Industrial Scale Synthesis of High-Purity Polyimide Precursors via Optimized SnAr Chemistry
Industrial Scale Synthesis of High-Purity Polyimide Precursors via Optimized SnAr Chemistry
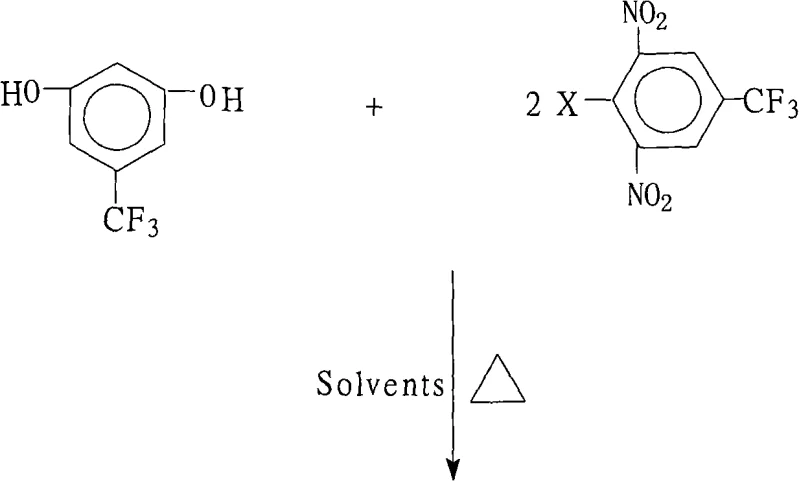
The rapid advancement of the optoelectronic industry, particularly in the sector of liquid crystal displays (LCDs) and flexible electronics, has created an insatiable demand for high-performance aromatic polyimides that possess superior thermal stability and mechanical strength. At the heart of manufacturing these advanced polymers lies the critical need for highly branched monomers, specifically 3,5-bis(2,6-dinitro-4-trifluoromethylphenoxy)benzotrifluoride, which serves as a pivotal precursor for generating aromatic polyvalent primary amines. Patent CN101270054A introduces a groundbreaking preparation method that addresses the historical lack of efficient synthetic routes for this complex fluorinated ether, offering a robust solution that combines high yield with exceptional purity. This technical insight report analyzes the proprietary chemistry disclosed in the patent, highlighting how this nucleophilic aromatic substitution strategy overcomes traditional synthetic bottlenecks to deliver a reliable supply chain for electronic chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovation detailed in CN101270054A, the synthesis of highly branched fluorinated ether intermediates was plagued by significant technical hurdles that hindered widespread industrial adoption. Conventional etherification processes often struggled with incomplete conversions due to the steric hindrance imposed by the ortho-nitro groups and the electron-withdrawing trifluoromethyl substituents on the aromatic rings. Traditional methods frequently relied on harsh reaction conditions or expensive phase-transfer catalysts that were difficult to remove, leading to product contamination with heavy metals or residual salts that are unacceptable for electronic-grade applications. Furthermore, many existing protocols failed to effectively manage the water byproduct generated during the condensation reaction, causing the equilibrium to shift backward and resulting in mediocre yields and a complex mixture of mono-substituted impurities that were arduous to separate. These inefficiencies not only inflated the cost of goods sold but also created substantial environmental burdens due to the generation of hazardous waste streams that required complex treatment before disposal.
The Novel Approach
The methodology presented in the patent revolutionizes this landscape by employing a meticulously optimized nucleophilic aromatic substitution (SnAr) reaction that leverages the activating power of the nitro groups while strictly controlling the reaction environment. By utilizing a specific molar ratio of 3,5-dihydroxybenzotrifluoride to 2,6-dinitro-4-trifluoromethyl halobenzene ranging from 1.0:2.0 to 1.0:2.2, the process ensures complete double substitution while minimizing the formation of unwanted byproducts. A key innovation lies in the use of a binary solvent system comprising a strong polar aprotic solvent and a water-insoluble organic solvent, which facilitates an azeotropic distillation process to continuously remove water from the reaction zone. This continuous water removal drives the reaction equilibrium decisively towards the product side, enabling high conversion rates without the need for excessive reagent loading. The result is a streamlined, one-pot synthesis that operates under atmospheric pressure, significantly simplifying the engineering requirements for scale-up.
Mechanistic Insights into SnAr Etherification
The core of this synthesis relies on a classic yet finely tuned nucleophilic aromatic substitution mechanism where the phenoxide anion acts as the nucleophile attacking the electron-deficient aromatic ring of the halobenzene. The presence of two strong electron-withdrawing nitro groups at the ortho positions relative to the leaving halogen atom dramatically activates the ring towards nucleophilic attack by stabilizing the negatively charged Meisenheimer complex intermediate. Simultaneously, the trifluoromethyl group at the para position further enhances this electrophilicity through its powerful inductive effect, lowering the activation energy required for the substitution to proceed. The choice of base, ranging from alkali metal carbonates to hydroxides, is critical for generating the reactive phenoxide species in situ without causing hydrolysis of the sensitive nitro groups or the trifluoromethyl moieties. This delicate balance ensures that the reaction proceeds selectively at the carbon-halogen bond, preserving the integrity of the rest of the molecular architecture which is essential for the subsequent polymerization steps.
Controlling the impurity profile is paramount for applications in high-end polyimides, where even trace amounts of mono-substituted intermediates can disrupt the polymer chain growth and degrade the thermal properties of the final film. The patented process mitigates this risk through the precise control of reaction temperature between 80°C and 180°C and the extended reflux period of 6 to 18 hours, which allows sufficient time for the second substitution event to occur after the first ether linkage is formed. The use of a mixed solvent system not only aids in water removal but also ensures that the intermediate mono-ether remains soluble and reactive, preventing premature precipitation that could halt the reaction. Post-reaction workup involving concentration and water precipitation effectively separates the highly lipophilic product from inorganic salts and polar byproducts, yielding crystals with purity levels consistently above 99%, thereby eliminating the need for costly and yield-reducing recrystallization steps.
