Advanced Synthesis of 1,7-Bis(2,4-Diaminophenoxy)Naphthalene for High-Performance Polyimide Resins
Advanced Synthesis of 1,7-Bis(2,4-Diaminophenoxy)Naphthalene for High-Performance Polyimide Resins
The rapid evolution of the flexible electronics industry has placed unprecedented demands on the thermal and mechanical properties of substrate materials, driving the need for advanced aromatic diamine monomers. Patent CN101250126A introduces a robust and highly efficient preparation method for 1,7-bis(2,4-diaminophenoxy)naphthalene, a critical building block for next-generation polyimide resins used in Flexible Copper Clad Laminates (FCCL). This technological breakthrough leverages a catalytic transfer hydrogenation strategy using hydrazine hydrate and palladium on carbon (Pd/C) within a saturated fatty alcohol solvent system. By operating under mild thermal conditions ranging from 70°C to 85°C, this process achieves exceptional product purity exceeding 99% without the need for complex downstream purification steps. For R&D directors and procurement specialists seeking a reliable polyimide intermediate supplier, this patent represents a significant leap forward in process intensification, offering a pathway to reduce manufacturing costs while ensuring the stringent quality standards required for high-performance electronic applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of high-purity aromatic diamines like 1,7-bis(2,4-diaminophenoxy)naphthalene has relied heavily on catalytic hydrogenation using molecular hydrogen gas or stoichiometric metal reductions such as iron-acid processes. The conventional catalytic hydrogenation route typically necessitates specialized high-pressure reactors capable of withstanding significant internal pressures, which imposes a heavy capital expenditure burden on manufacturing facilities and introduces complex safety protocols for handling explosive hydrogen gas. Furthermore, these high-pressure systems often require rigorous maintenance schedules and specialized operator training, leading to increased operational overheads and potential production bottlenecks. Alternatively, classical metal reduction methods generate substantial quantities of heavy metal waste sludge, creating severe environmental compliance challenges and escalating waste disposal costs. These legacy technologies often struggle to consistently achieve the ultra-high purity levels demanded by the semiconductor and advanced composite industries, frequently requiring multiple recrystallization steps that erode overall yield and extend production lead times significantly.
The Novel Approach
In stark contrast to these legacy methodologies, the novel approach detailed in the patent utilizes a heterogeneous catalytic system driven by hydrazine hydrate as the hydrogen donor, effectively circumventing the need for high-pressure infrastructure. This transfer hydrogenation protocol operates at atmospheric pressure within a temperature window of 70°C to 85°C, utilizing readily available saturated fatty alcohols such as ethanol, methanol, or ethylene glycol as the reaction medium. The use of Pd/C catalysts with varying palladium loadings allows for precise control over the reaction kinetics, ensuring complete conversion of the dinitro precursor to the desired diamine while minimizing side reactions. A distinct advantage of this method is the simplicity of the workup procedure; the catalyst is easily removed via hot filtration, and the product is isolated through a straightforward water-induced crystallization from the mother liquor. This streamlined workflow not only drastically simplifies the operational complexity but also ensures that the final crystal product meets the rigorous purity specifications of over 99% directly from the reactor, eliminating the need for energy-intensive distillation or chromatographic purification columns.
Mechanistic Insights into Pd/C-Catalyzed Transfer Hydrogenation
The core of this synthetic innovation lies in the efficient mechanism of catalytic transfer hydrogenation, where hydrazine hydrate serves as a safe and effective source of nascent hydrogen on the surface of the palladium catalyst. In this mechanistic pathway, the hydrazine molecule adsorbs onto the active sites of the Pd/C surface, where it undergoes dehydrogenation to release nitrogen gas and reactive hydrogen species. These activated hydrogen atoms are then transferred directly to the nitro groups of the 1,7-bis(2,4-dinitrophenoxy)naphthalene substrate, sequentially reducing them through nitroso and hydroxylamine intermediates to the final primary amine functionality. The choice of saturated fatty alcohol solvents is critical, as they provide excellent solubility for both the organic substrate and the hydrazine reagent while stabilizing the transition states involved in the reduction. This mechanism is highly selective for nitro group reduction, leaving the sensitive ether linkages and the naphthalene aromatic core intact, which is paramount for preserving the structural integrity required for high-performance polymer applications.
