Advanced Synthesis of Active Vinyl Polyimide Powder for High-Performance Electronic Materials
Advanced Synthesis of Active Vinyl Polyimide Powder for High-Performance Electronic Materials
The development of high-performance polymers for the electronics industry continues to drive innovation in material science, particularly in the realm of polyimides. Patent CN101423609B introduces a significant advancement in the preparation of active vinyl-containing polyimide powder, a material critical for next-generation flexible printed circuits and high-temperature composites. This technology addresses the growing demand for materials that combine exceptional thermal stability with reactive functionality, allowing for seamless integration into complex resin systems. By utilizing a controlled multi-step polymerization followed by thermal imidization, the process ensures high purity and consistent molecular weight distribution. As a reliable electronic chemical supplier, understanding the nuances of such patented methodologies is essential for delivering products that meet the rigorous standards of modern semiconductor and aerospace applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing polyimide powders often rely heavily on chemical imidization techniques that utilize aggressive reagents such as acetic anhydride and tertiary amines. These conventional pathways frequently result in the generation of substantial chemical waste, necessitating complex purification steps to remove residual salts and by-products that can compromise the dielectric properties of the final material. Furthermore, standard processes may struggle to incorporate reactive functional groups like vinyl moieties without causing premature cross-linking or gelation during the synthesis phase. The inability to precisely control the reaction environment often leads to batch-to-batch variability, which is unacceptable for high-precision electronic manufacturing where consistency is paramount. Additionally, the use of harsh chemical dehydrating agents can degrade sensitive monomer structures, limiting the scope of achievable polymer architectures.
The Novel Approach
The methodology outlined in the patent data presents a transformative approach by employing a thermal azeotropic dehydration technique that eliminates the need for stoichiometric chemical dehydrating agents. This novel route involves a carefully staged addition of monomers, starting with the reaction of aromatic quaternary primary amines and unsaturated acid anhydrides at low temperatures to preserve the integrity of the vinyl groups. By maintaining the reaction temperature between 0°C and 25°C during the polyamic acid formation stage, the process effectively prevents unwanted side reactions while ensuring a homogeneous solution. The subsequent imidization is achieved through heating with an azeotropic agent like toluene or xylene in the presence of an acid catalyst, which drives the equilibrium towards the imide ring closure efficiently. This results in a cleaner product profile with significantly reduced impurity levels, making it ideal for cost reduction in electronic chemical manufacturing.
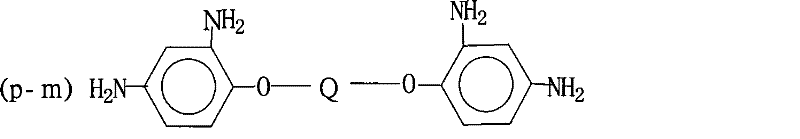
Mechanistic Insights into Azeotropic Dehydration Imidization
The core of this synthesis lies in the precise control of the polycondensation and cyclodehydration mechanisms. Initially, the nucleophilic attack of the amine groups on the carbonyl carbons of the anhydrides forms the polyamic acid precursor. The inclusion of unsaturated anhydrides, such as maleic anhydride or nadic anhydride, introduces the critical vinyl functionality directly into the polymer backbone or side chains. This step requires strict temperature regulation to avoid the polymerization of the vinyl groups before the polyimide structure is fully established. The transition from polyamic acid to polyimide is then facilitated by the removal of water generated during ring closure. Unlike chemical methods that trap water in salt forms, this thermal method physically removes water via azeotropic distillation. The use of catalysts like p-toluenesulfonic acid accelerates this cyclization without introducing metallic contaminants, thereby preserving the electrical insulation properties required for IC passivation layers.
Impurity control is inherently built into this mechanistic design through the selection of high-purity monomers and the efficiency of the azeotropic process. The use of strong polar aprotic solvents such as N-methyl-2-pyrrolidone (NMP) or N,N-dimethylacetamide ensures complete solubility of the high molecular weight intermediates, preventing phase separation that could lead to defects. The final precipitation and washing steps further purify the powder by removing any unreacted oligomers or residual solvent. This rigorous control over the reaction pathway ensures that the resulting polyimide powder exhibits the low water absorption and excellent dimensional stability cited in the patent benefits. For R&D directors, this level of mechanistic clarity offers confidence in the scalability and reproducibility of the material for critical applications.
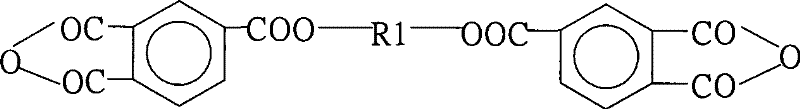
How to Synthesize Active Vinyl Polyimide Powder Efficiently
The synthesis protocol detailed in the patent provides a robust framework for producing high-quality polyimide powders suitable for industrial scale-up. The process begins with the dissolution of specific aromatic amines and anhydrides in a controlled solvent system, followed by a staged polymerization to build the desired molecular architecture. Critical parameters such as the molar ratio of quaternary amines to dibasic amines and the timing of anhydride addition are pivotal for achieving the target viscosity and molecular weight. The detailed standardized synthesis steps below outline the precise conditions required to replicate the high yields and purity levels demonstrated in the patent examples, ensuring that the active vinyl groups remain intact for subsequent curing processes.
