Industrial Scale-Up of 3,3'-Dinitro-4,4'-Bis(Phenoxy) Biphenyl for Advanced Polyimide Applications
Industrial Scale-Up of 3,3'-Dinitro-4,4'-Bis(Phenoxy) Biphenyl for Advanced Polyimide Applications
The rapid evolution of the electronic materials sector demands intermediates that offer exceptional thermal stability and dielectric properties, particularly for next-generation display technologies and aerospace composites. Patent CN101397253A introduces a robust and highly efficient methodology for the preparation of 3,3'-dinitro-4,4'-bis(2,6-dinitro-4-trifluoromethylphenoxy)biphenyl, a sophisticated aromatic ether that serves as a pivotal building block for advanced fluorinated polyimides. This specific intermediate enables the synthesis of highly branched aromatic fluorine-containing polybasic primary amines, which are indispensable for creating polyimide systems with superior temperature resistance and mechanical strength. For R&D directors and procurement specialists seeking a reliable polyimide monomer supplier, understanding the nuances of this synthetic route is critical for securing a stable supply chain of high-performance electronic chemicals.
The significance of this patent lies not only in the chemical structure itself but in the reproducible industrial process that overcomes common hurdles associated with multi-nitro substituted aromatic etherifications. Traditional methods for synthesizing such complex biphenyl derivatives often suffer from incomplete conversion, difficult purification steps, or the generation of hazardous waste streams that complicate regulatory compliance. By contrast, the disclosed method leverages a precise stoichiometric balance and a dual-solvent system to drive the reaction to near-quantitative completion while maintaining an environmentally friendly profile. This technological breakthrough positions the compound as a viable candidate for cost reduction in electronic chemical manufacturing, offering a pathway to high-purity materials without the prohibitive costs typically associated with specialty fluorinated intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of highly nitrated aromatic ethers has been plagued by kinetic challenges and side reactions that compromise both yield and product integrity. Conventional nucleophilic aromatic substitution reactions involving poly-nitro substrates often require harsh conditions that can lead to the degradation of sensitive functional groups or the formation of intractable tars that are difficult to separate from the desired product. Furthermore, many legacy processes rely on single-solvent systems that fail to adequately solubilize both the phenolic starting materials and the resulting ether products, leading to heterogeneous reaction mixtures that proceed slowly and unevenly. These inefficiencies result in significant batch-to-batch variability, forcing manufacturers to implement extensive and costly purification protocols such as column chromatography or multiple recrystallizations to meet the stringent purity specifications required for electronic grade materials.
In addition to technical limitations, conventional approaches often neglect the economic and environmental implications of solvent usage and waste generation. Processes that utilize excessive amounts of polar aprotic solvents without effective recovery mechanisms create substantial disposal burdens and increase the overall cost of goods sold. The lack of an integrated water removal strategy in standard etherification protocols can also shift the equilibrium backward, preventing the reaction from reaching high conversion rates and leaving unreacted starting materials that contaminate the final crystal lattice. For supply chain heads focused on sustainability and operational efficiency, these drawbacks represent significant bottlenecks that hinder the commercial scale-up of complex aromatic ethers, necessitating a shift toward more optimized and circular manufacturing paradigms.
The Novel Approach
The methodology outlined in CN101397253A represents a paradigm shift by introducing a synergistic solvent system and a controlled water-removal strategy that fundamentally alters the reaction thermodynamics. By employing a mixture of a water-insoluble organic solvent, such as toluene or xylene, combined with a strong polar aprotic solvent like N,N-dimethylformamide (DMF) or N-methyl-2-pyrrolidone (NMP), the process ensures optimal solubility for all reactants throughout the reaction timeline. This dual-solvent approach facilitates a homogeneous reaction environment that accelerates the nucleophilic attack of the phenoxide anion on the electron-deficient aromatic ring, thereby significantly enhancing the reaction rate and selectivity. Moreover, the integration of a reflux condensation setup designed for water separation effectively drives the equilibrium forward by continuously removing the water byproduct generated during the salt formation and substitution steps.