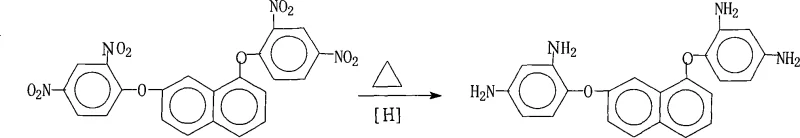
From an impurity control perspective, this mechanism offers superior advantages over traditional reduction methods by minimizing the formation of azo or azoxy byproducts that often plague nitro reductions. The controlled addition of hydrazine hydrate, combined with the specific temperature range of 70°C to 85°C, ensures that the reaction proceeds smoothly without localized overheating that could trigger decomposition or polymerization side reactions. The heterogeneous nature of the Pd/C catalyst facilitates easy separation, preventing metal contamination in the final product, which is a critical quality attribute for electronic grade chemicals. Furthermore, the crystallization step induced by the addition of pure water exploits the differential solubility of the diamine product in the alcohol-water mixture, effectively excluding soluble organic impurities and residual starting materials from the crystal lattice. This intrinsic purification capability embedded within the reaction design ensures that the resulting 1,7-bis(2,4-diaminophenoxy)naphthalene possesses the high purity and consistent quality necessary for synthesizing polyimides with low dielectric constants and excellent thermomechanical stability.
How to Synthesize 1,7-Bis(2,4-Diaminophenoxy)Naphthalene Efficiently
Implementing this synthesis route requires careful attention to reagent ratios and thermal management to maximize yield and safety. The process begins by charging a reaction vessel with the dinitro precursor, the Pd/C catalyst, and the selected alcohol solvent system, followed by heating to initiate the reaction. The controlled dropwise addition of hydrazine hydrate is the key operational parameter, as it regulates the rate of hydrogen generation and prevents exothermic runaway. Once the addition is complete, the mixture is maintained at the target temperature to ensure full conversion, monitored typically by HPLC or TLC. Upon completion, the hot filtration step removes the spent catalyst, and the subsequent cooling and water addition induce crystallization of the high-purity diamine. For a detailed breakdown of the specific reagent quantities, solvent choices, and step-by-step operational parameters validated by experimental data, please refer to the standardized synthesis guide below.
- Charge the reactor with 1,7-bis(2,4-dinitrophenoxy)naphthalene, Pd/C catalyst, and saturated fatty alcohol solvent, then heat to 60°C.
- Dropwise add hydrazine hydrate solution (60%-85% concentration) and maintain reaction temperature between 70°C and 85°C for 1 to 5 hours.
- Filter hot to remove catalyst, cool the mother liquor, add pure water to precipitate crystals, filter, and vacuum dry to obtain >99% purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this hydrazine-based reduction technology translates into tangible strategic advantages regarding cost structure and operational reliability. The elimination of high-pressure hydrogenation equipment significantly lowers the barrier to entry for manufacturing this high-value intermediate, allowing for more flexible production scheduling and reduced capital depreciation costs. The simplicity of the workup procedure, which relies on basic filtration and crystallization rather than complex distillation trains, reduces energy consumption and minimizes the requirement for specialized solvent recovery systems. This streamlined process flow enhances overall plant throughput, enabling manufacturers to respond more agilely to market demand fluctuations without compromising on product quality or delivery timelines. Furthermore, the use of commodity chemicals like hydrazine hydrate and common alcohols ensures a stable and resilient supply chain, mitigating the risks associated with sourcing exotic or highly regulated reagents.
