Advanced Synthesis of High-Purity Polyimide Monomers for Next-Generation Electronic Materials
The global demand for high-performance polyimides in the aerospace and microelectronics sectors has necessitated the development of robust synthetic routes for specialized monomers. Patent CN101381312A introduces a groundbreaking methodology for the preparation of 3,3'-dinitro-4,4'-bis(2,4-dinitrophenoxy)biphenyl, a critical precursor for highly branched aromatic polyimides. This specific compound serves as a foundational building block for creating polymers with exceptional thermal stability, mechanical strength, and electrical insulation properties required for advanced LCD displays and semiconductor applications. The disclosed process represents a significant leap forward in industrial chemistry, offering a pathway that combines high purity with operational simplicity. By leveraging a specific nucleophilic aromatic substitution strategy, the inventors have solved longstanding challenges related to solubility and reaction equilibrium in highly nitrated systems. For R&D directors and procurement specialists alike, understanding the nuances of this patent is essential for securing a reliable supply chain of next-generation electronic materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of complex nitro-ether biphenyl derivatives has been plagued by significant technical hurdles that impede large-scale commercialization. Conventional approaches often struggle with the poor solubility of highly nitrated starting materials in standard organic solvents, leading to heterogeneous reaction mixtures that result in inconsistent conversion rates and extended reaction times. Furthermore, without precise control over water removal, the reversible nature of the etherification reaction can lead to substantial hydrolysis of the sensitive nitro-halide reactants, drastically reducing overall yield. Many historical methods also rely on aggressive conditions or stoichiometric excesses of reagents that generate difficult-to-remove impurities, necessitating costly and time-consuming purification steps such as column chromatography which are entirely unsuitable for ton-scale production. The accumulation of these inefficiencies translates directly into higher manufacturing costs and unpredictable supply continuity for downstream polymer manufacturers.
The Novel Approach
The methodology outlined in patent CN101381312A elegantly circumvents these traditional bottlenecks through a sophisticated dual-solvent system and azeotropic water removal technique. By employing a mixture of a water-insoluble organic solvent, such as toluene or xylene, and a strong polar aprotic solvent like N,N-dimethylformamide (DMF), the process ensures complete solubilization of both the phenolic substrate and the nitro-halide electrophile. This homogeneity is critical for maintaining consistent reaction kinetics throughout the batch. Moreover, the integration of a water-dividing step during the reflux period actively shifts the chemical equilibrium toward the product side by continuously removing the water byproduct, thereby driving the reaction to near-completion without requiring extreme pressures. This approach not only simplifies the operational protocol to atmospheric pressure conditions but also facilitates the straightforward recovery and recycling of the organic solvents, aligning perfectly with modern green chemistry principles and cost-reduction mandates.
Mechanistic Insights into Nucleophilic Aromatic Substitution
The core chemical transformation driving this synthesis is a classic nucleophilic aromatic substitution ($S_NAr$), facilitated by the strong electron-withdrawing effects of the nitro groups positioned ortho and para to the leaving halogen atom. In this mechanism, the phenoxide anion, generated in situ by the deprotonation of 3,3'-dinitro-4,4'-dihydroxybiphenyl by the salt-forming agent, acts as a potent nucleophile. It attacks the electron-deficient aromatic ring of the 2,4-dinitrohalobenzene, forming a resonance-stabilized Meisenheimer complex intermediate. The presence of two nitro groups on the electrophilic ring significantly lowers the energy barrier for this addition step, making the reaction feasible under relatively mild thermal conditions compared to unsubstituted aryl halides. The subsequent elimination of the halide ion restores aromaticity, yielding the stable diaryl ether linkage. Understanding this electronic activation is crucial for process optimization, as it explains why the reaction proceeds efficiently even with less reactive leaving groups like chlorine when supported by the correct solvent environment.
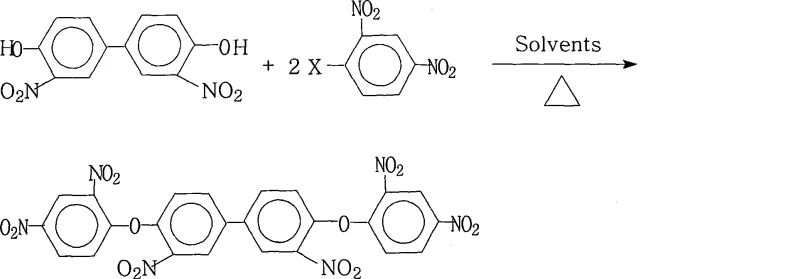
Beyond the primary substitution mechanism, the patent highlights a critical aspect of impurity control through the management of the salt-forming agent and reaction atmosphere. The use of bases such as potassium carbonate or potassium hydroxide must be carefully balanced; insufficient base leads to incomplete conversion of the phenol, while excessive base can promote side reactions or degradation of the sensitive nitro groups under prolonged heating. The nitrogen atmosphere specified in the protocol is not merely a precaution but a mechanistic necessity to prevent oxidative degradation of the electron-rich phenoxide intermediates and to exclude moisture that could compete with the nucleophile. By strictly controlling these parameters, the process achieves purities exceeding 99%, effectively minimizing the formation of mono-substituted byproducts or hydrolyzed phenols that would otherwise compromise the molecular weight and thermal properties of the final polyimide material.
