Advanced C-O Coupling Strategy for High-Purity Antiviral Luteolin Dimers
Introduction to Next-Generation Antiviral Flavonoid Synthesis
The global demand for potent antiviral agents has intensified the search for novel compounds derived from natural products, specifically flavonoids known for their broad-spectrum biological activities. Patent CN111995608A introduces a groundbreaking methodology for the semi-synthetic modification of luteolin and quercetin, two prominent flavonoids found in Grifola frondosa. This technology addresses the critical limitation of natural extraction, which often yields insufficient quantities of high-value dimers, by employing a directed chemical synthesis strategy. The core innovation lies in the sequential application of free radical bromination and copper-catalyzed C-O coupling, enabling the precise construction of luteolin-quercetin and luteolin-luteolin biflavonoid architectures. For pharmaceutical developers, this represents a significant leap forward, transforming low-abundance natural metabolites into accessible, high-purity intermediates suitable for drug discovery pipelines targeting viral proteases like SARS-CoV Mpro.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, obtaining complex biflavonoids such as luteolin-quercetin dimers relied heavily on direct isolation from plant sources or non-selective oxidative coupling. These conventional approaches suffer from severe drawbacks, primarily the extremely low natural abundance of these specific dimeric structures, which makes extraction economically unviable for commercial scale-up. Furthermore, direct oxidative coupling often lacks regioselectivity, resulting in a complex mixture of isomers that are notoriously difficult to separate, thereby drastically reducing the overall yield and purity of the final product. The reliance on chromatographic purification for isomer separation not only inflates production costs but also introduces solvent residues that are unacceptable for pharmaceutical grade materials. Consequently, the supply chain for these high-value antiviral candidates has historically been fragmented and unreliable, hindering their progression through clinical development stages.
The Novel Approach
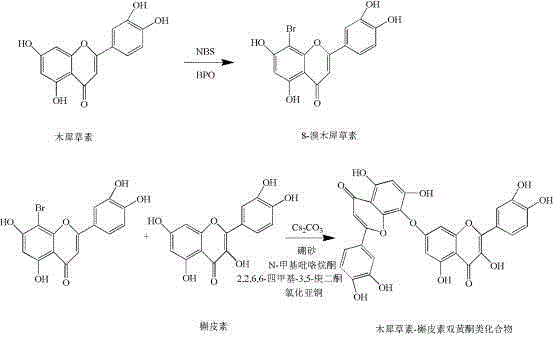
The methodology disclosed in the patent circumvents these historical bottlenecks by utilizing a rational, step-wise synthetic route that ensures high regioselectivity and operational simplicity. The process initiates with the selective bromination of luteolin at the C8 position using N-bromosuccinimide (NBS) initiated by dibenzoyl peroxide (BPO), creating a reactive handle for subsequent coupling. Crucially, the invention employs borax as a protecting agent to form boric acid complexes with the ortho-dihydroxyl groups of both the brominated intermediate and the coupling partner (quercetin or luteolin). This protection strategy masks reactive sites that would otherwise lead to polymerization or incorrect linkage, directing the reaction exclusively towards the desired C-O bond formation. By integrating this protection-deprotection logic with a copper-catalyzed coupling system, the process achieves a streamlined workflow that eliminates the need for complex isomer separation, directly yielding the target antiviral compounds with purity levels exceeding 85%.
Mechanistic Insights into Copper-Catalyzed C-O Coupling
The heart of this synthetic breakthrough is the copper-catalyzed Ullmann-type C-O coupling reaction, which facilitates the formation of the ether linkage between the two flavonoid units. In this mechanism, the cuprous chloride or cuprous iodide catalyst, coordinated with ligands such as 2,2,6,6-tetramethyl-3,5-heptanedione or 1,10-phenanthroline, activates the carbon-bromine bond of the 8-bromoluteolin boric acid complex. Simultaneously, the cesium carbonate base deprotonates the phenolic hydroxyl group of the partner flavonoid complex, generating a nucleophilic phenoxide species. The coordination of the borax protecting group is pivotal here; it not only prevents self-coupling or poly-substitution but also enhances the solubility of the flavonoid substrates in the polar aprotic solvent N-methylpyrrolidone (NMP). This homogeneous reaction environment allows for efficient collision frequency between the catalytic center and the substrates, driving the coupling reaction to completion at moderate temperatures ranging from 60°C to 120°C under nitrogen protection.
