Scalable Synthesis of High-Purity Diperylene Diimides for Next-Gen Optoelectronics
The rapid evolution of organic electronics demands materials with exceptional charge transport properties and thermal stability. A pivotal development in this sector is detailed in patent CN101205229A, which introduces a robust methodology for synthesizing diperylene tetracarboxylic acid diimide compounds. These bis-perylene structures represent a significant leap forward in the design of n-type organic semiconductors, offering extended pi-conjugation that is critical for high-performance applications such as organic field-effect transistors (OFETs) and photovoltaic devices. As a leading entity in the fine chemical sector, we recognize that the ability to reliably produce these complex architectures is a key differentiator for any reliable electronic chemical supplier. The patent outlines a copper-catalyzed dimerization strategy that transforms halogenated perylene precursors into sophisticated bis-structures, addressing the longstanding challenge of constructing large, planar aromatic systems with precision.
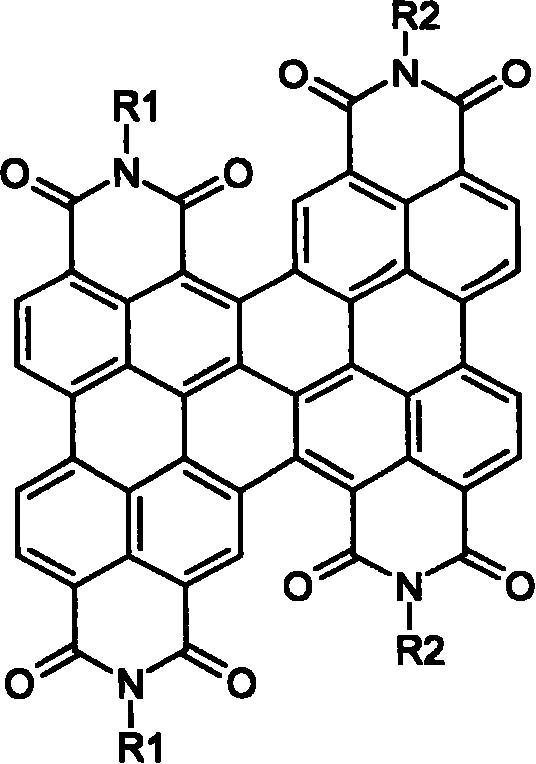
The structural versatility of these compounds allows for extensive customization. As illustrated in the general formula, the R1 and R2 groups can be independently selected from a vast array of substituents, including C1 to C30 alkyl chains, alkoxy groups, and various aryl moieties. This modularity is essential for cost reduction in electronic chemical manufacturing, as it permits the tuning of solubility profiles without compromising the core electronic properties of the perylene backbone. By adjusting these side chains, manufacturers can optimize the material for solution processing techniques, which are increasingly favored in the production of large-area organic electronic devices. The inherent stability of the perylene core, combined with the tunable periphery, makes these compounds ideal candidates for next-generation display technologies and energy storage solutions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of extended perylene arrays has been fraught with difficulties, often requiring harsh conditions that degrade the delicate imide functionalities or result in poor regioselectivity. Conventional routes frequently rely on multi-step condensations that suffer from low atom economy and generate significant amounts of hazardous waste. Furthermore, achieving the specific bay-area coupling required to link two perylene units often necessitates expensive palladium catalysts or intricate protecting group strategies that drive up the overall production cost. For procurement teams, these inefficiencies translate into volatile pricing and inconsistent supply chains for high-purity OLED material precursors. The reliance on precious metals also introduces purification bottlenecks, as removing trace metal residues to meet electronic-grade specifications can be technically demanding and time-consuming.
The Novel Approach
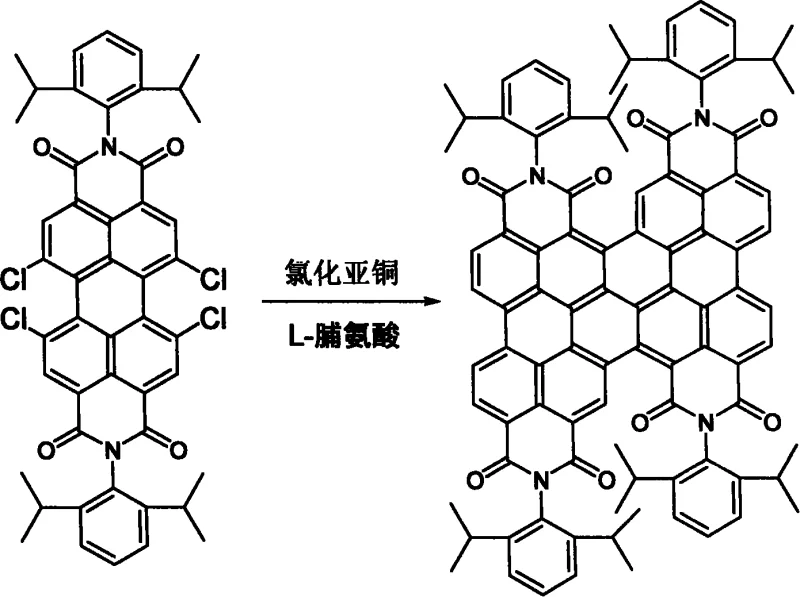
The methodology described in the patent offers a transformative alternative by utilizing a copper-mediated coupling mechanism that is both cost-effective and operationally simple. By employing inexpensive copper(I) salts such as cuprous chloride or cuprous iodide in conjunction with amino acid ligands like L-proline, the process achieves efficient C-C bond formation under relatively mild conditions. This approach eliminates the need for precious metal catalysts, thereby significantly lowering the raw material costs associated with production. The reaction proceeds in polar aprotic solvents like dimethyl sulfoxide (DMSO), which effectively solubilizes the reactants and facilitates the coupling event. This streamlined protocol not only simplifies the workflow but also enhances the scalability of the process, making it suitable for the commercial scale-up of complex polymer additives and semiconductor intermediates.
