Revolutionizing 2-Methylpyrrolidine Production: A Green, Metal-Free Hydroamination Strategy for Scale-Up
Revolutionizing 2-Methylpyrrolidine Production: A Green, Metal-Free Hydroamination Strategy for Scale-Up
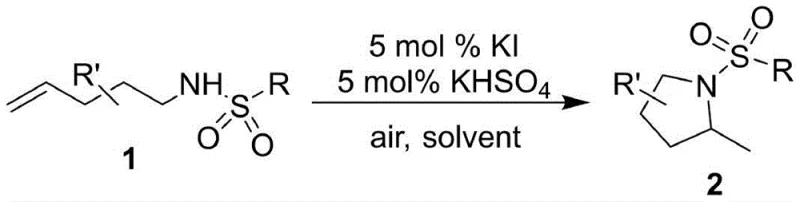
The landscape of nitrogen heterocycle synthesis is undergoing a significant paradigm shift, driven by the urgent need for sustainable and economically viable manufacturing processes. Patent CN112479968A introduces a groundbreaking methodology for the preparation of 2-methylpyrrolidine compounds, a structural motif ubiquitous in high-value pharmaceutical intermediates and bioactive natural products. This innovation leverages a dual-catalyst system comprising potassium iodide (KI) and potassium bisulfate (KHSO4) to facilitate an intramolecular hydroamination reaction under remarkably mild conditions. Unlike conventional approaches that demand rigorous exclusion of moisture or expensive noble metals, this technique operates efficiently in an open-air environment at room temperature. For R&D directors and process chemists, this represents a substantial reduction in operational complexity, while supply chain managers will recognize the immediate implications for cost stability and raw material security. The ability to transform readily available 4-pentenyl sulfonamides into cyclic amines with high atom economy positions this technology as a cornerstone for next-generation fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of pyrrolidine rings via hydroamination has been plagued by significant thermodynamic and kinetic barriers. The intrinsic electron repulsion between the lone pair of the nitrogen atom and the electron-rich pi-system of the olefin renders the uncatalyzed reaction virtually impossible. To overcome this, early methodologies relied heavily on strong bases to generate nucleophilic amide anions, which often resulted in poor functional group tolerance and harsh reaction profiles. More modern approaches have turned to lanthanide and Group IVB transition metal complexes, or late transition metals such as gold, palladium, and platinum. While effective, these catalytic systems introduce severe logistical burdens; they are frequently highly sensitive to air and moisture, necessitating expensive inert gas manifolds and anhydrous solvents. Furthermore, the residual presence of toxic heavy metals in the final API intermediate requires rigorous and costly purification protocols to meet stringent regulatory limits, creating a bottleneck in the commercialization of drug candidates.
The Novel Approach
The methodology disclosed in CN112479968A circumvents these historical challenges by utilizing an inexpensive, earth-abundant halide salt system. By employing 5 mol% KI and 5 mol% KHSO4, the reaction achieves efficient cyclization without the need for transition metals. This approach not only drastically reduces the raw material cost but also simplifies the engineering requirements for the reactor, as the process tolerates ambient atmosphere and moisture. The use of benign solvents like acetone/water or acetonitrile/water mixtures further aligns the process with green chemistry principles, minimizing the environmental footprint associated with volatile organic compound (VOC) emissions. For procurement teams, this shift away from precious metals mitigates the risk of price volatility associated with commodities like palladium or gold, ensuring a more predictable cost structure for long-term production campaigns.
Mechanistic Insights into KI/KHSO4-Catalyzed Cyclization
The mechanistic elegance of this transformation lies in the synergistic interaction between the iodide anion and the acidic proton source. While the precise catalytic cycle may involve the in situ generation of reactive iodine species or acid-mediated activation of the alkene, the net result is a highly efficient activation of the carbon-carbon double bond towards nucleophilic attack by the pendant sulfonamide nitrogen. The reaction proceeds through a concerted or stepwise addition that favors the formation of the five-membered pyrrolidine ring with excellent regioselectivity. The presence of KHSO4 likely serves to protonate the intermediate or activate the leaving group dynamics, ensuring rapid turnover of the catalyst. This mechanism is robust across a wide range of substrates, accommodating various sulfonamide protecting groups (Ts, Ms, Bs, Ns) and diverse substitution patterns on the carbon backbone, including bulky diphenyl groups and spirocyclic systems.
From an impurity control perspective, this catalytic system offers distinct advantages over transition metal alternatives. The absence of metal-ligand complexes eliminates the formation of metal-associated side products or decomposition pathways often seen with sensitive organometallic catalysts. The reaction profile described in the patent indicates high conversion rates, often exceeding 90% yield, which simplifies the downstream purification burden. The inorganic nature of the catalyst allows for potential removal via simple aqueous workup, although column chromatography is described for analytical purity. For quality assurance teams, the simplicity of the impurity profile translates to easier method validation and more consistent batch-to-batch reproducibility, a critical factor when scaling from gram-scale discovery to multi-kilogram commercial supply.
