Advanced Synthesis of 3,5-Bis(2,4-Diaminophenoxy)-2-Naphthoic Acid for Next-Gen Electronic Materials
Advanced Synthesis of 3,5-Bis(2,4-Diaminophenoxy)-2-Naphthoic Acid for Next-Gen Electronic Materials
The rapid evolution of the global electronics industry, particularly in the sectors of flexible displays and high-frequency communication devices, has placed unprecedented demands on the performance of underlying substrate materials. At the heart of this technological shift lies the critical need for advanced polyimide resins that can withstand extreme thermal processing while maintaining superior electrical insulation properties. Patent CN101245022A introduces a groundbreaking preparation method for 3,5-bis(2,4-diaminophenoxy)-2-naphthoic acid, a specialized aromatic diamine monomer that serves as a foundational building block for these next-generation polymers. This proprietary synthesis route leverages a sophisticated palladium-catalyzed reduction strategy to convert nitro-precursors into high-purity amines with exceptional efficiency. By addressing the longstanding challenges of impurity control and process scalability, this technology offers a robust solution for manufacturers seeking to enhance the reliability of their supply chains for high-performance electronic chemicals. The method not only ensures product purity levels exceeding 99% but also streamlines the operational workflow, making it an ideal candidate for large-scale industrial adoption in the competitive landscape of optoelectronic material production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex aromatic diamines like 3,5-bis(2,4-diaminophenoxy)-2-naphthoic acid has been plagued by significant technical hurdles that hindered widespread commercial adoption. Traditional reduction methods often relied on harsh chemical environments, such as iron powder in acidic media, which generated substantial quantities of hazardous solid waste and required extensive downstream purification to remove metal residues. Alternatively, high-pressure catalytic hydrogenation, while cleaner, necessitated expensive reactor infrastructure capable of withstanding extreme pressures, thereby increasing capital expenditure and operational risk for chemical manufacturers. Furthermore, conventional processes frequently struggled with selectivity issues, leading to the formation of partially reduced intermediates or over-reduced byproducts that compromised the thermal stability of the final polyimide resin. These inefficiencies resulted in prolonged production cycles, elevated energy consumption, and inconsistent batch-to-batch quality, creating bottlenecks for procurement teams aiming to secure reliable sources of high-purity polyimide intermediates. The environmental footprint of these legacy methods also posed compliance challenges, as the disposal of heavy metal sludge and acidic wastewater became increasingly regulated under global sustainability mandates.
The Novel Approach
In stark contrast to these legacy techniques, the novel approach detailed in the patent data utilizes a mild yet highly effective liquid-phase reduction system driven by hydrazine hydrate and a heterogeneous palladium on carbon (Pd/C) catalyst. This methodology operates under atmospheric pressure conditions within a moderate temperature range of 70°C to 85°C, significantly lowering the energy barrier and safety risks associated with the synthesis. The use of saturated fatty alcohols as the solvent medium provides an optimal environment for the dissolution of the organic substrate while facilitating efficient mass transfer during the catalytic cycle. A distinct advantage of this system is its ability to achieve near-quantitative conversion without the generation of toxic heavy metal waste, as the Pd/C catalyst can be easily separated via hot filtration and potentially recycled. The process design inherently minimizes the formation of side products, ensuring that the crude product obtained after precipitation with water already meets stringent purity specifications without the need for recrystallization or column chromatography. This streamlined workflow represents a paradigm shift in cost reduction in electronic chemical manufacturing, allowing producers to deliver premium-grade monomers with drastically simplified post-processing requirements.
Mechanistic Insights into Pd/C Catalyzed Nitro Reduction
The core of this synthetic breakthrough lies in the intricate mechanistic pathway of the catalytic transfer hydrogenation facilitated by the palladium surface. In this reaction system, hydrazine hydrate acts as the hydrogen donor, decomposing on the active sites of the Pd/C catalyst to generate nascent hydrogen species in situ. These reactive hydrogen atoms are then transferred to the nitro groups (-NO2) located at the 2 and 4 positions of the phenoxy rings attached to the naphthalene core. The reduction proceeds through a series of well-defined intermediates, typically involving the formation of nitroso and hydroxylamine species before finally arriving at the stable primary amine (-NH2) functionality. The presence of the electron-withdrawing carboxylic acid group on the naphthalene ring and the ether linkages requires a catalyst with sufficient activity to overcome potential deactivation effects, which the selected Pd/C formulation effectively addresses. The choice of solvent plays a crucial role in stabilizing these transition states and preventing the aggregation of catalyst particles, ensuring sustained catalytic activity throughout the 1 to 5-hour reaction window. Understanding this mechanism is vital for R&D directors aiming to optimize reaction parameters for maximum throughput while maintaining the structural integrity of the sensitive naphthoic acid backbone.
