Advanced Synthesis of Polyimide Intermediates: Scaling High-Purity Electronic Materials for Industrial Applications
The rapid evolution of the optoelectronics and aerospace sectors has created an insatiable demand for high-performance polymers capable of withstanding extreme thermal and mechanical stress. At the heart of this material revolution lies the development of advanced aromatic polyimides, which serve as the backbone for next-generation liquid crystal displays (LCDs) and flexible circuitry. A pivotal breakthrough in this domain is documented in Chinese patent CN101279921A, which discloses a robust and scalable preparation method for 1,4-bis(2,6-dinitro-4-trifluoromethylphenoxy)-2-tert-butylbenzene. This specific chemical entity acts as a critical monomer intermediate, enabling the synthesis of highly branched polyimide systems that exhibit exceptional temperature resistance and comprehensive performance metrics. For R&D directors and procurement strategists alike, understanding the nuances of this synthesis is paramount, as it represents a significant leap forward in manufacturing efficiency and product purity. The patent outlines a process that not only simplifies operational complexity but also aligns with modern environmental standards by minimizing waste and facilitating solvent recovery. As the global supply chain for electronic chemicals tightens, mastering such proprietary synthetic routes becomes a competitive necessity for ensuring continuity in the production of high-value functional materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex diaryl ethers, particularly those intended for high-performance polymer applications, has been plagued by significant technical and economic hurdles. Traditional nucleophilic aromatic substitution reactions often necessitate the use of expensive phase-transfer catalysts or harsh reaction conditions that can degrade sensitive functional groups. Furthermore, conventional methodologies frequently struggle with incomplete conversion, leading to difficult purification processes and suboptimal yields that inflate the overall cost of goods sold. Many legacy processes operate under high pressure or require strictly anhydrous conditions that are difficult to maintain on a multi-ton scale, introducing substantial safety risks and equipment investment costs. The generation of stoichiometric amounts of salt byproducts and the inability to effectively recycle high-boiling polar aprotic solvents further exacerbate the environmental footprint, creating disposal challenges that conflict with increasingly stringent global regulations. These inefficiencies create bottlenecks in the supply chain, resulting in longer lead times and higher volatility in pricing for downstream manufacturers of polyimide films and coatings.
The Novel Approach
In stark contrast to these legacy challenges, the methodology described in patent CN101279921A introduces a streamlined and economically viable pathway that addresses the core inefficiencies of traditional etherification. By leveraging a specific molar ratio of 2-tert-butylhydroquinone to 2,6-dinitro-4-trifluoromethyl halobenzene ranging from 1.0:2.0 to 1.0:2.2, the process ensures complete consumption of the limiting reagent while minimizing side reactions. The innovation lies in the strategic use of a mixed solvent system combined with a heating reflux water separation technique, which continuously drives the equilibrium toward product formation without the need for exotic catalysts. This approach operates comfortably at atmospheric pressure within a temperature range of 80°C to 200°C, drastically reducing the capital expenditure required for specialized high-pressure reactors. Moreover, the protocol explicitly emphasizes the recoverability and recyclability of the organic solvent system, transforming what was once a waste stream into a reusable asset. This novel route not only delivers products with purity levels exceeding 99% but also establishes a foundation for sustainable industrial production that resonates with the cost-reduction goals of modern procurement teams.
Mechanistic Insights into Nucleophilic Aromatic Substitution
The chemical transformation at the core of this synthesis is a classic yet highly optimized example of nucleophilic aromatic substitution (SnAr), driven by the powerful electron-withdrawing effects of the substituents on the aromatic ring. In this reaction, the phenoxide anion, generated in situ from 2-tert-butylhydroquinone by the action of a base such as potassium carbonate or sodium hydroxide, acts as a potent nucleophile. It attacks the electron-deficient aromatic ring of the 2,6-dinitro-4-trifluoromethyl halobenzene, specifically targeting the carbon atom bearing the halogen leaving group. The presence of two nitro groups ortho to the leaving group, along with the strongly electron-withdrawing trifluoromethyl group at the para position, significantly lowers the energy of the transition state, thereby accelerating the reaction rate even under relatively mild thermal conditions. This electronic activation is crucial, as it allows the reaction to proceed efficiently without the need for transition metal catalysts, which are often sources of heavy metal contamination in electronic-grade materials. The mechanistic pathway involves the formation of a Meisenheimer complex intermediate, which subsequently collapses to expel the halide ion and restore aromaticity, resulting in the formation of the stable ether linkage.
