Advanced Synthesis of 2,2'-Difluoro-4,4',5,5'-Biphenyl Tetracarboxylic Dianhydride for Next-Gen Electronics
The rapid evolution of the microelectronics and 5G communication sectors demands materials that can withstand extreme thermal conditions while maintaining superior electrical insulation and dimensional stability. In this context, patent CN114349726B introduces a groundbreaking advancement in the field of organic synthesis and polymer material preparation, specifically detailing the synthesis and application of 2,2'-difluoro-4,4',5,5'-biphenyltetracarboxylic dianhydride monomer. This novel monomer addresses the critical trade-off often found in polyimide materials between thermal mechanical performance and photoelectric properties. By strategically introducing fluorine substituents at the 2,2' positions of the biphenyl backbone, the invention achieves a unique molecular configuration that preserves the high thermal resistance and low thermal expansion coefficient characteristic of rigid rod polymers, while simultaneously enhancing optical transparency and reducing dielectric constants. This technological leap positions the monomer as a cornerstone for next-generation aerospace, flexible display, and high-frequency circuit board applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional fully aromatic polyimides, such as those derived from 3,3',4,4'-biphenyltetracarboxylic dianhydride (BPDA), are renowned for their exceptional thermal stability and mechanical strength. However, their molecular chains tend to stack densely due to the planar nature of the aromatic rings and heterocyclic structures. While this dense packing confers excellent physical properties, it often leads to poor solubility and, more critically for modern electronics, suboptimal photoelectric performance. The high density of electron clouds in these conventional structures facilitates intermolecular charge transfer, resulting in deep coloration and higher dielectric constants that are detrimental to high-speed signal transmission. Furthermore, attempts to improve solubility and optical properties by introducing bulky side groups often come at the expense of thermal stability and linear thermal expansion coefficients, rendering them unsuitable for precision applications like flexible copper-clad laminates where dimensional integrity under thermal cycling is paramount.
The Novel Approach
The methodology outlined in the patent data presents a sophisticated solution by utilizing 3,4-dimethylaniline as a starting raw material to construct the fluorinated biphenyl skeleton. Unlike conventional methods that might rely on direct fluorination of existing dianhydrides which can be harsh and unselective, this approach builds the fluorine functionality into the backbone early in the synthesis. The process involves a sequence of amine protection, palladium-catalyzed oxidative coupling, deprotection, and a specific Balz-Schiemann fluorination reaction. This stepwise construction allows for precise control over the substitution pattern, ensuring that fluorine atoms are located exactly at the 2,2' positions. This specific placement leverages the small van der Waals volume of fluorine to avoid disrupting molecular chain stacking, thus retaining the low CTE, while its strong electron-withdrawing nature effectively suppresses charge transfer complexes. The result is a monomer that enables the production of polyimides with a rare combination of high thermal decomposition temperatures, low dielectric loss, and enhanced optical transparency.
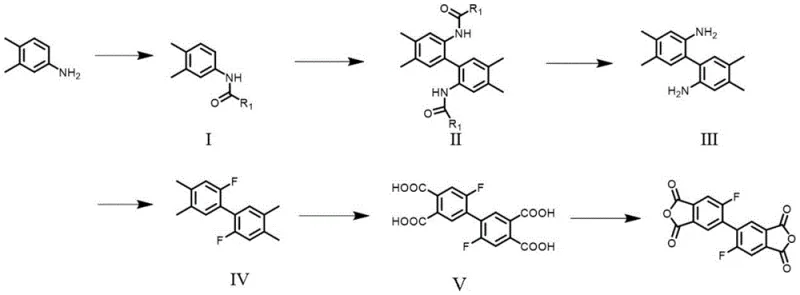
Mechanistic Insights into Pd-Catalyzed Oxidative Coupling and Fluorination
The core of this synthetic innovation lies in the efficient construction of the biaryl bond and the subsequent introduction of fluorine. The coupling reaction utilizes potassium persulfate as an oxidant and palladium acetate as a catalyst in a mixed solvent system of trifluoroacetic acid and methanesulfonic acid. This catalytic system facilitates the oxidative homocoupling of the protected aniline intermediates at room temperature, a condition that is remarkably mild compared to traditional Ullmann-type couplings which often require high temperatures and stoichiometric amounts of copper. The mechanism likely involves the generation of aryl radical cations or palladium-aryl intermediates that couple to form the biphenyl linkage with high regioselectivity. Following the coupling, the amine protecting groups are removed under acidic or basic conditions to regenerate the free amino groups, which are then subjected to diazotization using sodium nitrite and tetrafluoroboric acid. The resulting diazonium tetrafluoroborate salts are thermally decomposed to yield the aryl fluoride bonds, a classic Balz-Schiemann transformation that ensures the fluorine is covalently bonded to the aromatic ring with high stability.
Impurity control is meticulously managed throughout this multi-step sequence. The initial protection of the amine group prevents unwanted side reactions during the oxidative coupling phase, ensuring that the catalyst interacts primarily with the desired positions on the aromatic ring. The use of specific alkyl acid chlorides for protection allows for tunable solubility of the intermediates, facilitating purification via recrystallization or column chromatography before the critical fluorination step. Furthermore, the oxidation of the methyl groups to carboxylic acids is performed using potent oxidants like potassium permanganate or nitric acid under controlled temperatures, ensuring complete conversion to the tetra-acid intermediate without degrading the sensitive fluorine-substituted backbone. The final dehydration and cyclization step, typically conducted at temperatures exceeding 150°C or via acetic anhydride reflux, drives the equilibrium towards the formation of the five-membered anhydride rings, yielding the high-purity dianhydride monomer essential for polymerization.
