Advanced Synthesis of 3,3'-Dinitro-4,4'-bis(4-nitrophenoxy)biphenyl for High-Performance Polyimide Manufacturing
Advanced Synthesis of 3,3'-Dinitro-4,4'-bis(4-nitrophenoxy)biphenyl for High-Performance Polyimide Manufacturing
The global demand for high-performance aromatic polyimides continues to surge, driven by critical applications in liquid crystal displays (LCDs), aerospace composites, and flexible electronics. At the heart of this supply chain lies the efficient production of specialized monomers, specifically 3,3'-dinitro-4,4'-bis(4-nitrophenoxy)biphenyl. Patent CN101225047A introduces a robust and scalable preparation method that addresses historical bottlenecks in synthesizing this complex aromatic ether. By leveraging a precise nucleophilic aromatic substitution strategy, this technology enables the production of high-purity intermediates essential for next-generation functional materials. For R&D directors and procurement leaders, understanding this pathway is vital for securing a stable supply of electronic grade chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex diaryl ethers with multiple nitro substituents has presented significant challenges regarding reaction control and purification. Traditional approaches often suffer from incomplete conversion due to steric hindrance around the biphenyl core, leading to difficult-to-remove impurities that compromise the thermal properties of the final polyimide. Furthermore, many legacy processes require harsh conditions, such as high-pressure reactors or expensive phase-transfer catalysts, which escalate capital expenditure and operational risks. The lack of standardized, open-literature protocols for this specific dinitro-biphenyl derivative has previously forced manufacturers to rely on proprietary, often inefficient routes that generate substantial hazardous waste.
The Novel Approach
The methodology disclosed in the patent data revolutionizes this landscape by employing a straightforward reflux technique under atmospheric pressure. By utilizing a dual-solvent system comprising a water-insoluble organic solvent and a strongly polar aprotic solvent, the reaction achieves excellent homogeneity and kinetic energy for the substitution. This novel approach eliminates the need for specialized high-pressure equipment, drastically reducing infrastructure investment. Moreover, the integration of a water-separation mechanism during reflux drives the equilibrium forward, ensuring high conversion rates without the need for excessive reagent loading. This simplicity translates directly into a more reliable supply chain for high-purity electronic chemical intermediates.
Mechanistic Insights into Nucleophilic Aromatic Substitution
The core chemical transformation relies on a classic nucleophilic aromatic substitution (SnAr) mechanism, facilitated by the strong electron-withdrawing nature of the nitro groups. In this reaction, the phenoxide anion, generated in situ by the deprotonation of 3,3'-dinitro-4,4'-dihydroxybiphenyl using a carbonate or hydroxide base, acts as a potent nucleophile. It attacks the electron-deficient aromatic ring of the 4-halonitrobenzene at the position ortho or para to the nitro group. The presence of the nitro group stabilizes the negatively charged Meisenheimer complex intermediate, lowering the activation energy required for the displacement of the halide leaving group.
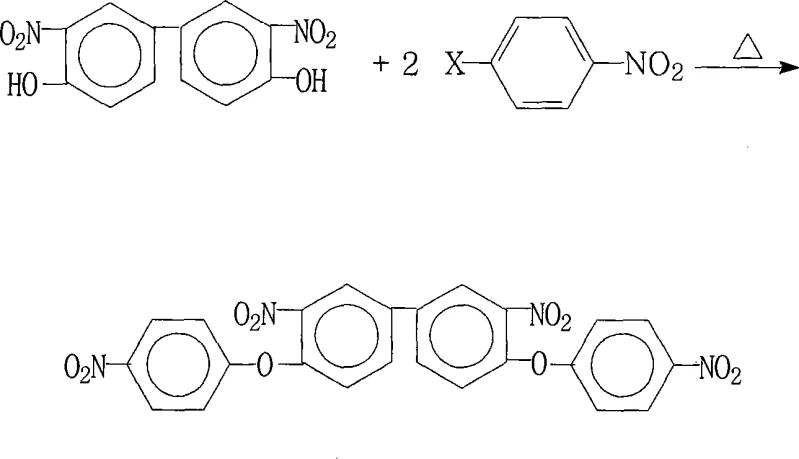
Impurity control is intrinsically managed through the selection of the leaving group and the reaction temperature window of 80°C to 200°C. The patent specifies that halogens such as fluorine, chlorine, bromine, or iodine can serve as the leaving group, allowing flexibility based on cost and reactivity profiles. Fluorine, being the most electronegative, typically offers the fastest reaction rates in SnAr processes due to its ability to stabilize the transition state, while chlorine offers a cost-effective balance. The rigorous control of the molar ratio between the dihydroxy precursor and the halonitrobenzene ensures that mono-substituted byproducts are minimized, resulting in a crude product with purity levels suitable for direct crystallization.