- Cost Reduction in Manufacturing: The transition from high-pressure catalytic hydrogenation to atmospheric transfer hydrogenation results in substantial cost savings by removing the need for expensive pressure-rated reactors and associated safety infrastructure. The process inherently reduces energy consumption due to lower operating temperatures and the elimination of energy-intensive purification steps like column chromatography or multiple recrystallizations. Additionally, the high selectivity of the Pd/C catalyst minimizes raw material waste, ensuring that the expensive dinitro precursor is converted efficiently into the desired diamine product with minimal loss to byproducts. These factors collectively drive down the cost of goods sold (COGS), making the final polyimide resin more competitive in the price-sensitive electronics market.
- Enhanced Supply Chain Reliability: By utilizing widely available solvents such as ethanol, methanol, and ethylene glycol, the process decouples production from the volatility of niche chemical markets. The robustness of the reaction conditions allows for consistent batch-to-batch reproducibility, which is critical for maintaining long-term supply contracts with major polymer manufacturers. The simplified operational protocol also reduces the dependency on highly specialized labor, allowing for easier scaling of production capacity across different manufacturing sites. This flexibility ensures a continuous supply of high-purity 1,7-bis(2,4-diaminophenoxy)naphthalene, safeguarding downstream customers against production disruptions and inventory shortages.
- Scalability and Environmental Compliance: The generation of nitrogen gas as the only gaseous byproduct simplifies off-gas treatment compared to processes that emit volatile organic compounds or acidic fumes. The absence of heavy metal sludge, typical of iron-acid reductions, significantly lowers hazardous waste disposal costs and aligns with increasingly stringent global environmental regulations. The process is inherently scalable from pilot plant to commercial tonnage scales without requiring fundamental changes to the reaction engineering, facilitating rapid capacity expansion. This environmental and operational efficiency positions the manufacturer as a sustainable partner, appealing to multinational corporations with strict carbon footprint and waste reduction mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 1,7-bis(2,4-diaminophenoxy)naphthalene. These insights are derived directly from the patented methodology and practical experience in scaling aromatic amine synthesis. Understanding these nuances is essential for evaluating the feasibility of integrating this intermediate into your specific polymer formulation or supply chain strategy. The answers provided reflect the consensus on best practices for handling, storage, and quality assurance of this sensitive chemical entity.
Q: What are the critical purity specifications for FCCL grade diamines?
A: For Flexible Copper Clad Laminate (FCCL) applications, the 1,7-bis(2,4-diaminophenoxy)naphthalene must exceed 99% purity to ensure optimal dielectric properties and thermal stability in the final polyimide resin.
Q: How does the hydrazine reduction method compare to catalytic hydrogenation?
A: The hydrazine reduction method described in CN101250126A eliminates the need for high-pressure hydrogenation equipment, significantly reducing capital expenditure and safety risks while maintaining high yields and selectivity.
Q: Is the Pd/C catalyst recoverable in this process?
A: Yes, the heterogeneous Pd/C catalyst is removed via hot filtration before crystallization, allowing for potential recovery and recycling, which further enhances the economic viability of the process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,7-Bis(2,4-Diaminophenoxy)Naphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced electronic materials hinges on the availability of ultra-high purity intermediates produced via robust and scalable pathways. Our technical team has extensively analyzed the hydrazine reduction route described in CN101250126A and possesses the expertise to optimize this chemistry for large-scale manufacturing. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that every batch meets stringent purity specifications required for FCCL and aerospace applications. Our rigorous QC labs employ state-of-the-art analytical instrumentation to verify identity, assay, and impurity profiles, guaranteeing that our 1,7-bis(2,4-diaminophenoxy)naphthalene consistently exceeds the 99% purity benchmark essential for high-performance polyimide synthesis.
We invite you to collaborate with us to leverage these technological advancements for your supply chain optimization. Our team is prepared to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments that demonstrate how our optimized manufacturing processes can enhance your product performance while reducing total landed costs. Let us be your strategic partner in navigating the complexities of the electronic chemical supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