Beyond the primary substitution mechanism, the control of impurities and side products is meticulously managed through the choice of reaction parameters and workup procedures. The use of a mixed solvent system comprising both water-insoluble organic solvents (like benzene or toluene) and strong polar aprotic solvents (such as NMP or DMF) plays a dual role: it solubilizes both the organic reactants and the inorganic salts while facilitating the azeotropic removal of water produced during the salt formation step. This continuous removal of water prevents the hydrolysis of the halobenzene starting material, a common side reaction that can severely impact yield and purity. Furthermore, the precise control of the base-to-substrate molar ratio ensures that the phenol is fully deprotonated without creating an overly basic environment that could promote decomposition of the sensitive nitro groups. The final crystallization step, induced by the addition of water to the concentrated reaction mass, leverages the differential solubility of the product versus unreacted starting materials and inorganic salts, effectively purifying the crude material in a single unit operation. 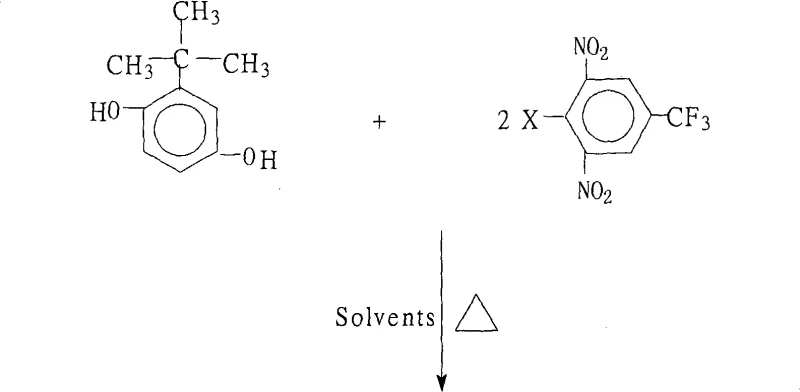
How to Synthesize 1,4-Bis(2,6-Dinitro-4-Trifluoromethylphenoxy)-2-Tert-Butylbenzene Efficiently
Executing this synthesis on an industrial scale requires strict adherence to the optimized parameters defined in the patent to ensure consistent quality and yield. The process begins with the careful charging of reactants into a reactor equipped with a water separator, followed by the controlled addition of the base and solvent mixture. Operators must monitor the reflux temperature closely to maintain the optimal kinetic window while ensuring efficient water removal. Once the reaction time of 6 to 18 hours is complete, the workup procedure involves concentrating the mixture to recover valuable solvents before inducing precipitation.
- Prepare the reaction mixture by combining 2-tert-butylhydroquinone and 2,6-dinitro-4-trifluoromethyl halobenzene in a molar ratio of 1.0: 2.0-2.2 with a salt-forming agent.
- Utilize a mixed solvent system comprising water-insoluble organic solvents and strong polar aprotic solvents, then heat to reflux for water separation over 6 to 18 hours.