- Dissolve aromatic quaternary primary amine and unsaturated acid anhydride (1: 2 molar ratio) in polar aprotic solvent at 0-25°C, then add aromatic dibasic primary amine.
- Add aromatic dibasic anhydride to the solution and react at 0-25°C for 2-4 hours to form a homogeneous viscous polyamic acid solution.
- Add azeotropic dehydrating agent and catalyst, then heat to 80-150°C for 5-12 hours to complete imidization, followed by precipitation and drying.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthesis route offers distinct advantages that translate directly into operational efficiency and risk mitigation. The reliance on general-purpose equipment rather than specialized reactors lowers the barrier to entry for production and reduces capital expenditure requirements. The elimination of expensive and hazardous chemical imidization reagents simplifies the raw material sourcing strategy, reducing dependency on volatile supply chains for niche chemicals. Furthermore, the environmental friendliness of the process, characterized by the recovery and recycling of organic solvents, aligns with increasingly stringent global regulations on industrial emissions. This compliance reduces the administrative burden and potential fines associated with waste disposal, contributing to substantial cost savings over the lifecycle of the product.
- Cost Reduction in Manufacturing: The process achieves cost optimization primarily through the simplification of the reaction workflow and the avoidance of costly post-processing purification steps. By utilizing thermal dehydration instead of chemical agents, the method eliminates the generation of salt by-products that require extensive washing and wastewater treatment. The high yields reported in the patent examples, ranging significantly above typical industry averages for similar complex polymers, indicate a highly efficient conversion of raw materials into finished product. This efficiency minimizes raw material waste and maximizes output per batch, driving down the unit cost of production without compromising on the quality specifications required for high-end electronic applications.
- Enhanced Supply Chain Reliability: The raw materials utilized in this synthesis, such as common aromatic diamines and anhydrides, are widely available commodities in the fine chemical market. This abundance ensures a stable supply base that is less susceptible to the disruptions often seen with exotic or proprietary catalysts. The ability to perform the synthesis in standard solvents like NMP or DMAc further enhances supply security, as these are produced at massive scales globally. For supply chain heads, this translates to reduced lead times for high-purity polyimide powders and a lower risk of production stoppages due to material shortages, ensuring continuous availability for downstream manufacturing partners.
- Scalability and Environmental Compliance: The commercial scale-up of complex polyimide additives is often hindered by heat transfer issues and solvent management, but this azeotropic method is inherently scalable. The use of reflux conditions allows for effective temperature control even in large reactors, mitigating the risk of thermal runaway. Additionally, the closed-loop nature of the solvent recovery system significantly reduces volatile organic compound (VOC) emissions. This environmental compatibility facilitates easier permitting for new production facilities and supports corporate sustainability goals. The robustness of the process ensures that quality remains consistent whether producing pilot batches or multi-ton commercial runs, providing a reliable foundation for long-term supply agreements.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of active vinyl polyimide powders. These answers are derived directly from the technical specifications and beneficial effects described in the patent literature, providing clarity on the material's capabilities and processing requirements. Understanding these details is crucial for integrating this advanced material into existing manufacturing workflows and maximizing its performance benefits in final applications.
Q: What are the key advantages of this polyimide powder synthesis method?
A: The method described in patent CN101423609B offers a simple process with low cost and environmental friendliness. It utilizes general-purpose equipment suitable for industrial production, avoiding complex chemical imidization reagents.
Q: What applications does active vinyl polyimide powder serve?
A: Due to its reactive vinyl groups, this powder acts as a high-temperature toughening modifier for epoxy and bismaleimide resins. It is also used in FPC and PCB substrate materials for its excellent dielectric and mechanical properties.
Q: How is the imidization reaction completed in this process?
A: The process employs thermal azeotropic dehydration. A dehydrating agent like toluene or xylene is added along with an acid catalyst, and the mixture is heated to 80-150°C to remove water and cyclize the polyamic acid into polyimide.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Active Vinyl Polyimide Powder Supplier
The technological potential of active vinyl polyimide powders represents a significant opportunity for advancing high-performance materials in the electronics and aerospace sectors. NINGBO INNO PHARMCHEM stands ready to support your development goals with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art reactors capable of handling the precise temperature controls and solvent management required for this sophisticated synthesis. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch meets the exacting standards necessary for critical applications like FPC substrates and high-temperature adhesives.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis route to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized manufacturing processes can reduce your overall material costs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will demonstrate our commitment to quality and reliability as your strategic partner in advanced polymer solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
