Revolutionizing High-Performance Polymer Production: A Deep Dive into Polythioetherimide Synthesis
The landscape of high-performance engineering plastics is undergoing a significant transformation driven by the need for materials that balance thermal stability with processability. Patent CN101531758A introduces a groundbreaking methodology for the preparation of polythioetherimide resins, addressing critical bottlenecks in traditional polymer synthesis. This technology leverages a direct coupling strategy using elemental sulfur, bypassing the cumbersome preparation of thioether dianhydrides that has long plagued the industry. By utilizing mono-substituted phthalic anhydride isomers, such as chlorinated or nitro-substituted variants, as direct feedstocks, the process achieves a remarkable simplification of the synthetic route. The resulting polymers exhibit exceptional comprehensive properties, including high toughness, low melt viscosity, and superior heat resistance, making them ideal candidates for demanding applications in aerospace, automotive, and electronic components. 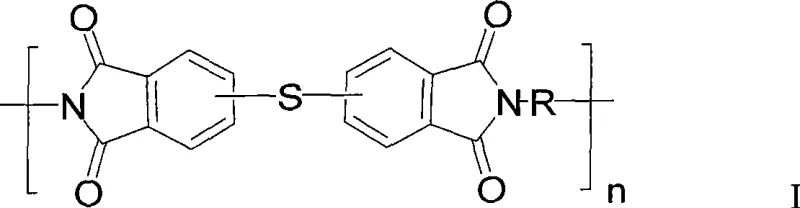
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of polythioetherimides has been hindered by a multi-step pathway that is both economically and operationally inefficient. Traditional methods typically require the initial preparation of thioether-containing aromatic tetra-acid dianhydrides. This involves reacting nitro- or chloro-substituted phthalimides with alkali metal sulfides like sodium sulfide, followed by a rigorous sequence of hydrolysis, acidification, and dehydration to obtain the dianhydride monomer. Each of these steps introduces potential yield losses, increases solvent consumption, and generates significant waste streams. Furthermore, the handling of alkali metal sulfides poses safety challenges due to their tendency to oxidize and deliquesce, requiring strict anhydrous conditions that escalate production costs. The cumulative effect of these繁琐 steps results in a high-cost final product with limited scalability, restricting its adoption in cost-sensitive industrial sectors despite its superior performance characteristics.
The Novel Approach
The innovative protocol described in the patent data fundamentally reengineers this workflow by eliminating the dianhydride synthesis entirely. Instead, it employs a direct coupling polymerization between a disubstituted phthalimide intermediate and elemental sulfur. This approach reduces the synthetic sequence by more than three distinct operational steps, drastically cutting down on reaction time and energy consumption. By using elemental sulfur as the coupling agent, the process avoids the instability issues associated with alkali metal sulfides, allowing for more robust stoichiometric control. The reaction can be conducted in a variety of solvent systems, including polar aprotic solvents or even under melt conditions, offering unparalleled flexibility for process optimization. This streamlined methodology not only lowers the barrier to entry for manufacturing but also enhances the environmental profile of the production cycle by minimizing waste generation and solvent usage.
Mechanistic Insights into Direct Sulfur Coupling Polymerization
The core of this technological advancement lies in the nucleophilic substitution mechanism facilitated by elemental sulfur in the presence of reducing agents. In the first stage, mono-substituted phthalic anhydrides react with organic diamines at temperatures ranging from 100°C to 350°C to form the disubstituted phthalimide intermediate. This step is critical for establishing the imide ring structure which provides the thermal backbone of the polymer.  In the subsequent polymerization stage, the disubstituted phthalimide undergoes a coupling reaction with sulfur. The reducing agents, which can range from formates and hydrazines to metal powders like iron or zinc, activate the sulfur species, enabling them to displace the halogen or nitro substituents on the phthalimide ring. This creates the flexible thioether linkage (-S-) that connects the rigid imide units. The presence of catalysts and auxiliaries, such as carbonates or halides, further accelerates this substitution, ensuring high conversion rates and consistent chain growth. The ability to tune the ratio of 3-substituted to 4-substituted isomers in the feedstock allows for precise control over the polymer's crystallinity and solubility, offering a powerful tool for tailoring material properties to specific end-use requirements.
In the subsequent polymerization stage, the disubstituted phthalimide undergoes a coupling reaction with sulfur. The reducing agents, which can range from formates and hydrazines to metal powders like iron or zinc, activate the sulfur species, enabling them to displace the halogen or nitro substituents on the phthalimide ring. This creates the flexible thioether linkage (-S-) that connects the rigid imide units. The presence of catalysts and auxiliaries, such as carbonates or halides, further accelerates this substitution, ensuring high conversion rates and consistent chain growth. The ability to tune the ratio of 3-substituted to 4-substituted isomers in the feedstock allows for precise control over the polymer's crystallinity and solubility, offering a powerful tool for tailoring material properties to specific end-use requirements.
Impurity control is inherently superior in this system due to the avoidance of aqueous workups associated with dianhydride hydrolysis. Traditional routes often leave behind salt residues or incomplete hydrolysis byproducts that can act as degradation initiators at high temperatures. In contrast, the direct coupling method produces a cleaner polymer matrix. The use of chain end-capping agents, such as aryl compounds with halogen or nitro groups, effectively terminates the polymerization at the desired molecular weight, preventing uncontrolled cross-linking or gelation. This results in a resin with a narrow polydispersity index, typically between 1.8 and 5.4, which translates to consistent melt flow behavior during processing. The inherent viscosity of the final product, measurable in m-cresol, serves as a reliable indicator of this structural integrity, with values typically falling between 0.13 dL/g and 1.90 dL/g, confirming the formation of high molecular weight chains suitable for load-bearing applications.