Controlling the impurity profile is equally critical, as even trace amounts of unreacted nitro compounds or hydroxylamine intermediates can act as defect sites in the final polyimide polymer chain, severely degrading its dielectric properties and thermal resistance. The patented process mitigates this risk through the precise control of the hydrazine-to-substrate molar ratio, maintained between 1:20 and 1:50, which ensures a vast excess of reducing agent to drive the equilibrium completely towards the diamine product. Additionally, the hot filtration step immediately following the reaction serves a dual purpose: it removes the spent catalyst to prevent any further unwanted catalytic activity during cooling, and it eliminates insoluble particulate matter that could act as nucleation sites for impurities. The subsequent addition of pure water to the cooled mother liquor induces crystallization based on solubility differences, effectively excluding soluble organic byproducts from the crystal lattice. This crystallization-driven purification mechanism is what allows the process to achieve purity levels greater than 99% directly from the reactor, bypassing the need for energy-intensive distillation or solvent-heavy recrystallization steps that are common in less optimized synthetic routes.
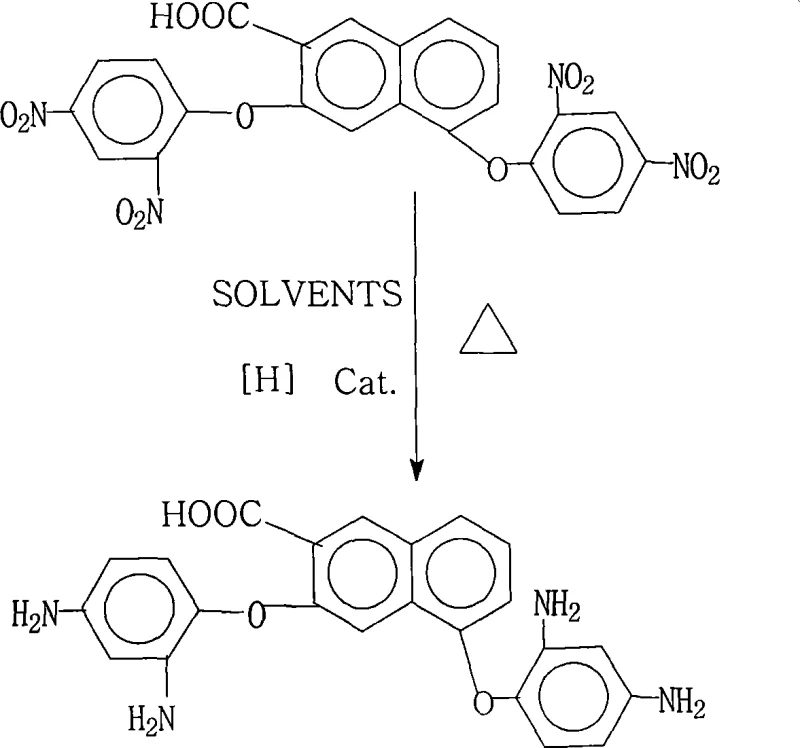
How to Synthesize 3,5-Bis(2,4-Diaminophenoxy)-2-Naphthoic Acid Efficiently
Implementing this synthesis route in a pilot or production setting requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and safety. The process begins with the careful charging of the reactor with the dinitro precursor, the Pd/C catalyst, and the selected alcohol solvent system, followed by the controlled addition of hydrazine hydrate to manage exothermic potential. Maintaining the temperature within the specified 70°C to 85°C window is critical; temperatures below this range may result in incomplete reduction, while excessive heat could lead to solvent degradation or safety hazards. The reaction progress should be monitored to confirm the disappearance of the starting material, typically requiring between 1 to 5 hours depending on the specific catalyst loading and solvent mix employed. Once the reaction is complete, the immediate hot filtration step is non-negotiable to preserve product quality, followed by a controlled cooling and precipitation phase to maximize yield recovery. For a comprehensive breakdown of the exact reagent quantities, specific solvent ratios, and detailed operational safety protocols, please refer to the standardized synthesis guide provided below.
- Combine 3,5-bis(2,4-dinitrophenoxy)-2-naphthoic acid with Pd/C catalyst (1-15% Pd) and saturated fatty alcohol solvents in a reaction vessel.
- Add hydrazine hydrate solution (60-85% concentration) and maintain the reaction temperature between 70°C and 85°C for 1 to 5 hours with continuous stirring.