How to Synthesize 3,3'-Dinitro-4,4'-bis(2,4-dinitrophenoxy)biphenyl Efficiently
To implement this synthesis effectively in a pilot or production plant, operators must adhere to the precise stoichiometric ratios and thermal profiles defined in the patent examples. The process begins with the careful charging of the reactor under an inert nitrogen blanket to ensure an oxygen-free environment, followed by the sequential addition of the dihydroxybiphenyl substrate, the nitro-halide electrophile, and the chosen inorganic base. The selection of the solvent mixture is paramount, typically involving a volume ratio that balances the solvating power of the polar aprotic component with the water-carrying capacity of the non-polar component. Once the mixture is heated to reflux, the water separator becomes the focal point of the operation, as the rate of water collection serves as a real-time indicator of reaction progress. Detailed standardized operating procedures for scaling this specific pathway from laboratory glassware to industrial reactors are provided in the technical guide below.
- Charge the reactor with 3,3'-dinitro-4,4'-dihydroxybiphenyl and 2,4-dinitrohalobenzene in a molar ratio of 1.0: 2.0 to 2.2, along with a salt-forming agent and a mixed solvent system.
- Heat the mixture under a nitrogen atmosphere to reflux temperatures between 80°C and 200°C, utilizing a water separator to remove byproduct water for 3 to 18 hours.
- Concentrate the reaction solution, cool the system, add water to precipitate the solid product, then filter, wash with hot water, and dry to obtain the final crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN101381312A offers tangible strategic benefits that extend far beyond simple chemical yield. The most significant advantage lies in the drastic simplification of the downstream processing workflow. Unlike traditional methods that may require complex extraction sequences or chromatographic purification to remove heavy metal catalysts or stubborn impurities, this process relies on a simple precipitation and filtration step. By adding water to the concentrated reaction mass, the product crystallizes out in high purity, allowing the mother liquor containing the valuable organic solvents to be easily separated and recycled. This closed-loop solvent capability significantly reduces the consumption of raw materials and minimizes the volume of hazardous waste requiring disposal, directly translating to lower operational expenditures and a reduced environmental footprint for the manufacturing facility.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by its ability to operate at atmospheric pressure, eliminating the need for expensive high-pressure autoclaves and the associated safety infrastructure. Furthermore, the flexibility in choosing the halogenated starting material allows procurement teams to optimize costs based on market fluctuations; for instance, switching between chloro- and fluoro-derivatives depending on availability without altering the core process parameters. The high conversion rates achieved through azeotropic water removal mean that raw material utilization is maximized, reducing the cost per kilogram of the active pharmaceutical or electronic intermediate. Additionally, the elimination of transition metal catalysts removes the costly and time-consuming step of metal scavenging, which is often a regulatory requirement for electronic grade materials.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the robustness of the reaction conditions, which tolerate minor variations in temperature and mixing without catastrophic failure, ensuring consistent batch-to-batch quality. The use of commodity chemicals such as toluene, xylene, and potassium carbonate as key reagents ensures that the supply chain is not dependent on exotic or single-source specialty reagents that are prone to logistical bottlenecks. The short reaction times, ranging from just 3 to 18 hours depending on the specific halide used, allow for faster turnaround times and higher throughput in existing reactor trains. This agility enables suppliers to respond more rapidly to spikes in demand from the LCD or semiconductor industries, securing a competitive edge in a fast-moving market.
- Scalability and Environmental Compliance: Scaling this chemistry from the bench to multi-ton production is straightforward due to the absence of exothermic runaways or gas evolution hazards. The process generates minimal three-waste (wastewater, waste gas, solid waste), primarily consisting of inorganic salts and aqueous washings that are easier to treat than organic solvent sludge. The ability to recycle the organic solvent system repeatedly without significant loss of efficiency aligns with increasingly stringent global environmental regulations, future-proofing the manufacturing asset against tighter emission standards. This environmental compatibility not only reduces compliance costs but also enhances the brand reputation of the end-product as a sustainably manufactured electronic material.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and claims within the patent documentation to provide clarity for potential partners and licensees. Understanding these specifics is vital for assessing the feasibility of integrating this monomer into your existing polymer production lines.
Q: What are the critical reaction conditions for maximizing yield in this synthesis?
A: The patent specifies a critical balance of temperature (80°C to 200°C) and the use of a water-insoluble organic solvent mixed with a polar aprotic solvent. Crucially, the process requires heating under reflux with water separation (azeotropic distillation) for 3 to 18 hours to drive the equilibrium forward and prevent hydrolysis.
Q: Which halogenated starting materials are suitable for this reaction?
A: The process is versatile regarding the leaving group. The patent explicitly lists 2,4-dinitrofluorobenzene, 2,4-dinitrochlorobenzene, 2,4-dinitrobromobenzene, and 2,4-dinitroiodobenzene as viable reactants, allowing procurement teams to select based on availability and cost without compromising the reaction pathway.
Q: How does this method address environmental and waste disposal concerns?
A: This method significantly reduces environmental impact by utilizing a solvent system that allows for easy recovery and repeated recycling. Furthermore, the workup involves simple water precipitation and washing, avoiding the generation of complex hazardous waste streams typical of traditional heavy metal-catalyzed couplings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3'-Dinitro-4,4'-bis(2,4-dinitrophenoxy)biphenyl Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced polyimides hinges on the availability of ultra-high purity monomers produced via scalable and safe processes. Our team of process chemists has extensively analyzed the route disclosed in CN101381312A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this technology to fruition. We are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of 3,3'-dinitro-4,4'-bis(2,4-dinitrophenoxy)biphenyl meets the exacting standards demanded by the electronic materials sector. Our commitment to quality assurance means that we can deliver materials that consistently support the synthesis of polyimides with superior thermal and mechanical performance.
We invite you to collaborate with us to optimize your supply chain for high-performance polymer precursors. By leveraging our expertise in nucleophilic aromatic substitutions and solvent engineering, we can help you achieve significant efficiencies in your manufacturing operations. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can support your long-term production goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