How to Synthesize Polythioetherimide Efficiently
The synthesis of this advanced polymer resin is designed for industrial feasibility, balancing high yield with operational simplicity. The process begins with the condensation of phthalic anhydride derivatives and diamines, followed by the critical sulfur coupling step. Detailed standard operating procedures regarding specific solvent ratios, heating ramps, and purification protocols are essential for reproducibility. For a comprehensive guide on executing this synthesis with precision, please refer to the standardized protocol below.
- React mono-substituted phthalic anhydride isomers with organic diamines at 100-350°C to form disubstituted phthalimide intermediates.
- Perform coupling polymerization of the phthalimide intermediate with elemental sulfur using reducing agents and catalysts at 60-260°C.
- Control molecular weight using chain end-capping agents and isolate the resin via precipitation and washing.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement strategists and supply chain directors, the shift to this novel synthesis route represents a substantial opportunity for cost optimization and risk mitigation. The elimination of the dianhydride intermediate step removes a significant portion of the variable costs associated with labor, energy, and solvent recovery. Moreover, the reliance on commodity chemicals like elemental sulfur and common phthalic anhydrides insulates the supply chain from the volatility often seen with specialized fine chemical intermediates. This robustness ensures a more predictable lead time and enhances the overall reliability of the supply network, crucial for maintaining continuous production lines in downstream manufacturing sectors.
- Cost Reduction in Manufacturing: The most immediate financial benefit stems from the drastic reduction in process steps. By skipping the hydrolysis, acidification, and dehydration stages required for dianhydride preparation, manufacturers can achieve significant savings in utility costs and reactor occupancy time. The use of inexpensive elemental sulfur as a coupling agent, compared to costly and sensitive alkali metal sulfides, further drives down the raw material bill of materials. Additionally, the simplified workflow reduces the need for complex equipment and extensive quality control testing at intermediate stages, leading to a leaner and more cost-effective production model that improves overall margin potential without compromising product quality.
- Enhanced Supply Chain Reliability: The raw materials for this process, specifically chlorinated or nitro-substituted phthalic anhydrides, are widely available commodity chemicals with established global supply networks. This abundance mitigates the risk of supply disruptions that can occur with niche, custom-synthesized monomers. The process tolerance for isomer mixtures (3- and 4- substituted) means that manufacturers do not need to invest in expensive separation technologies to obtain pure isomers, allowing for the use of lower-grade, more readily available feedstocks. This flexibility ensures a steady flow of materials even during market tightness, securing the continuity of supply for critical engineering plastic applications.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this technology offers distinct advantages. The potential for melt polymerization eliminates the need for large volumes of organic solvents, aligning with increasingly stringent environmental regulations regarding VOC emissions. The reduction in waste streams, particularly saline wastewater from hydrolysis steps, simplifies effluent treatment and lowers disposal costs. The process is inherently scalable, capable of transitioning from laboratory bench scales to multi-ton commercial production with minimal re-engineering. This scalability supports rapid capacity expansion to meet growing market demand for high-performance thermoplastics in emerging sectors like electric vehicles and renewable energy infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this polythioetherimide technology. These insights are derived directly from the patent specifications and are intended to clarify the operational parameters and performance expectations for potential adopters. Understanding these details is crucial for evaluating the feasibility of integrating this material into your existing product portfolios.
Q: What are the primary advantages of this polythioetherimide synthesis method over traditional routes?
A: This method eliminates the need to synthesize thioether dianhydride intermediates, saving more than three reaction steps including hydrolysis and dehydration. It utilizes inexpensive elemental sulfur instead of unstable alkali metal sulfides, significantly reducing raw material costs and operational complexity.
Q: What thermal properties can be expected from the resulting polythioetherimide resin?
A: The synthesized resin exhibits excellent heat resistance with a glass transition temperature (Tg) ranging from approximately 200°C to 350°C. It also demonstrates low melt viscosity, making it highly suitable for melt processing techniques like injection molding and extrusion.
Q: Can the molecular weight of the polymer be controlled during this process?
A: Yes, the molecular weight is adjustable through the use of specific chain end-capping agents. The process allows for the production of resins with weight average molecular weights between 3,000 and 140,000, tailored to specific application requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polythioetherimide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced polymer synthesis route for the high-performance materials sector. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this patented methodology into commercial reality. Our facilities are equipped to handle complex polymerization reactions with precision, ensuring that every batch meets stringent purity specifications and performance criteria. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, providing our clients with the confidence that their supply needs will be met reliably and consistently. Our rigorous QC labs employ state-of-the-art analytical techniques, including GPC and DSC, to verify molecular weight distribution and thermal properties, guaranteeing that the delivered resin performs exactly as designed in your final applications.
We invite forward-thinking organizations to collaborate with us to optimize their material supply chains. Whether you are looking to reduce costs in engineering plastic manufacturing or secure a stable source of high-purity polythioetherimide, our team is ready to assist. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to understand how we can support your next-generation product development initiatives.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