- Filter the mixture while hot to remove the catalyst, cool the mother liquor, add pure water to precipitate crystals, then filter and vacuum dry to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthesis method translates into tangible strategic benefits that extend far beyond simple unit cost metrics. The elimination of high-pressure hydrogenation equipment significantly reduces the capital intensity required for production facilities, allowing for more flexible manufacturing setups that can be scaled up or down based on market demand without massive infrastructure investments. Furthermore, the simplicity of the workup procedure, which relies on basic filtration and water precipitation rather than complex chromatographic separations, drastically shortens the overall production cycle time. This acceleration in throughput enhances supply chain responsiveness, enabling suppliers to reduce lead times for high-purity polyimide intermediates and better accommodate urgent orders from downstream electronics manufacturers. The robustness of the process also implies a lower risk of batch failures due to operator error or equipment malfunction, thereby ensuring a more consistent and reliable flow of materials to the production line. By minimizing the reliance on exotic reagents and specialized high-pressure vessels, the supply chain becomes more resilient to disruptions, securing a steady source of critical raw materials for the fabrication of advanced flexible copper clad laminates.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the substitution of expensive and hazardous high-pressure hydrogen gas with readily available hydrazine hydrate, which lowers both raw material costs and safety compliance expenditures. The heterogeneous nature of the Pd/C catalyst allows for its potential recovery and reuse, further diminishing the variable cost per kilogram of the final product. Moreover, the absence of a dedicated refining or purification stage post-reaction eliminates the consumption of additional solvents and energy associated with recrystallization or distillation, resulting in substantial operational savings. These cumulative efficiencies allow manufacturers to offer competitive pricing structures without compromising on the stringent quality standards required by the semiconductor and display industries. The reduction in waste treatment costs, due to the lack of heavy metal sludge and acidic effluents, also contributes to a leaner cost structure that aligns with modern green chemistry principles.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as saturated fatty alcohols and hydrazine hydrate ensures that the raw material supply base is broad and stable, reducing the risk of shortages that often plague specialty chemical markets. The operational simplicity of the process means that it can be easily transferred between different manufacturing sites or scaled from pilot plants to multi-ton reactors with minimal technical friction. This scalability is crucial for meeting the surging demand for high-performance polyimides in the expanding fields of 5G communications and flexible electronics. By adopting a process that is less sensitive to minor fluctuations in operating conditions, suppliers can guarantee higher on-time delivery rates and maintain buffer stocks more effectively. This reliability is a key differentiator for partners looking to secure long-term contracts for critical electronic chemical components in a volatile global market.
- Scalability and Environmental Compliance: From an environmental perspective, this synthesis route offers a significantly cleaner profile compared to traditional methods, generating minimal three wastes (waste water, waste gas, and solid residue). The use of alcohol solvents, which are generally easier to recover and recycle than chlorinated or aromatic solvents, further reduces the environmental footprint of the manufacturing process. This alignment with strict environmental regulations facilitates smoother permitting processes for new production lines and reduces the liability associated with waste disposal. The ability to scale this process to industrial levels without encountering the engineering bottlenecks typical of high-pressure systems makes it an attractive option for expanding production capacity. Consequently, this method supports sustainable growth strategies, allowing companies to increase output to meet market needs while adhering to increasingly rigorous global sustainability and carbon neutrality goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 3,5-bis(2,4-diaminophenoxy)-2-naphthoic acid, derived directly from the technical specifications of the patented process. These insights are intended to clarify the operational advantages and quality benchmarks associated with this specific synthetic route for stakeholders evaluating potential suppliers. Understanding these details is essential for making informed decisions about integrating this high-performance monomer into your polymer synthesis workflows. The answers reflect the proven capabilities of the technology to deliver consistent quality and operational efficiency in an industrial setting.
Q: What is the achievable purity of 3,5-bis(2,4-diaminophenoxy)-2-naphthoic acid using this method?
A: According to patent CN101245022A, the described process consistently yields products with a purity exceeding 99%, eliminating the need for complex subsequent refining or purification steps.
Q: Why is this diamine critical for Flexible Copper Clad Laminates (FCCL)?
A: This specific aromatic diamine serves as a key monomer for synthesizing highly branched polyimide resins, which provide the essential thermal stability, low dielectric constant, and mechanical strength required for advanced FCCL applications.
Q: What are the safety advantages of using hydrazine hydrate over high-pressure hydrogenation?
A: The use of hydrazine hydrate as a reducing agent in a liquid-phase system avoids the significant infrastructure costs and safety risks associated with high-pressure hydrogen gas reactors, simplifying the operational workflow.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,5-Bis(2,4-Diaminophenoxy)-2-Naphthoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature are faithfully reproduced at an industrial scale. We are committed to delivering 3,5-bis(2,4-diaminophenoxy)-2-naphthoic acid that meets stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our facility is designed to handle complex organic syntheses with a focus on safety, quality, and environmental stewardship, making us an ideal strategic partner for your electronic material supply needs. We understand the critical nature of your timelines and the importance of material consistency in high-tech applications.
We invite you to engage with our technical procurement team to discuss how we can tailor our production capabilities to your specific volume requirements and quality standards. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of how our optimized synthesis route can improve your overall project economics. We encourage potential partners to reach out for specific COA data and route feasibility assessments to verify our capability to support your R&D and manufacturing goals. Let us collaborate to accelerate the development of next-generation polyimide materials that will power the future of flexible electronics and advanced composites.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
