Industrial Synthesis of 3,5-Bis(2,4-Diaminophenoxy)Toluene for High-Performance Polyimide Resins
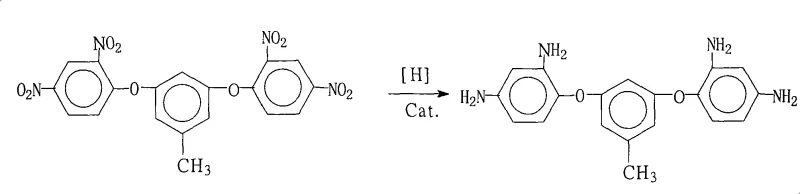
The escalating demand for high-performance flexible copper clad laminates (FCCL) and advanced polyimide composites in the aerospace and microelectronics sectors has necessitated the development of robust synthetic routes for specialized aromatic diamines. Patent CN101260060A introduces a pivotal advancement in the preparation of 3,5-bis(2,4-diaminophenoxy)toluene, a critical monomer for synthesizing highly branched polyimide resins with superior thermal stability and dielectric properties. This technical disclosure outlines a catalytic transfer hydrogenation protocol that effectively converts the corresponding dinitro precursor into the target diamine with exceptional efficiency. By leveraging a palladium on carbon (Pd/C) catalyst system in conjunction with hydrazine hydrate, the process circumvents the safety hazards and infrastructure costs associated with high-pressure gaseous hydrogenation. For R&D directors and procurement specialists seeking a reliable polyimide monomer supplier, understanding the nuances of this patented methodology is essential for securing a supply chain capable of delivering high-purity electronic chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methodologies for reducing aromatic nitro compounds to amines often rely on stoichiometric metal reductions using iron or zinc in acidic media, or catalytic hydrogenation using molecular hydrogen gas. The metal-acid reduction pathways generate substantial quantities of toxic metal sludge and acidic wastewater, creating severe environmental compliance burdens and escalating waste treatment costs for manufacturers. Furthermore, these methods frequently suffer from poor selectivity, leading to the formation of azo and hydrazo byproducts that are difficult to separate from the desired diamine, thereby compromising the optical and electrical performance of the final polyimide material. On the other hand, conventional catalytic hydrogenation, while cleaner, typically requires specialized high-pressure reactors and rigorous safety protocols to handle explosive hydrogen gas, representing a significant capital expenditure barrier for many fine chemical producers. These legacy processes often necessitate complex downstream purification steps, such as column chromatography or multiple recrystallizations, which drastically reduce overall yield and extend production lead times.
The Novel Approach
The innovative strategy detailed in the patent data employs a transfer hydrogenation mechanism using hydrazine hydrate as the hydrogen donor in the presence of a heterogeneous Pd/C catalyst. This approach operates under atmospheric pressure and moderate temperatures ranging from 70°C to 85°C, significantly lowering the energy consumption and equipment requirements compared to high-pressure hydrogenation. The use of saturated fatty alcohols or glycols as solvents provides an optimal medium where the starting dinitro material is soluble at reaction temperatures but allows the product diamine to crystallize out upon the addition of water during the workup phase. This "reaction-crystallization" integration eliminates the need for solvent swapping or extensive distillation, streamlining the isolation process. By avoiding harsh acidic conditions and heavy metal reductants, this novel route ensures a cleaner impurity profile, directly addressing the stringent quality standards required for electronic grade materials used in flexible circuit boards and high-temperature composites.
Mechanistic Insights into Pd/C-Catalyzed Transfer Hydrogenation
The core of this synthesis lies in the efficient activation of hydrazine on the palladium surface, which facilitates the stepwise reduction of the nitro groups (-NO2) to amino groups (-NH2). In this catalytic cycle, hydrazine adsorbs onto the active sites of the Pd/C catalyst and decomposes to generate reactive nitrogen species and atomic hydrogen equivalents in situ. These active hydrogen species are then transferred to the nitro groups of the 3,5-bis(2,4-dinitrophenoxy)toluene substrate, progressing through nitroso and hydroxylamine intermediates before finally yielding the stable amine. The choice of Pd/C is critical, as palladium exhibits superior activity for nitro reduction compared to other transition metals, while the carbon support ensures high dispersion and stability of the metal particles. Crucially, the reaction conditions are tuned to prevent the hydrogenolysis of the aryl-oxygen-aryl ether linkages, which are susceptible to cleavage under more vigorous reducing conditions, thus preserving the structural integrity of the monomer backbone.
Impurity control is inherently managed by the specificity of the catalytic system and the solubility characteristics of the reaction mixture. The mild temperature window of 70°C to 85°C minimizes thermal degradation and side reactions such as condensation between amino and nitro groups, which could otherwise form insoluble azoxy polymers. Furthermore, the hot filtration step effectively removes the solid catalyst and any insoluble particulate matter before the product crystallization begins, acting as a primary purification barrier. As the mother liquor cools and water is introduced, the polarity of the solvent system shifts, causing the highly polar diamine product to precipitate while non-polar organic impurities and residual hydrazine byproducts remain in the solution. This thermodynamic driving force ensures that the isolated crystals possess a purity exceeding 99%, as evidenced by the experimental data where purities of 99.5% to 99.8% were consistently achieved across multiple embodiments without further refinement.