How to Synthesize 3,3'-Dinitro-4,4'-bis(4-nitrophenoxy)biphenyl Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for scaling this reaction from laboratory benchtop to industrial kiloton production. The process begins with the precise charging of reactants into a reactor equipped with a Dean-Stark trap or similar water-separation apparatus. The choice of base, ranging from potassium carbonate to sodium hydroxide, allows for fine-tuning the pH and reaction kinetics to match specific facility capabilities. Detailed standardized synthetic steps see the guide below for exact parameters regarding solvent ratios and thermal profiles.
- Mix 3,3'-dinitro-4,4'-dihydroxybiphenyl and 4-halonitrobenzene in a molar ratio of 1.00: 2.00-2.22 with a base catalyst.
- Heat the mixture in a dual-solvent system (water-insoluble + polar aprotic) under reflux with water separation for 4 to 18 hours.
- Concentrate the reaction, cool, add water to precipitate the solid, then filter, wash, and dry to obtain the final crystal product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of this patented process are profound, offering a pathway to significant cost optimization without compromising quality. The ability to operate under atmospheric pressure removes the safety liabilities and maintenance costs associated with autoclaves, while the use of commodity chemical feedstocks ensures long-term availability. The process design inherently supports a circular economy model within the plant, as the solvent system is engineered for easy recovery and reuse, minimizing raw material consumption and waste disposal fees.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and high-pressure vessels leads to a drastic simplification of the production line. By recovering and recycling the organic solvent mixture, which constitutes a major portion of the reaction mass, manufacturers can achieve substantial reductions in variable operating costs. This efficiency allows for competitive pricing strategies in the volatile electronic chemical market while maintaining healthy margins.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials like 4-chloronitrobenzene and common inorganic bases mitigates the risk of supply disruptions caused by niche reagent shortages. The robustness of the reaction conditions means that production can be maintained consistently across different geographic locations, ensuring a steady flow of critical intermediates to downstream polymer manufacturers who cannot afford downtime.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, primarily consisting of inorganic salts that are easier to treat than heavy metal residues. This environmental friendliness simplifies regulatory compliance and permitting for capacity expansion. The straightforward workup procedure involving water precipitation and filtration is easily adaptable to large-scale continuous or batch processing, facilitating rapid commercial scale-up of complex polymer additives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of adopting this technology for their specific production needs.
Q: What is the primary application of 3,3'-dinitro-4,4'-bis(4-nitrophenoxy)biphenyl?
A: This compound serves as a critical intermediate for synthesizing aromatic polyimide monomers, which are essential for high-performance materials in LCD displays, aerospace, and microelectronics due to their thermal stability.
Q: How does this patent improve upon traditional etherification methods?
A: The patented method utilizes a specific mixed solvent system and atmospheric pressure reflux, eliminating the need for high-pressure equipment while achieving yields over 90% and purity exceeding 99%.
Q: Can the solvents used in this process be recycled?
A: Yes, the process design explicitly allows for the concentration and recovery of organic solvents like toluene and DMF, enabling repeated reuse which significantly lowers operational costs and environmental impact.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3'-Dinitro-4,4'-bis(4-nitrophenoxy)biphenyl Supplier
As the electronics industry pushes the boundaries of material performance, having a partner with deep technical expertise in aromatic monomer synthesis is indispensable. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of 3,3'-dinitro-4,4'-bis(4-nitrophenoxy)biphenyl meets the exacting standards required for high-end polyimide applications.
We invite you to collaborate with us to optimize your material sourcing strategy and reduce your overall cost of goods. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and manufacturing goals immediately.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