Mechanistic Insights into Cu-Catalyzed Dimerization
The core of this synthetic breakthrough lies in the copper-catalyzed Ullmann-type coupling mechanism, which facilitates the homocoupling of halogenated perylene diimides. In this catalytic cycle, the copper(I) species undergoes oxidative addition into the carbon-halogen bond of the perylene precursor, forming an organocopper intermediate. The presence of the amino acid ligand is crucial, as it stabilizes the copper center and enhances its reactivity towards the sterically hindered bay positions of the perylene core. Subsequent reductive elimination releases the coupled bis-perylene product and regenerates the active copper catalyst. This mechanism is particularly advantageous because it tolerates a wide range of functional groups on the imide nitrogen, allowing for the synthesis of diverse derivatives without the need for extensive protection-deprotection sequences.
Impurity control is another critical aspect where this method excels. The use of inorganic bases such as potassium carbonate or cesium carbonate helps to neutralize acidic byproducts and drives the reaction equilibrium towards the desired product. Moreover, the reaction conditions—typically heating at 50°C to 150°C for 1 to 5 days under inert gas protection—are optimized to minimize side reactions such as dehalogenation or over-coupling. The resulting crude products can be effectively purified using standard silica gel column chromatography, yielding materials with high purity levels suitable for electronic applications. This robustness in impurity management ensures that the final high-purity diperylene diimides meet the stringent quality standards required for use in sensitive optoelectronic devices.
How to Synthesize Diperylene Tetracarboxylic Acid Diimide Efficiently
The practical implementation of this synthesis involves a straightforward mixing of reagents followed by thermal treatment. The process begins with the combination of the halogenated precursor, catalyst, ligand, and base in a specific molar ratio, ensuring optimal catalytic turnover. The choice of solvent and temperature is then adjusted based on the specific reactivity of the halogenated starting material, with chlorinated precursors generally requiring slightly higher activation energies than their brominated counterparts. Detailed operational parameters are critical for reproducibility, and the following guide outlines the standardized procedure derived from the patent examples.
- Mix halogenated perylene diimide precursor with Cu(I) catalyst, amino acid ligand, and inorganic base in a molar ratio of approximately 1: 6:7:8.
- Add dimethyl sulfoxide (DMSO) or its derivatives as the solvent to achieve a concentration of 0.01 to 1.0 mmol/ml.
- Heat the mixture to 50°C–150°C under inert gas protection for 1 to 5 days, followed by purification via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain managers and procurement specialists, the adoption of this copper-catalyzed route presents several compelling economic and logistical benefits. The shift away from precious metal catalysts to abundant copper salts drastically reduces the dependency on volatile commodity markets, leading to more stable pricing structures for raw materials. Additionally, the simplicity of the workup procedure, which involves basic aqueous washes and solvent evaporation, minimizes the consumption of specialized reagents and reduces the generation of hazardous waste streams. These factors collectively contribute to a more sustainable and cost-efficient manufacturing process that aligns with modern green chemistry principles.
- Cost Reduction in Manufacturing: The elimination of expensive palladium or platinum catalysts results in substantial cost savings on a per-kilogram basis. Furthermore, the use of inexpensive amino acid ligands and common inorganic bases lowers the overall reagent cost profile. The simplified purification process also reduces labor and solvent costs associated with downstream processing, making the final product more competitive in the global market.
- Enhanced Supply Chain Reliability: The starting materials, specifically halogenated perylene diimides, are commercially available or can be synthesized via established literature methods, ensuring a steady supply of feedstock. The robustness of the reaction conditions means that production is less susceptible to minor fluctuations in temperature or pressure, reducing the risk of batch failures and ensuring consistent delivery schedules for clients.
- Scalability and Environmental Compliance: The reaction uses DMSO, a solvent with a high boiling point that can be recovered and recycled, minimizing environmental impact. The absence of heavy metal residues simplifies waste disposal and regulatory compliance, facilitating easier permitting for large-scale production facilities. This scalability is essential for meeting the growing demand for organic electronic materials in the consumer electronics sector.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the synthesis and application of these advanced materials. The answers are derived directly from the experimental data and claims presented in the patent documentation, providing accurate insights for researchers and engineers evaluating this technology for their own processes.
Q: What is the primary advantage of this dimerization method?
A: The method utilizes a simple Cu-catalyzed coupling in DMSO, avoiding complex transition metal systems and allowing for the direct formation of extended pi-conjugated systems from readily available halogenated precursors.
Q: What types of substituents are compatible with this synthesis?
A: The process is highly versatile, accommodating R groups such as C1–C30 alkyl, alkoxy, aryl, and heteroaryl groups, enabling fine-tuning of solubility and electronic properties.
Q: What are the typical yields for this reaction?
A: According to the patent data, purified yields typically range from 10% to 50%, depending on the specific halogenated precursor and substituent groups used.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diperylene Tetracarboxylic Acid Diimide Supplier
The synthesis of diperylene tetracarboxylic acid diimides represents a sophisticated intersection of organic synthesis and materials science, requiring a partner with deep technical expertise and manufacturing capability. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. Our rigorous QC labs and stringent purity specifications guarantee that every batch of material delivered meets the exacting standards necessary for high-performance electronic applications.
We invite you to collaborate with us to optimize your supply chain for these critical materials. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. By leveraging our process knowledge, we can help you navigate the complexities of sourcing high-value intermediates. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our capabilities can support your innovation goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