Beyond the reaction kinetics, the novel approach excels in its downstream processing simplicity, which is a key factor for reducing lead time for high-purity polyimide precursors. Instead of complex extraction sequences, the product is isolated through a straightforward concentration and precipitation step where the addition of water causes the highly pure crystalline product to crash out of the solution. This elegant workup procedure not only minimizes solvent loss but also allows for the direct recycling of the mother liquor solvents, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing process. The result is a streamlined production workflow that delivers yields consistently above 90% and purities exceeding 99%, providing a reliable foundation for the mass production of high-performance polyimide monomers needed in the LCD and semiconductor industries.
Mechanistic Insights into Nucleophilic Aromatic Substitution
The core chemical transformation driving this synthesis is a classic Nucleophilic Aromatic Substitution (SnAr), facilitated by the powerful electron-withdrawing effects of the nitro groups positioned ortho and para to the leaving halogen atom. In the reaction mechanism, the base—typically an alkali metal carbonate or hydroxide—first deprotonates the hydroxyl groups of the 3,3'-dinitro-4,4'-dihydroxybiphenyl to generate a highly nucleophilic phenoxide anion. This anion then attacks the electron-deficient carbon atom bearing the halogen on the 2,6-dinitro-4-trifluoromethyl halobenzene ring. The presence of two nitro groups ortho to the reaction site and one para, along with the trifluoromethyl group, creates a profound deficiency of electron density on the aromatic ring, stabilizing the negatively charged Meisenheimer complex intermediate and lowering the activation energy for the substitution. This electronic activation is crucial for allowing the reaction to proceed efficiently under relatively mild thermal conditions compared to non-activated systems.
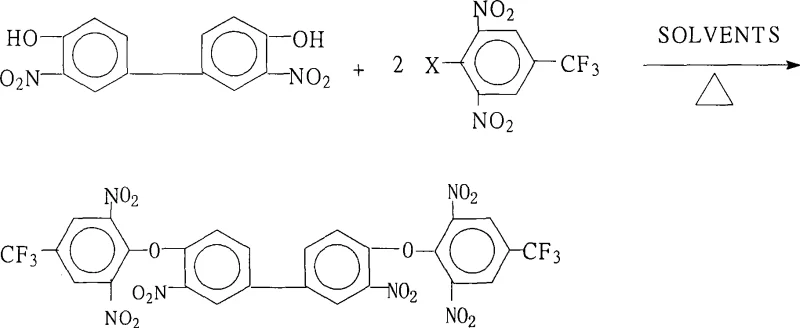
Controlling the impurity profile in such a highly nitrated system requires precise management of the reaction stoichiometry and temperature profile to prevent over-reaction or hydrolysis of the sensitive nitro groups. The patent specifies a molar ratio of diol to halobenzene between 1.0:2.0 and 1.0:2.2, ensuring a slight excess of the electrophile to drive the double substitution to completion without leaving significant amounts of the mono-substituted intermediate. Additionally, the choice of base and its molar ratio (0.80 to 8.00 relative to the diol) plays a pivotal role in scavenging the acid byproduct (HX) formed during the substitution, preventing acid-catalyzed decomposition of the product. By maintaining the reaction temperature between 80°C and 180°C, the process provides sufficient thermal energy to overcome the steric hindrance of the bulky substituents while avoiding thermal degradation, resulting in a clean impurity spectrum that simplifies the final purification steps.
How to Synthesize 3,3'-Dinitro-4,4'-Bis(Phenoxy) Biphenyl Efficiently
Executing this synthesis on a commercial scale requires strict adherence to the optimized parameters defined in the patent to ensure consistent quality and safety. The process begins with the careful charging of reactors with the specific solvent blends and reagents, followed by a controlled heating ramp to initiate the azeotropic removal of water. Operators must monitor the water separation closely to determine the reaction endpoint, as the cessation of water collection indicates the completion of the etherification. Following the reaction, the concentration and precipitation steps must be managed to control crystal morphology and particle size, which can impact downstream filtering and drying efficiency. For a detailed breakdown of the specific equipment requirements, safety protocols, and step-by-step operational parameters, please refer to the standardized synthesis guide below.
- Combine 3,3'-dinitro-4,4'-dihydroxybiphenyl and 2,6-dinitro-4-trifluoromethyl halobenzene in a molar ratio of 1.0: 2.0~2.2 with a salt-forming agent in a mixed solvent system.