How to Synthesize 3,5-Bis(2,4-Diaminophenoxy)Toluene Efficiently
Implementing this synthesis route requires precise control over reagent stoichiometry and thermal profiles to maximize yield and safety. The process begins with the suspension of the dinitro precursor in a selected alcohol or glycol solvent system, followed by the addition of the Pd/C catalyst. The molar ratio of hydrazine hydrate to the substrate is maintained between 1:20 and 1:50 to ensure complete conversion while minimizing excess reagent waste. Detailed operational parameters, including specific catalyst loadings and solvent combinations that optimize the balance between reaction rate and product isolation, are critical for successful technology transfer. The following guide summarizes the standardized operational sequence derived from the patent examples to assist process engineers in replicating this high-efficiency pathway.
- Charge the reactor with 3,5-bis(2,4-dinitrophenoxy)toluene, Pd/C catalyst (1-15% Pd loading), and a saturated fatty alcohol solvent system.
- Heat the mixture to 60°C and slowly add hydrazine hydrate solution (60-85% concentration), then maintain reaction temperature between 70°C and 85°C for 1 to 5 hours.
- Filter the hot reaction mixture to remove the catalyst, cool the mother liquor, add purified water to induce crystallization, and vacuum dry the resulting crystals.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis route offers profound advantages in terms of cost structure and supply chain resilience, making it an attractive option for cost reduction in electronic chemical manufacturing. The elimination of high-pressure hydrogenation equipment reduces both capital investment and ongoing maintenance costs, while the use of commodity chemicals like hydrazine hydrate and ethanol ensures raw material availability and price stability. The simplified workup procedure, which relies on crystallization rather than complex distillation or chromatography, significantly reduces utility consumption (steam and electricity) and shortens the batch cycle time. For supply chain heads, the robustness of this process means reduced risk of production delays caused by equipment failure or complex purification bottlenecks, ensuring a steady flow of critical monomers for downstream polymerization.
- Cost Reduction in Manufacturing: The economic benefits are driven primarily by the operational simplicity and the high atom economy of the transfer hydrogenation reaction. By removing the need for expensive high-pressure reactors and the associated safety infrastructure, manufacturers can achieve substantial capital expenditure savings. Additionally, the ability to recover and potentially recycle the heterogeneous Pd/C catalyst after hot filtration mitigates the cost impact of using precious metals. The high yield reported in the patent examples, ranging from 87.6% to 95.4%, combined with the absence of costly purification steps, results in a significantly lower cost per kilogram of the final active pharmaceutical ingredient or electronic intermediate compared to traditional reduction methods.
- Enhanced Supply Chain Reliability: The reliance on widely available solvents such as methanol, ethanol, and ethylene glycol decouples the production process from the volatility of specialized reagent markets. Unlike processes that depend on niche catalysts or hazardous gases requiring special transport logistics, this method utilizes liquid reagents that are easy to store and handle in standard chemical warehouses. The short reaction time of 1 to 5 hours allows for rapid turnover of reactor vessels, increasing the overall throughput capacity of existing facilities. This agility enables suppliers to respond quickly to fluctuations in market demand for polyimide precursors, thereby reducing lead time for high-purity polyimide monomers and enhancing the reliability of supply for key account customers.
- Scalability and Environmental Compliance: The process is inherently green, generating minimal three wastes (waste water, waste gas, and solid residue) compared to metal-acid reduction techniques. The primary byproduct of the hydrazine oxidation is nitrogen gas, which vents safely, and the aqueous waste streams are easier to treat due to the absence of heavy metal ions. This environmental profile simplifies regulatory compliance and reduces the burden on wastewater treatment facilities, which is a critical factor for scaling up production in regions with strict environmental regulations. The crystallization-based isolation is easily scalable from pilot plant to multi-ton commercial production without losing efficiency, ensuring that the quality and consistency of the product remain stable as volumes increase to meet global demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 3,5-bis(2,4-diaminophenoxy)toluene. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing clarity on the feasibility and advantages of this specific manufacturing route. Understanding these details helps stakeholders evaluate the suitability of this material for their specific high-performance polymer applications.
Q: What is the achievable purity of 3,5-bis(2,4-diaminophenoxy)toluene using this method?
A: According to patent CN101260060A, this specific catalytic reduction pathway consistently achieves product purity exceeding 99%, with specific examples demonstrating purity levels of 99.5% to 99.8% without requiring complex chromatographic purification.
Q: Can the palladium catalyst be recovered in this process?
A: Yes, the process utilizes a heterogeneous Pd/C catalyst which is removed via hot filtration before the crystallization step. This allows for potential catalyst recovery and recycling, significantly reducing the cost of goods sold (COGS) associated with precious metal usage.
Q: Is this synthesis route scalable for commercial production?
A: The method is explicitly designed for industrialization, utilizing standard unit operations such as stirred tank reactors, filtration, and crystallization. The avoidance of high-pressure hydrogen gas and the use of common solvents like ethanol and ethylene glycol make it highly amenable to large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,5-Bis(2,4-Diaminophenoxy)Toluene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity monomers play in the performance of next-generation electronic materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory synthesis to industrial manufacturing is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of 3,5-bis(2,4-diaminophenoxy)toluene meets the exacting standards required for FCCL and advanced composite applications. Our commitment to quality assurance guarantees that our clients receive materials that enable the fabrication of polyimides with consistent thermal and mechanical properties.
We invite procurement leaders and technical directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized catalytic processes, we can help you reduce total landed costs while securing a stable supply of critical intermediates. Please contact our technical procurement team to request specific COA data and route feasibility assessments for your upcoming projects. We are dedicated to being your strategic partner in navigating the complexities of the global fine chemical supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
