Advanced Synthesis of 1,4-Bis(2,4-Dinitrophenoxy)-2-Tert-Butylbenzene for High-Performance Polyimides
Advanced Synthesis of 1,4-Bis(2,4-Dinitrophenoxy)-2-Tert-Butylbenzene for High-Performance Polyimides
The rapid evolution of the electronic materials sector demands intermediates that combine exceptional thermal stability with precise structural integrity, particularly for applications in liquid crystal displays (LCDs) and aerospace composites. Patent CN101279919A introduces a groundbreaking preparation method for 1,4-bis(2,4-dinitrophenoxy)-2-tert-butylbenzene, a critical precursor for synthesizing aromatic polyimides with superior mechanical and electrical properties. This novel synthetic route addresses the longstanding challenges associated with producing high-purity aromatic ethers, offering a robust solution for manufacturers seeking to enhance the performance of next-generation functional materials. By leveraging a carefully optimized nucleophilic aromatic substitution strategy, this technology enables the efficient construction of complex molecular architectures essential for high-temperature resistant polymers. As a reliable electronic chemical supplier, understanding the nuances of such patented methodologies is crucial for securing a competitive edge in the global supply chain of advanced optoelectronic materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sterically hindered aromatic diethers like 1,4-bis(2,4-dinitrophenoxy)-2-tert-butylbenzene has been plagued by significant technical hurdles that impede commercial viability. Traditional approaches often rely on harsh reaction conditions, requiring elevated pressures or the use of expensive transition metal catalysts that introduce difficult-to-remove impurities into the final product stream. Furthermore, conventional methods frequently suffer from poor atom economy and generate substantial quantities of hazardous waste, creating severe environmental compliance burdens for production facilities. The lack of publicly available literature prior to this invention indicates that existing pathways were likely proprietary, inefficient, or simply incapable of delivering the requisite purity levels needed for high-end electronic applications. These inefficiencies translate directly into inflated production costs and extended lead times, posing a significant risk to supply chain continuity for downstream polymer manufacturers who depend on consistent quality.
The Novel Approach
In stark contrast to these legacy issues, the methodology disclosed in CN101279919A presents a streamlined, environmentally friendly process that operates under atmospheric pressure with remarkable efficiency. This innovative approach utilizes a dual-solvent system that facilitates continuous water removal, driving the reaction equilibrium towards completion without the need for exotic reagents or complex equipment. The process is designed to be inherently scalable, utilizing readily available starting materials such as 2-tert-butylhydroquinone and various 2,4-dinitrohalobenzenes to achieve yields that significantly outperform traditional benchmarks. By eliminating the need for corrosive substances and simplifying the workup procedure to a straightforward filtration and drying step, this method drastically reduces the operational complexity typically associated with fine chemical manufacturing. This represents a paradigm shift in cost reduction in display material manufacturing, allowing producers to allocate resources more effectively while minimizing their environmental footprint through solvent recycling protocols.
Mechanistic Insights into Nucleophilic Aromatic Substitution
The core of this synthetic breakthrough lies in a highly controlled nucleophilic aromatic substitution (SnAr) mechanism, where the electron-deficient nature of the 2,4-dinitrohalobenzene ring is exploited to facilitate bond formation. The presence of two strongly electron-withdrawing nitro groups ortho and para to the halogen leaving group dramatically activates the aromatic ring towards nucleophilic attack by the phenoxide anion derived from 2-tert-butylhydroquinone. This activation allows the reaction to proceed smoothly under relatively mild thermal conditions compared to unactivated systems, ensuring that the sensitive tert-butyl group remains intact throughout the process. The choice of base, ranging from carbonates to hydroxides, plays a pivotal role in generating the reactive phenoxide species in situ, while the mixed solvent system ensures optimal solubility of both the organic substrates and the inorganic salts involved. Understanding these mechanistic details is vital for R&D teams aiming to replicate or further optimize this pathway for specific grade requirements in high-purity OLED material or polymer additive production.
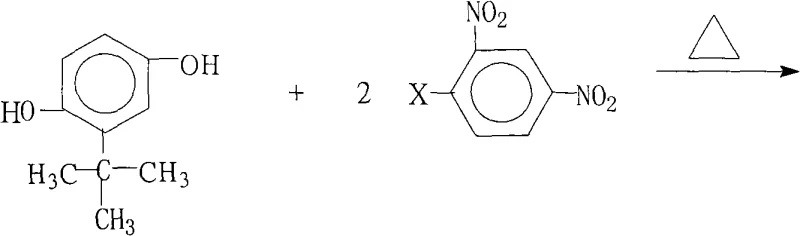
Furthermore, the meticulous control over reaction parameters described in the patent ensures exceptional impurity profiling, which is critical for the subsequent polymerization steps. The use of a water-separating reflux setup prevents the hydrolysis of the halogenated starting material, a common side reaction that can degrade yield and introduce phenolic impurities difficult to separate. The stoichiometry is carefully balanced with a slight excess of the halogenated component (molar ratio 1.0:2.0-2.2) to ensure complete consumption of the more valuable hydroquinone derivative, thereby maximizing resource utilization. Post-reaction processing involves concentrating the solution and inducing precipitation by adding water, a technique that leverages the differential solubility of the product to achieve high purity without the need for energy-intensive chromatographic purification. This level of control over the chemical environment demonstrates a sophisticated understanding of process chemistry, enabling the commercial scale-up of complex electronic intermediates with consistent batch-to-batch reproducibility.