- Heat the mixture to reflux (80°C–180°C) with water separation for 6 to 18 hours to drive the nucleophilic aromatic substitution to completion.
- Concentrate the reaction liquid, cool, add water to precipitate the solid product, then filter, wash with hot water, and dry to obtain crystals with >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis route offers tangible strategic advantages that extend beyond mere chemical yield. The process is designed with industrial practicality in mind, utilizing commodity chemicals and solvents that are readily available in the global market, thereby mitigating the risk of raw material shortages that often plague specialty chemical supply chains. The ability to operate under atmospheric pressure eliminates the need for expensive high-pressure reactors, reducing capital expenditure (CAPEX) for manufacturing facilities and lowering the barrier to entry for scaling production. Furthermore, the simplicity of the workup procedure, which avoids complex extractions and relies on basic filtration, translates directly into reduced labor costs and shorter cycle times, enabling faster turnaround from raw material intake to finished goods shipment.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the efficient recovery and recycling of the organic solvent system. Since the solvents used, such as toluene and DMF, are recovered through distillation and reused in subsequent batches, the consumption of fresh solvent per kilogram of product is drastically minimized. This closed-loop solvent management not only lowers direct material costs but also reduces the expenses associated with hazardous waste disposal and environmental compliance. Additionally, the high selectivity of the reaction means that fewer resources are wasted on purifying side products, leading to a more efficient utilization of expensive fluorinated starting materials and a lower overall cost of goods sold for the final polyimide monomer.
- Enhanced Supply Chain Reliability: The robustness of the synthetic method contributes to a more resilient supply chain by ensuring consistent output quality regardless of minor fluctuations in operating conditions. The use of stable, non-corrosive reagents and the absence of sensitive catalysts that require inert atmosphere handling simplify the logistics of storage and transportation. This operational stability allows manufacturers to maintain steady production schedules and meet tight delivery windows, which is critical for customers in the fast-paced electronics industry who rely on just-in-time inventory models. By partnering with a supplier utilizing this technology, buyers can secure a dependable source of high-purity intermediates that supports their own production continuity without the risk of quality-related disruptions.
- Scalability and Environmental Compliance: From a sustainability perspective, the process aligns well with modern environmental regulations due to its low waste generation and energy-efficient design. The absence of heavy metal catalysts or toxic reagents simplifies the treatment of effluent streams, making it easier for manufacturing sites to obtain and maintain necessary environmental permits. The scalability of the reaction is proven by its successful demonstration in multi-gram to kilogram scales within the patent examples, indicating a clear path to ton-scale production without fundamental changes to the chemistry. This scalability ensures that as demand for fluorinated polyimides grows in sectors like 5G communications and flexible displays, the supply of this key intermediate can expand seamlessly to meet market needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized biphenyl derivative. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders make informed decisions about integrating this material into their supply chains and product development pipelines.
Q: What is the primary application of 3,3'-dinitro-4,4'-bis(2,6-dinitro-4-trifluoromethylphenoxy)biphenyl?
A: This compound serves as a critical precursor for synthesizing highly branched aromatic fluorine-containing polybasic primary amines, which are essential monomers for producing high-performance fluorinated polyimides used in LCDs and aerospace materials.
Q: How does the patented method improve yield compared to traditional etherification?
A: By utilizing a specific mixed solvent system of water-insoluble organics and polar aprotic solvents alongside azeotropic water removal, the process achieves yields exceeding 97% and purity levels up to 99.7%, significantly reducing downstream purification costs.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the reaction operates under atmospheric pressure without corrosive byproducts, uses recyclable solvents like toluene and DMF, and involves simple filtration workups, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3'-Dinitro-4,4'-Bis(Phenoxy) Biphenyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance intermediates play in the advancement of electronic materials and aerospace technologies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ state-of-the-art analytical techniques to verify every batch against the highest industry standards. Whether you require custom synthesis modifications or bulk supply of standard intermediates, our infrastructure is designed to support your long-term growth and innovation goals.
We invite you to collaborate with us to optimize your sourcing strategy and achieve significant operational efficiencies. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage potential partners to reach out for specific COA data and route feasibility assessments to verify how our capabilities align with your project milestones. Let us help you secure a competitive advantage in the market with reliable access to premium chemical building blocks.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