How to Synthesize 3,5-Bis(2,6-Dinitro-4-Trifluoromethylphenoxy)Benzotrifluoride Efficiently
Implementing this synthesis route requires strict adherence to the stoichiometric ratios and solvent compositions defined in the patent to ensure reproducibility and safety. The process begins with the charging of the reactor with the dihydroxy starting material, the halobenzene electrophile, and the selected base system, followed by the addition of the binary solvent mixture. Operators must monitor the water separation carefully, as the efficiency of the azeotropic distillation directly correlates with the final yield of the reaction. Once the reaction is deemed complete via HPLC or TLC monitoring, the workup procedure involves concentrating the mixture to recover valuable solvents, cooling the residue, and inducing crystallization through the addition of water. For a detailed breakdown of the specific operational parameters, reagent grades, and safety precautions required for this synthesis, please refer to the standardized protocol below.
- Charge the reactor with 3,5-dihydroxybenzotrifluoride, 2,6-dinitro-4-trifluoromethyl halobenzene, and a mixed base system in a solvent mixture of polar aprotic and water-insoluble organic solvents.
- Heat the reaction mixture to reflux (80°C to 180°C) while continuously removing water via azeotropic distillation for 6 to 18 hours to drive the equilibrium forward.
- Concentrate the reaction solution, cool the system, add water to precipitate the solid product, then filter, wash with hot water, and dry to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers transformative benefits that extend far beyond simple chemical yield improvements. The process is designed with inherent cost-saving mechanisms that reduce the total cost of ownership for this critical intermediate, making it a strategically sound choice for long-term sourcing agreements. By eliminating the need for exotic catalysts or high-pressure reactors, the capital expenditure required for manufacturing is significantly lowered, while the operational simplicity reduces the risk of batch failures. Furthermore, the ability to recycle solvents repeatedly creates a closed-loop system that minimizes raw material consumption and aligns with modern sustainability goals, providing a competitive edge in markets where environmental compliance is increasingly scrutinized by downstream customers.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of readily available and inexpensive starting materials such as 3,5-dihydroxybenzotrifluoride and common halogenated nitrobenzenes, which are produced on a massive global scale. The elimination of expensive transition metal catalysts removes the necessity for costly purification steps to meet low-metal specifications, which is a frequent requirement in the electronics industry. Additionally, the high atom economy of the substitution reaction combined with the efficient recovery and reuse of the organic solvent system drastically reduces the variable costs associated with waste disposal and fresh solvent procurement. These factors collectively contribute to a substantially lower cost basis per kilogram of finished product, allowing for more aggressive pricing strategies or improved margin retention.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the robustness of the reaction conditions, which do not rely on scarce or geographically constrained reagents. The use of standard alkali bases and common organic solvents ensures that production can be maintained even during fluctuations in the availability of specialty chemicals. The atmospheric pressure operation reduces the maintenance burden on reactor vessels and associated piping, leading to higher equipment uptime and fewer unplanned shutdowns. This reliability translates into shorter lead times for order fulfillment and a more predictable delivery schedule, which is critical for manufacturers of polyimides who operate on tight production timelines to meet the demands of the consumer electronics sector.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively from laboratory bench scales to multi-kilogram pilot batches without loss of efficiency. The "three-waste" output is minimized due to the high conversion rates and solvent recycling, simplifying the wastewater treatment requirements and reducing the environmental footprint of the manufacturing facility. The absence of corrosive byproducts means that standard stainless steel equipment can be used, avoiding the need for lined reactors or Hastelloy, which further lowers the barrier to entry for contract manufacturing organizations. This combination of scalability and environmental friendliness makes the technology future-proof against tightening regulatory standards regarding industrial emissions and chemical handling.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this intermediate into their supply chain, we have compiled answers to the most common inquiries regarding the patent technology. These responses address critical concerns about purity specifications, process safety, and the adaptability of the method to different production volumes. Understanding these technical nuances is essential for making informed decisions about vendor qualification and process validation.
Q: What represents the primary advantage of this synthesis method regarding product purity?
A: The patented process achieves exceptional purity levels exceeding 99% by utilizing a specific solvent system and water-removal technique that minimizes side reactions and hydrolysis, ensuring the final polyimide monomer meets stringent electronic grade specifications.
Q: How does the solvent recovery system impact overall production costs?
A: The method employs a binary solvent system where the organic components can be easily separated and recycled repeatedly without significant degradation, drastically reducing raw material consumption and waste disposal costs compared to single-use solvent protocols.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the reaction operates under atmospheric pressure without requiring specialized high-pressure equipment or corrosive reagents, making it highly scalable and safe for commercial production facilities ranging from pilot plants to multi-ton annual capacity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,5-Bis(2,6-Dinitro-4-Trifluoromethylphenoxy)Benzotrifluoride Supplier
As the global demand for high-performance polyimides continues to surge, securing a stable source of high-quality monomers like 3,5-bis(2,6-dinitro-4-trifluoromethylphenoxy)benzotrifluoride is paramount for maintaining product leadership. NINGBO INNO PHARMCHEM stands ready to support your development and production needs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch meets the exacting standards required for electronic material applications. We understand the critical nature of this intermediate in the value chain and are committed to delivering consistent quality that enables your downstream polymerization processes to run smoothly and efficiently.
We invite you to engage with our technical procurement team to discuss how we can tailor our manufacturing capabilities to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized process can reduce your overall material costs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to validate our capabilities against your internal standards with confidence and speed.
