Advanced Synthesis of Trichlorohydrazinopyridine Hydrate for Commercial Scale-up
The pharmaceutical and agrochemical industries are constantly seeking robust supply chains for critical heterocyclic intermediates that serve as the backbone for complex active ingredients. Patent CN102093288B introduces a highly efficient preparation method for trichlorohydrazinopyridine hydrate, a pivotal precursor in the synthesis of advanced antibacterial and antiviral agents. This technology addresses long-standing challenges in the production of chlorinated pyridine derivatives by utilizing a direct hydrazinolysis approach on 2,3,5,6-tetrachloropyridine. By optimizing solvent systems and reaction parameters, this method achieves exceptional yields and purity levels that are essential for downstream pharmaceutical applications. For R&D directors and procurement specialists, understanding this pathway offers a strategic advantage in securing reliable sources for high-quality pyridine intermediates. The process not only simplifies the synthetic route but also aligns with modern green chemistry principles by minimizing waste and maximizing raw material utilization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the production of chlorinated pyridine derivatives has relied heavily on the direct chlorination of pyridine or its derivatives, a process fraught with significant technical and environmental hurdles. These conventional routes often suffer from poor regioselectivity, resulting in complex mixtures of isomers such as 2-chloropyridine, 2,6-dichloropyridine, and various trichloro-isomers that are difficult and costly to separate. Furthermore, the use of elemental chlorine gas poses severe safety risks and generates substantial amounts of hazardous waste, complicating compliance with increasingly stringent environmental regulations. The low yields associated with these multi-step chlorination processes inevitably drive up the cost of goods sold, making the final intermediates less competitive in the global market. Additionally, the presence of trace impurities from harsh chlorination conditions can negatively impact the efficacy and safety profiles of the final drug substances, necessitating expensive and time-consuming purification steps.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a nucleophilic substitution strategy that bypasses the complexities of direct chlorination entirely. By reacting 2,3,5,6-tetrachloropyridine with hydrazine hydrate in a controlled alcoholic environment, the process achieves high selectivity for the target trichlorohydrazinopyridine hydrate. This method operates under normal pressure and moderate reflux temperatures, significantly reducing energy consumption and equipment stress compared to high-pressure chlorination reactors. The ability to use common alcohols like ethanol as solvents not only lowers material costs but also facilitates easier solvent recovery and recycling, creating a more sustainable manufacturing loop. This streamlined approach eliminates the need for extensive isomer separation, delivering a crude product with purity levels exceeding 98% HPLC content, which is ready for immediate use in subsequent synthetic transformations.
Mechanistic Insights into Hydrazinolysis of Tetrachloropyridine
The core of this synthesis lies in the nucleophilic aromatic substitution mechanism, where the hydrazine molecule acts as a potent nucleophile attacking the electron-deficient pyridine ring. The presence of four electronegative chlorine atoms on the pyridine ring significantly activates the structure towards nucleophilic attack, particularly at the 6-position which is sterically accessible and electronically favorable. The reaction proceeds through a Meisenheimer-like complex intermediate, where the addition of the hydrazine is followed by the elimination of a chloride ion, driven by the restoration of aromaticity. The choice of solvent plays a critical role in stabilizing the transition state and solubilizing both the organic substrate and the aqueous hydrazine component, ensuring a homogeneous reaction environment that maximizes collision frequency. Understanding this mechanistic pathway allows chemists to fine-tune reaction conditions, such as stirring speed and temperature, to prevent side reactions and ensure complete conversion of the starting material.
Impurity control in this process is inherently managed by the specificity of the hydrazinolysis reaction and the physical properties of the product. Unlike radical chlorination which generates a statistical distribution of isomers, this substitution is highly directed, minimizing the formation of structural analogs that are difficult to remove. The patent highlights that the crude product does not require recrystallization or chromatographic purification to achieve high purity, indicating that by-products are either minimal or easily separated during the filtration and washing steps. The hydration state of the product also contributes to its stability and crystallinity, allowing for efficient isolation via simple cooling and filtration. This inherent purity reduces the burden on quality control laboratories and ensures that the intermediate meets the rigorous specifications required for GMP manufacturing of active pharmaceutical ingredients.
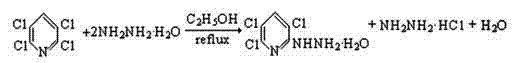
How to Synthesize Trichlorohydrazinopyridine Hydrate Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and process control to replicate the high yields reported in the patent data. The procedure involves charging a reactor with precise molar ratios of 2,3,5,6-tetrachloropyridine and hydrazine hydrate solution, typically ranging from 1:2.5 to 1:4, to ensure complete consumption of the valuable chlorinated substrate. The reaction is conducted under reflux in a C1-C4 alcohol, with ethanol being the preferred solvent due to its optimal boiling point and solvation properties. Maintaining an appropriate stirring speed is vital to ensure heat transfer and mixing without causing the reaction mass to overflow into the condenser system. After the reaction period of 1 to 3 hours, the mixture is cooled to induce crystallization, followed by filtration and drying to isolate the final white solid product.