How to Synthesize 2,2'-Difluoro-BPDA Efficiently
The synthesis of this high-performance monomer requires precise adherence to reaction conditions to maximize yield and purity. The process begins with the protection of 3,4-dimethylaniline, followed by the critical palladium-catalyzed coupling which forms the biphenyl core. Subsequent steps involve deprotection, fluorination via the Balz-Schiemann reaction, oxidation of methyl groups to carboxylic acids, and finally thermal or chemical imidization to close the anhydride rings. Each step has been optimized in the patent examples to demonstrate scalability and reproducibility, with specific attention paid to solvent ratios, catalyst loading, and temperature profiles to minimize byproduct formation. For detailed operational parameters and stoichiometry, refer to the standardized guide below.
- Protect the amino group of 3,4-dimethylaniline with alkyl acid chloride to form Intermediate I.
- Perform oxidative coupling of Intermediate I using potassium persulfate and palladium acetate catalyst to form the biphenyl backbone (Intermediate II).
- Deprotect the amine groups to yield Intermediate III, followed by Balz-Schiemann fluorination to introduce fluorine atoms at the 2,2' positions.
- Oxidize the methyl groups to carboxylic acids using potassium permanganate or nitric acid, followed by dehydration and cyclization to form the final dianhydride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers significant strategic advantages beyond mere technical performance. The reliance on 3,4-dimethylaniline as a starting material leverages a widely available and cost-effective commodity chemical, reducing dependency on exotic or scarce precursors that often plague the supply chains of specialized electronic chemicals. The synthetic pathway avoids the use of expensive transition metal catalysts in stoichiometric quantities, utilizing palladium acetate in catalytic amounts (2% to 8% molar ratio), which drastically reduces the raw material cost per kilogram of the final product. Moreover, the ability to perform the coupling reaction at room temperature translates to substantial energy savings in a commercial manufacturing setting, lowering the overall utility costs associated with heating and cooling large-scale reactors.
- Cost Reduction in Manufacturing: The elimination of harsh reaction conditions and the use of catalytic rather than stoichiometric metal reagents significantly lower the operational expenditure. The process avoids the need for complex downstream processing to remove heavy metal residues, as the catalyst loading is minimal and the workup procedures described involve standard aqueous washes and extractions. This simplification of the purification train reduces solvent consumption and waste disposal costs, contributing to a leaner and more cost-efficient manufacturing process for high-purity electronic chemical intermediates.
- Enhanced Supply Chain Reliability: By utilizing a synthetic route based on robust and well-understood chemical transformations such as amide protection and oxidative coupling, the risk of batch failure is minimized. The intermediates formed during the synthesis are stable and can be isolated and stored if necessary, providing flexibility in production scheduling. This robustness ensures a consistent supply of the monomer, which is critical for downstream polymer manufacturers who require uninterrupted feedstock to maintain their own production schedules for polyimide films and resins used in critical aerospace and microelectronics applications.
- Scalability and Environmental Compliance: The reaction conditions described, such as the use of mixed acid solvents and standard oxidants, are readily scalable from laboratory to industrial tonnage without requiring specialized high-pressure or cryogenic equipment. The process design inherently supports green chemistry principles by maximizing atom economy in the coupling step and allowing for the recovery and reuse of solvents. This alignment with environmental compliance standards reduces the regulatory burden and potential liabilities associated with hazardous waste generation, making it a sustainable choice for long-term commercial production of advanced polymer additives.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the properties and processing of the fluorinated dianhydride monomer described in the patent. These insights are derived directly from the experimental data and comparative analysis provided in the documentation, offering clarity on how this material performs relative to standard industry benchmarks. Understanding these nuances is essential for R&D teams evaluating this monomer for specific formulation requirements in high-end electronic applications.
Q: How does the introduction of fluorine atoms affect the thermal expansion coefficient (CTE) of the resulting polyimide?
A: According to patent CN114349726B, the fluorine atom has the smallest van der Waals volume except for hydrogen. Its introduction into the 2,2' position of the biphenyl tetracarboxylic dianhydride structure does not disrupt the close stacking of molecular chains, thereby maintaining a low coefficient of thermal expansion (CTE) comparable to non-fluorinated analogs while improving other properties.
Q: What are the dielectric advantages of using this fluorinated monomer in 5G applications?
A: The strong electron-withdrawing effect of the fluorine atoms inhibits the formation of intermolecular charge transfer complexes. This results in significantly improved optical transparency and reduced dielectric constant (Dk) and dielectric loss (Dl), which are critical for high-frequency signal transmission in 5G communication and flexible display technologies.
Q: Does the fluorination process compromise the thermal stability of the polyimide?
A: No, the thermal stability remains robust. The aromatic C-F bond possesses high bond energy and a short bond length, contributing to excellent thermal stability. Experimental data indicates that the glass transition temperature (Tg) remains above 390°C and the thermal decomposition temperature (T5%) exceeds 565°C, ensuring reliability in high-temperature processing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2'-Difluoro-BPDA Supplier
As the demand for high-performance polyimides continues to surge in the 5G and flexible electronics markets, securing a reliable supply of advanced monomers like 2,2'-difluoro-4,4',5,5'-biphenyltetracarboxylic dianhydride is crucial. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for electronic grade materials. We understand that the consistency of the monomer directly impacts the dielectric and thermal properties of the final polyimide, and our commitment to quality assurance ensures that every batch meets the exacting standards necessary for aerospace and microelectronics applications.
We invite you to collaborate with us to optimize your material sourcing strategy. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. By partnering with us, you gain access to not just a product, but a comprehensive supply chain solution that includes specific COA data and route feasibility assessments. Contact our technical procurement team today to discuss how we can support your next-generation material development projects with reliable, high-quality fluorinated dianhydride monomers.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