How to Synthesize 2-Methylpyrrolidine Efficiently
The operational simplicity of this synthesis makes it highly accessible for both laboratory optimization and industrial implementation. The process begins with the direct charging of the 4-pentenyl sulfonamide substrate along with the catalytic amounts of KI and KHSO4 into a standard reaction vessel. The use of a mixed solvent system, specifically acetone/water or acetonitrile/water, provides the necessary solubility for both the organic substrate and the inorganic salts while maintaining a homogeneous reaction medium. The reaction is allowed to stir at room temperature for approximately 10 hours under open air, removing the need for specialized pressure vessels or cryogenic cooling. Following the reaction, the solvent is removed under reduced pressure, and the crude product is purified. Detailed standardized operating procedures for this transformation are provided below to ensure consistent replication of the high yields reported in the patent literature.
- Charge a reactor with 4-pentenyl sulfonamide substrate, 5 mol% KI, and 5 mol% KHSO4 under open air conditions.
- Stir the mixture in acetone/water or acetonitrile/water solvent at room temperature for approximately 10 hours.
- Remove solvent under reduced pressure and purify the crude residue via silica gel column chromatography to isolate the target 2-methylpyrrolidine.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for the economic viability and continuity of chemical supply chains, the adoption of this KI-catalyzed hydroamination route offers transformative benefits. The primary advantage lies in the drastic simplification of the bill of materials; replacing expensive, supply-constrained noble metals with commodity chemicals like potassium iodide and potassium bisulfate fundamentally alters the cost basis of the molecule. This shift not only lowers the direct material cost but also reduces the dependency on geopolitical regions that dominate the mining and refining of precious metals. Furthermore, the operational conditions—room temperature and open air—significantly lower the energy consumption and capital expenditure required for reactor infrastructure, as there is no need for complex inert gas systems or high-pressure ratings.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts such as gold, palladium, or platinum removes a major cost driver from the manufacturing process. These noble metals are subject to significant market volatility and require expensive recovery or scavenging processes to meet regulatory standards for residual metals in pharmaceuticals. By utilizing inexpensive inorganic salts, the process achieves substantial cost savings without compromising reaction efficiency. Additionally, the high yields reported (often above 90%) minimize waste generation and maximize the throughput of the starting material, further enhancing the overall economic efficiency of the production campaign.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including 4-pentenyl sulfonamides, KI, and KHSO4, are commercially available in bulk quantities from multiple global suppliers. This diversity of supply sources mitigates the risk of single-source bottlenecks that often plague specialized ligand or catalyst markets. The robustness of the reaction to air and moisture also means that storage and handling requirements are less stringent, reducing the risk of material degradation during logistics and warehousing. This reliability ensures a steady flow of intermediates, supporting uninterrupted manufacturing schedules for downstream API production.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new safety and environmental challenges, but this methodology is inherently designed for scalability. The exotherm is manageable at room temperature, and the use of water-miscible solvents like acetone facilitates easier solvent recovery and waste treatment compared to chlorinated or aromatic solvents. The avoidance of heavy metals simplifies the environmental compliance landscape, reducing the burden of hazardous waste disposal and effluent treatment. This green profile aligns perfectly with the increasing regulatory pressure on pharmaceutical manufacturers to adopt sustainable practices, making it an attractive option for companies aiming to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydroamination technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for project managers evaluating the feasibility of integrating this route into existing production workflows.
Q: Why is the KI/KHSO4 catalytic system superior to traditional transition metal catalysts?
A: Traditional methods often rely on expensive and air-sensitive lanthanide or noble metal catalysts (Au, Pd, Pt). The KI/KHSO4 system operates under open air at room temperature, eliminating the need for inert atmospheres and costly metal removal steps.
Q: What are the typical reaction conditions for this hydroamination process?
A: The reaction proceeds efficiently at room temperature using a mixed solvent system of acetone/water or acetonitrile/water. It requires only 5 mol% loading of both KI and KHSO4 catalysts.
Q: Is column chromatography strictly required for purification?
A: While the patent describes column chromatography for high purity, the inorganic catalysts (KI/KHSO4) can potentially be removed via aqueous extraction due to their solubility differences, offering a pathway to simplify downstream processing for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methylpyrrolidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting innovative synthetic routes like the KI-catalyzed hydroamination described in CN112479968A. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our facility is equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of 2-methylpyrrolidine intermediate meets the highest standards required for pharmaceutical applications. We are committed to leveraging our technical expertise to optimize this green chemistry approach for your specific supply chain needs.
We invite you to engage with our technical team to explore how this cost-effective methodology can be tailored to your project requirements. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this metal-free protocol. Our experts are ready to provide specific COA data and route feasibility assessments, helping you make informed decisions that drive value and efficiency in your drug development pipeline. Contact our technical procurement team today to initiate a discussion on securing a reliable, high-quality supply of these critical nitrogen heterocycles.
