Furthermore, the use of inexpensive inorganic bases such as potassium carbonate or sodium carbonate, rather than costly organic bases or transition metal catalysts, removes the need for specialized metal scavenging steps, further streamlining the cost structure. These cumulative efficiencies result in a significantly lower cost of goods sold (COGS), enabling buyers to secure high-purity electronic chemical intermediates at a more sustainable price point.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on exotic or single-source reagents, but this method utilizes widely available starting materials that are produced by multiple global suppliers. The robustness of the reaction conditions, which tolerate a range of halogenated substrates (fluoro, chloro, bromo, iodo), provides procurement teams with the flexibility to switch raw material sources based on availability and price without compromising the final product quality. This adaptability mitigates the risk of production stoppages due to raw material shortages, ensuring that downstream polymer manufacturers receive their critical monomers on schedule. Additionally, the atmospheric pressure operation reduces the need for specialized high-pressure reactors, allowing for production to be scaled across a wider network of qualified contract manufacturing organizations.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial waste disposal intensify, the environmental profile of a chemical process becomes a key determinant of its long-term viability. This synthesis generates minimal saline waste and avoids the use of corrosive acids or bases that require neutralization before discharge, simplifying wastewater treatment protocols. The closed-loop solvent recovery system ensures that volatile organic compound (VOC) emissions are kept to a minimum, facilitating compliance with stringent environmental regulations in major manufacturing hubs. Consequently, scaling this process from pilot plant to multi-ton commercial production involves fewer regulatory hurdles and lower capital expenditure on waste management infrastructure, accelerating the time-to-market for new polyimide formulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this fluorinated intermediate, derived directly from the proprietary data within the patent documentation. These insights are intended to clarify the operational feasibility and quality standards associated with this specific synthetic route for potential partners and technical evaluators. Understanding these details is crucial for integrating this material into your existing supply chain and product development pipelines.
Q: What are the critical reaction conditions for synthesizing this fluorinated monomer?
A: The synthesis requires a biphasic solvent system comprising a water-insoluble solvent (like benzene or toluene) and a polar aprotic solvent (like DMF or NMP), heated to reflux between 80°C and 180°C with active water removal.
Q: How does this method improve upon traditional polyimide precursor synthesis?
A: This patented approach eliminates the need for corrosive reagents and operates under atmospheric pressure, significantly simplifying equipment requirements while achieving yields exceeding 94% and purity above 99%.
Q: Is the solvent system suitable for large-scale industrial recycling?
A: Yes, the process utilizes a recoverable organic solvent system that can be distilled and reused multiple times, drastically reducing waste generation and operational costs for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4-Bis(2,6-Dinitro-4-Trifluoromethylphenoxy)Benzotrifluoride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced electronic materials hinges on the reliability and purity of the underlying chemical building blocks. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 2,4-bis(2,6-dinitro-4-trifluoromethylphenoxy)benzotrifluoride meets the exacting standards required for high-performance polyimide synthesis. Our commitment to quality assurance means that your downstream polymerization processes will proceed without interruption due to feedstock variability.
We invite you to collaborate with us to optimize your material sourcing strategy and achieve significant operational efficiencies. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage potential partners to reach out for specific COA data and route feasibility assessments, allowing you to validate our capabilities against your internal standards before committing to a supply agreement. Let us be your strategic partner in delivering the high-quality fluorinated intermediates necessary to power the next generation of electronic displays and aerospace composites.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