- Concentrate the reaction solution, cool the system, precipitate the solid product with water, and purify via filtration, washing, and drying to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible strategic advantages that extend far beyond simple chemical yield. The most significant benefit lies in the drastic simplification of the manufacturing infrastructure; because the reaction proceeds at atmospheric pressure and avoids corrosive reagents, facilities can utilize standard glass-lined or stainless steel reactors without needing expensive high-pressure ratings or specialized corrosion-resistant alloys. This directly translates to lower capital depreciation costs and reduced maintenance overheads, allowing for more competitive pricing structures in the final electronic chemical market. Additionally, the ability to recover and recycle the organic solvent system multiple times significantly reduces the raw material consumption per kilogram of product, insulating the supply chain from volatility in solvent pricing and minimizing hazardous waste disposal fees. The robustness of the process also implies a higher reliability of supply, as the risk of batch failure due to sensitive catalytic poisoning or equipment malfunction is substantially mitigated compared to more complex transition-metal-catalyzed alternatives.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the ability to operate under atmospheric pressure removes significant cost drivers from the production budget. By avoiding the need for rigorous heavy metal scavenging steps typically required in pharmaceutical or electronic grade synthesis, the downstream processing costs are significantly lowered. Furthermore, the high atom economy and the potential for solvent recycling mean that the variable cost per unit is optimized, providing a buffer against raw material price fluctuations. This efficiency allows suppliers to offer more stable long-term contracts to downstream polyimide manufacturers, fostering stronger commercial partnerships.
- Enhanced Supply Chain Reliability: The reliance on readily available commodity chemicals such as carbonates, hydroxides, and common industrial solvents ensures that the supply chain is not vulnerable to the bottlenecks often associated with specialty reagents. The simplicity of the operation reduces the dependency on highly specialized labor, making it easier to scale production across different geographic locations to mitigate regional risks. The high purity achieved directly from crystallization reduces the need for complex chromatographic purification, shortening the production cycle time and enabling faster turnaround for urgent orders. This agility is critical for maintaining just-in-time inventory levels in the fast-paced electronics manufacturing sector.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with the water separation technique being easily adaptable from pilot plant to multi-ton commercial reactors. The reduction in three wastes (waste water, gas, and solids) aligns perfectly with modern environmental, social, and governance (ESG) mandates, reducing the regulatory burden on manufacturing sites. The ability to recycle solvents not only cuts costs but also minimizes the carbon footprint of the manufacturing process, a key metric for multinational corporations seeking sustainable suppliers. This environmental compatibility ensures long-term operational continuity without the risk of shutdowns due to non-compliance with evolving environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized polyimide intermediate. These insights are derived directly from the experimental data and claims presented in the underlying patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this material into their existing supply chains.
Q: What are the critical reaction conditions for synthesizing this polyimide intermediate?
A: The synthesis requires a mixed solvent system of water-insoluble organics (like toluene) and polar aprotic solvents (like DMF or NMP), heated to reflux between 80°C and 200°C for 6 to 18 hours to facilitate water separation and drive the equilibrium forward.
Q: Why is this specific intermediate important for the electronics industry?
A: This compound serves as a crucial precursor for highly branched aromatic polyimides, which offer superior thermal stability and mechanical strength required for advanced LCD displays and aerospace applications.
Q: How does this method improve upon traditional etherification processes?
A: The patented method operates at atmospheric pressure without corrosive reagents, allows for efficient solvent recovery and recycling, and achieves high yields (over 90% in optimized examples) with minimal waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Bis(2,6-Dinitro-4-Trifluoromethylphenoxy)-2-Tert-Butylbenzene Supplier
As the demand for high-performance polyimides continues to surge in the LCD and semiconductor industries, securing a reliable source for critical intermediates like 1,4-bis(2,6-dinitro-4-trifluoromethylphenoxy)-2-tert-butylbenzene is essential for maintaining production schedules. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to meet the rigorous demands of global clients. Our state-of-the-art facilities are equipped with stringent purity specifications and rigorous QC labs to ensure that every batch meets the exacting standards required for electronic applications. We understand that consistency is key in polymer synthesis, and our dedicated technical team works closely with customers to validate route feasibility and optimize process parameters for maximum efficiency.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By partnering with our technical procurement team, you can access specific COA data and comprehensive route feasibility assessments that demonstrate the viability of this synthesis for your operations. Whether you are looking to diversify your supplier base or optimize your current manufacturing costs, our expertise in fine chemical intermediates provides the stability and quality assurance your business needs to thrive in a competitive market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