How to Synthesize 1,4-Bis(2,4-Dinitrophenoxy)-2-Tert-Butylbenzene Efficiently
Implementing this synthesis requires precise adherence to the specified molar ratios and solvent compositions to guarantee the high yields and purity reported in the patent data. The process begins with the combination of 2-tert-butylhydroquinone and the selected 2,4-dinitrohalobenzene in a reactor equipped with a water separator, followed by the addition of the base and the binary solvent mixture. Operators must maintain the reaction temperature within the 80°C to 200°C range, monitoring the water collection to determine the reaction endpoint, which typically occurs between 6 to 18 hours depending on the specific halogen used. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in validating this route within their own pilot facilities.
- Mix 2-tert-butylhydroquinone and 2,4-dinitrohalobenzene in a molar ratio of 1.0: 2.0-2.2 with a salt-forming agent in a mixed solvent system.
- Heat the mixture to reflux (80°C-200°C) with water separation for 6 to 18 hours to drive the nucleophilic substitution to completion.
- Concentrate the reaction solution, cool, add water to precipitate the solid product, then filter, wash, and dry to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers transformative benefits that extend far beyond simple chemical transformation. The elimination of expensive transition metal catalysts and the ability to operate at atmospheric pressure significantly lower the capital expenditure required for reactor infrastructure, making it accessible for a wider range of manufacturing partners. Moreover, the robustness of the reaction conditions means that production schedules are less susceptible to delays caused by equipment maintenance or safety incidents related to high-pressure operations. This reliability is paramount for reducing lead time for high-purity monomers, ensuring that downstream polymer producers can maintain their own production timelines without interruption. The strategic value of this method lies in its ability to decouple production costs from volatile catalyst markets while providing a stable, scalable source of critical electronic materials.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally sound due to the avoidance of precious metal catalysts and the implementation of a closed-loop solvent recovery system. By utilizing common inorganic bases like potassium carbonate and recyclable organic solvents such as toluene and DMF, the variable cost per kilogram of product is drastically minimized compared to catalytic coupling methods. The high yield achieved minimizes raw material waste, ensuring that every gram of input contributes effectively to the final output, which translates into substantial cost savings over large production volumes. Additionally, the simplicity of the isolation procedure reduces labor hours and energy consumption associated with purification, further enhancing the overall cost efficiency of the manufacturing operation.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, specifically 2-tert-butylhydroquinone and various nitrohalobenzenes, are commodity chemicals with well-established global supply chains. This abundance ensures that production is not bottlenecked by the scarcity of exotic reagents, providing a secure foundation for long-term supply agreements. The flexibility to use different halogenated benzenes (fluoro, chloro, bromo, or iodo) allows procurement teams to pivot between suppliers based on price and availability without altering the core process, adding a layer of resilience against market fluctuations. This adaptability is crucial for maintaining continuous supply in the face of geopolitical or logistical disruptions that often impact the fine chemical sector.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns perfectly with modern green chemistry principles by generating minimal waste and avoiding the release of corrosive byproducts. The ability to recycle solvents repeatedly not only lowers costs but also significantly reduces the volume of hazardous waste requiring disposal, simplifying regulatory compliance and permitting processes. The atmospheric pressure operation eliminates the risks associated with high-pressure vessels, enhancing workplace safety and reducing insurance premiums. These factors combined make the process highly scalable, allowing manufacturers to ramp up production from pilot scale to hundreds of tons annually with confidence in meeting stringent environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and industry best practices. These insights are intended to clarify the operational feasibility and strategic benefits for stakeholders evaluating this technology for integration into their supply chains. Understanding these details is essential for making informed decisions about sourcing and production planning.
Q: What are the key advantages of this synthesis method for industrial production?
A: The method described in patent CN101279919A offers significant operational simplicity by conducting the reaction under atmospheric pressure without corrosive substances. It utilizes a recyclable solvent system and achieves high yields and purity, making it highly suitable for large-scale manufacturing of polyimide precursors.
Q: Which halogenated benzenes are compatible with this reaction pathway?
A: The process is versatile and accommodates various 2,4-dinitrohalobenzenes, including fluoro-, chloro-, bromo-, and iodo- derivatives. This flexibility allows manufacturers to optimize costs based on the availability and pricing of specific halogenated starting materials while maintaining high product quality.
Q: How does the solvent system contribute to reaction efficiency?
A: The use of a mixed solvent system comprising a water-insoluble organic solvent and a strong polar aprotic solvent facilitates effective water removal via azeotropic distillation. This drives the equilibrium towards product formation, ensuring high conversion rates and simplifying the subsequent isolation of the solid product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Bis(2,4-Dinitrophenoxy)-2-Tert-Butylbenzene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance intermediates play in the advancement of electronic and aerospace materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 1,4-bis(2,4-dinitrophenoxy)-2-tert-butylbenzene performs consistently in your polymerization processes. Our expertise in process optimization allows us to tailor production parameters to meet specific customer requirements, ensuring that you receive a material that is perfectly suited for your unique application needs.
We invite you to collaborate with us to explore how this advanced synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic goals for high-quality electronic material sourcing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
