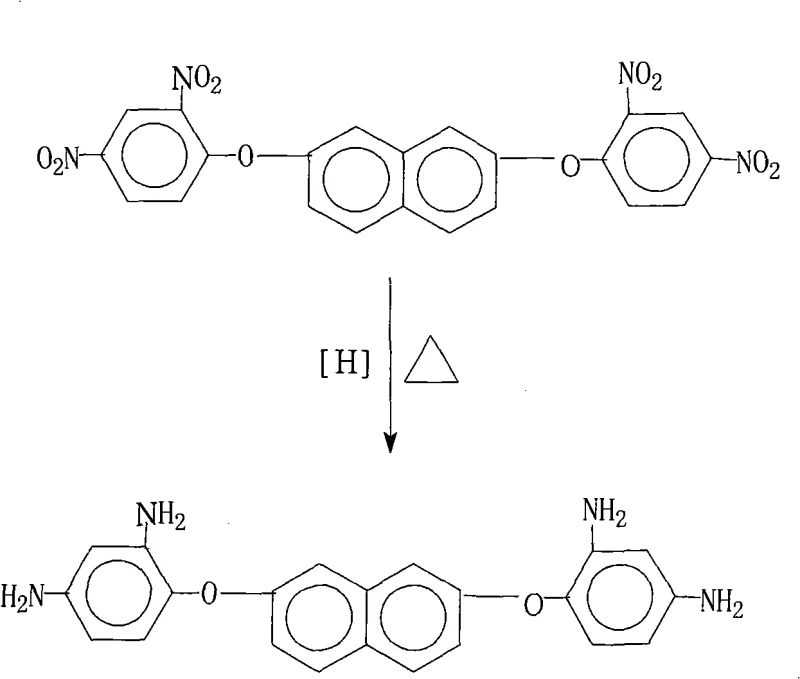
Scalable Synthesis of 2,7-Bis(2,4-Diaminophenoxy)Naphthalene for High-Performance Polyimide Applications
The rapid evolution of the flexible electronics sector has placed unprecedented demands on the thermal and mechanical properties of insulating materials, specifically driving the need for advanced polyimide precursors. Patent CN101265202A introduces a robust and highly efficient preparation method for 2,7-bis(2,4-diaminophenoxy)naphthalene, a critical monomer for synthesizing high-performance polyimide resins used in Flexible Copper Clad Laminates (FCCL). This technical breakthrough addresses the longstanding challenges of achieving high purity and yield in aromatic diamine synthesis without resorting to energy-intensive purification processes. By leveraging a catalytic transfer hydrogenation strategy using hydrazine hydrate and palladium on carbon, the process offers a streamlined pathway that is inherently safer and more cost-effective than traditional high-pressure hydrogenation methods. For R&D directors and procurement specialists in the electronic chemicals industry, this methodology represents a significant optimization in the supply chain for high-grade polymer additives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for aromatic diamines often rely on catalytic hydrogenation using molecular hydrogen under high pressure, which necessitates specialized, expensive reactor infrastructure and rigorous safety protocols to mitigate explosion risks. Furthermore, conventional reduction methods frequently suffer from incomplete conversion or the formation of azo and azoxy by-products, which can severely compromise the thermal stability and dielectric properties of the resulting polyimide films. The purification of these crude products typically requires multiple recrystallization steps or column chromatography, leading to substantial material loss and increased solvent waste, thereby inflating the overall manufacturing cost. Additionally, the use of harsh reducing agents like iron powder in acidic media generates massive amounts of solid waste sludge, creating significant environmental compliance burdens for modern chemical manufacturers striving for green production standards.
The Novel Approach
The novel approach detailed in the patent utilizes a catalytic transfer hydrogenation system where hydrazine hydrate serves as the hydrogen donor in the presence of a Pd/C catalyst within a saturated fatty alcohol solvent matrix. This method operates at atmospheric pressure and moderate temperatures ranging from 70°C to 85°C, drastically reducing the energy consumption and equipment complexity required for the reaction. The specific selection of solvents, such as mixtures of 2-methoxyethanol and ethylene glycol, plays a dual role by facilitating the reaction kinetics and enabling a unique crystallization-driven purification mechanism. By simply cooling the mother liquor and adding water, the target 2,7-bis(2,4-diaminophenoxy)naphthalene precipitates as high-purity crystals, effectively bypassing the need for downstream refining operations. This operational simplicity not only shortens the production cycle but also ensures a consistent impurity profile that is critical for downstream polymerization processes.
Mechanistic Insights into Pd/C-Catalyzed Transfer Hydrogenation
The core of this synthesis lies in the efficient transfer of hydrogen from hydrazine to the nitro groups of the naphthalene derivative, mediated by the palladium surface. In this catalytic cycle, hydrazine adsorbs onto the Pd/C active sites and decomposes to release nitrogen gas and reactive hydrogen species, which are immediately transferred to the electron-deficient nitro groups. This mechanism avoids the accumulation of free hydrogen gas, thereby minimizing the risk of over-reduction or hydrogenolysis of the ether linkages, which is a common side reaction in high-pressure hydrogenation of aryl ethers. The reaction proceeds through nitroso and hydroxylamine intermediates, which are rapidly reduced to the final amine functionality due to the high activity of the palladium catalyst in this specific solvent environment. The careful control of the hydrazine addition rate and temperature ensures that the exothermic nature of the decomposition is managed effectively, preventing thermal runaway and maintaining the structural integrity of the sensitive naphthalene backbone.