- Charge a reactor with 2,3,5,6-tetrachloropyridine and hydrazine hydrate solution (80% concentration) in an alcoholic solvent such as ethanol.
- Stir the reaction mixture under reflux conditions at normal pressure for 1 to 3 hours, ensuring adequate stirring speed to prevent overflow while maintaining complete reaction.
- Cool the reaction mass to room temperature, filter the precipitate, wash with solvent, and dry to obtain the high-purity trichlorohydrazinopyridine hydrate product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthesis method presents compelling economic and operational benefits that directly impact the bottom line. The high yield of the process, consistently reaching above 96% in optimized embodiments, translates to a significant reduction in the cost per kilogram of the intermediate, allowing for more competitive pricing strategies in the final drug market. Furthermore, the ability to recycle the alcoholic solvent and recover hydrazine hydrochloride from the aqueous waste stream drastically reduces raw material consumption and waste disposal costs. This closed-loop solvent system not only enhances sustainability metrics but also mitigates the risk of supply disruptions associated with volatile solvent markets. The simplicity of the work-up procedure, which avoids complex purification columns or extensive washing sequences, shortens the overall production cycle time and increases facility throughput.
- Cost Reduction in Manufacturing: The elimination of expensive purification steps and the high efficiency of raw material conversion lead to substantial cost savings in the manufacturing process. By avoiding the need for additional reagents to remove isomers or by-products, the overall material cost is significantly lowered. The recovery and reuse of ethanol solvent further decrease the variable costs associated with each production batch, enhancing the overall margin profile. Additionally, the reduced energy demand from operating at normal pressure and moderate temperatures contributes to lower utility expenses over the long term.
- Enhanced Supply Chain Reliability: The reliance on readily available commodity chemicals such as ethanol and hydrazine hydrate ensures a stable and resilient supply chain that is less susceptible to geopolitical or logistical disruptions. The robustness of the reaction conditions means that the process can be easily transferred between different manufacturing sites without significant re-validation efforts. High crude purity reduces the dependency on specialized purification services, allowing for more flexible sourcing options and faster turnaround times for order fulfillment. This reliability is crucial for maintaining continuous production schedules for downstream API manufacturers who depend on timely delivery of key intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from laboratory scale to potential industrial tonnage without fundamental changes to the chemistry. The reduction in hazardous waste generation, particularly the avoidance of chlorine gas and the minimization of aqueous effluent through recycling, simplifies environmental permitting and compliance management. The solid nature of the product facilitates safe handling, storage, and transportation, reducing the risks associated with liquid intermediates. These factors combined make the technology an attractive option for companies looking to expand their capacity while adhering to strict environmental, social, and governance (ESG) goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of trichlorohydrazinopyridine hydrate based on the patented technology. These insights are derived directly from the experimental data and beneficial effects described in the intellectual property documentation. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their supply chains. The answers reflect the practical realities of scaling this chemistry for commercial purposes while maintaining high quality standards.
Q: What is the expected yield and purity of this synthesis method?
A: According to patent CN102093288B, when using ethanol as the solvent, the yield can generally reach above 96%, and the HPLC purity of the crude product can exceed 98% without further purification.
Q: Can the solvent be recycled in this process?
A: Yes, the process allows for solvent recycling. The filtrate can be separated into an alcohol layer for direct reuse in the next batch and a water layer for hydrazine hydrochloride recovery, significantly reducing waste.
Q: What are the downstream applications of this intermediate?
A: This intermediate is crucial for producing 2,5-dichloropyridine and 2,3,5-trichloropyridine, which are key precursors for synthetic antibacterial agents, sterilants, and antiviral drugs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trichlorohydrazinopyridine Hydrate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your pharmaceutical development programs. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of trichlorohydrazinopyridine hydrate meets the highest industry standards. We are committed to providing a seamless supply chain experience, leveraging our technical expertise to optimize processes for cost and efficiency while maintaining full regulatory compliance.
We invite you to collaborate with us to explore how this advanced synthesis route can enhance your product portfolio and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality expectations. Please contact us to request specific COA data and route feasibility assessments that will demonstrate the tangible benefits of partnering with NINGBO INNO PHARMCHEM. Let us help you accelerate your timeline to market with a supply partner you can trust for consistency and excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