Impurity control is intrinsically built into the physicochemical properties of the chosen solvent system. The saturated fatty alcohols exhibit a temperature-dependent solubility profile where the starting dinitro compound and potential intermediate by-products remain soluble at elevated temperatures, while the final diamine product has significantly lower solubility upon cooling. This differential solubility allows for the selective crystallization of the target molecule, leaving trace impurities and catalyst residues in the mother liquor. The hot filtration step prior to cooling is crucial for removing the heterogeneous Pd/C catalyst, ensuring that no heavy metal contaminants carry over into the final product, which is essential for meeting the stringent electrical specifications of electronic grade materials. The result is a product with purity exceeding 99%, achieved through a single crystallization event rather than iterative purification cycles.
How to Synthesize 2,7-Bis(2,4-Diaminophenoxy)Naphthalene Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for scaling this reaction from laboratory to commercial production, emphasizing the critical balance between catalyst loading, solvent composition, and thermal management. To achieve optimal results, operators must strictly adhere to the specified molar ratios of hydrazine to substrate, ensuring an excess of the reducing agent to drive the reaction to completion while avoiding unnecessary waste. The choice of solvent mixture is equally vital, as it dictates the crystallization behavior and the ease of product isolation; therefore, precise control over the water addition rate during the precipitation phase is necessary to maximize crystal growth and filterability. Detailed standardized synthesis steps see the guide below.
- Charge the reactor with 2,7-bis(2,4-dinitrophenoxy)naphthalene, Pd/C catalyst (1-15% Pd loading), and a saturated fatty alcohol solvent system (e.g., 2-methoxyethanol/ethylene glycol mixture).
- Heat the mixture to 60°C and slowly add hydrazine hydrate solution (60-85% concentration) while maintaining strict temperature control.
- Maintain reaction temperature between 70°C and 85°C for 1 to 5 hours, then filter hot to remove catalyst, cool the mother liquor, and precipitate the product with water.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers transformative benefits in terms of cost structure and operational reliability. The elimination of high-pressure hydrogenation equipment significantly lowers the barrier to entry for production, allowing for manufacturing in facilities that do not possess specialized high-pressure infrastructure, thus expanding the potential supplier base. The simplified workflow, which integrates reaction and purification into a seamless continuous process, reduces labor costs and minimizes the turnaround time between batches, enhancing overall plant throughput. Furthermore, the use of commercially available and relatively inexpensive reagents like hydrazine hydrate and common fatty alcohols ensures a stable and resilient supply chain that is less susceptible to the volatility often seen with specialty gases or exotic catalysts.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by removing the need for expensive downstream purification steps such as column chromatography or multiple recrystallizations, which traditionally consume large volumes of solvents and energy. The ability to recover and potentially recycle the saturated alcohol solvents further contributes to a lower cost of goods sold (COGS), making the final polyimide monomer more price-competitive in the global market. Additionally, the high atom economy of the transfer hydrogenation reaction minimizes raw material waste, directly translating to improved margin potential for large-scale manufacturers.
- Enhanced Supply Chain Reliability: By relying on liquid-phase reagents that are easy to store and transport compared to compressed gases, the logistical complexity of the supply chain is drastically reduced. The robustness of the reaction conditions, which tolerate minor fluctuations in temperature and mixing without compromising product quality, ensures consistent batch-to-batch performance, a critical factor for long-term supply agreements with major electronics manufacturers. This reliability mitigates the risk of production delays caused by equipment failure or stringent safety shutdowns often associated with high-pressure hydrogen operations.
- Scalability and Environmental Compliance: The generation of nitrogen gas as the only by-product of the reducing agent makes this process environmentally benign, significantly simplifying waste treatment protocols and reducing the regulatory burden on the manufacturing site. The scalability is proven by the straightforward nature of the unit operations—mixing, heating, filtering, and crystallizing—which can be easily replicated in larger reactors without the engineering challenges of managing high-pressure gas-liquid interfaces. This alignment with green chemistry principles not only future-proofs the production facility against tightening environmental regulations but also enhances the brand value for customers seeking sustainable supply chain partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized aromatic diamine, derived directly from the patented methodology. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this monomer into their existing polyimide formulations or supply chains. The answers reflect the specific advantages of the hydrazine-based reduction route over legacy technologies.
Q: What is the primary advantage of using hydrazine hydrate over hydrogen gas for this reduction?
A: Using hydrazine hydrate with a Pd/C catalyst eliminates the need for high-pressure hydrogenation equipment, significantly reducing capital expenditure and safety risks associated with high-pressure gas handling in industrial settings.
Q: How is high purity (>99%) achieved without complex purification steps?
A: The process utilizes a specific saturated fatty alcohol solvent system that ensures high solubility of reactants during the reaction but allows for selective crystallization of the product upon cooling and water addition, effectively excluding impurities without recrystallization.
Q: Is this synthesis method suitable for large-scale polyimide production?
A: Yes, the method is explicitly designed for industrial scalability, featuring simple operational steps, readily available raw materials, and minimal waste generation, making it ideal for the mass production of polyimide precursors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,7-Bis(2,4-Diaminophenoxy)Naphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity monomers play in the performance of next-generation electronic materials, and we have positioned ourselves as a leader in the commercialization of such complex intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full industrial output is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 2,7-bis(2,4-diaminophenoxy)naphthalene meets the exacting standards required for FCCL and advanced composite applications.
We invite you to collaborate with us to optimize your material sourcing strategy and leverage our technical expertise for your specific formulation needs. Our team is prepared to provide a Customized Cost-Saving Analysis that demonstrates how switching to our optimized supply chain can reduce your total landed costs. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your volume requirements and quality targets.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