Following the coupling event, the reaction mixture undergoes a straightforward workup involving centrifugation and acidification with hydrochloric acid. This acidic treatment serves a dual purpose: it quenches the excess base and, more importantly, hydrolyzes the borate ester bonds, regenerating the free phenolic hydroxyl groups essential for the biological activity of the final dimer. The regeneration of the catechol moiety is critical, as these groups are responsible for the hydrogen bonding interactions with the viral protease active site. The robustness of this mechanism is evidenced by its applicability to both heterodimers (luteolin-quercetin) and homodimers (luteolin-luteolin), demonstrating the versatility of the borax protection strategy in managing the complex reactivity profiles of polyphenolic natural products.
How to Synthesize Antiviral Luteolin Derivatives Efficiently
The synthesis protocol outlined in the patent offers a reproducible pathway for manufacturing these high-value intermediates, balancing reaction efficiency with ease of operation. The process is divided into three distinct phases: the generation of the electrophilic brominated precursor, the formation of the nucleophilic borax complexes, and the final catalytic coupling and deprotection. Each step utilizes commercially available reagents and standard laboratory equipment, such as three-neck flasks and rotary evaporators, ensuring that the transition from bench-scale optimization to pilot plant production is seamless. The detailed stoichiometry, such as maintaining a molar ratio of luteolin to NBS between 1:1 and 1:1.5, is critical for minimizing poly-brominated byproducts while maximizing the conversion to the desired 8-bromo intermediate. For a comprehensive breakdown of the specific temperature profiles, reaction times, and workup procedures required to achieve optimal yields, please refer to the standardized synthesis guide below.
- Perform radical bromination of luteolin using NBS and BPO in acetone to generate 8-bromoluteolin.
- Protect the hydroxyl groups of 8-bromoluteolin and quercetin using borax in aqueous solution to form boric acid complexes.
- Execute C-O coupling using cuprous chloride, cesium carbonate, and a ligand in NMP, followed by acid deprotection.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented technology offers substantial strategic advantages by decoupling the production of high-value antiviral intermediates from the volatility of agricultural harvests. Traditional sourcing of rare biflavonoids is subject to seasonal variations, geographical constraints, and the ecological impact of mass plant extraction. In contrast, this semi-synthetic approach relies on luteolin and quercetin, which are abundant, commodity-grade natural products available from multiple global suppliers. This shift to a synthesis-based model significantly enhances supply chain reliability, ensuring consistent availability of raw materials regardless of external environmental factors. Furthermore, the elimination of extensive chromatographic purification steps, replaced by simpler crystallization and washing protocols, drastically reduces the consumption of high-purity solvents and silica gel, leading to a leaner and more cost-effective manufacturing process.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive inorganic reagents like borax and cesium carbonate, which replace costly specialty protecting groups often used in fine chemical synthesis. By avoiding the need for transition metal scavengers typically required for palladium-catalyzed reactions, the downstream purification burden is significantly lightened, resulting in lower operational expenditures. The high atom economy of the coupling reaction, combined with the ability to recover and recycle solvents like acetone and NMP, contributes to a substantial reduction in the overall cost of goods sold (COGS) for these complex molecules.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures that production timelines are predictable and controllable, unlike extraction processes which can be delayed by crop failures or logistics issues. The use of stable intermediates, such as the freeze-dried boric acid complexes, allows for inventory buffering, enabling manufacturers to respond rapidly to surges in demand for antiviral drug development. This stability is crucial for long-term supply agreements with pharmaceutical partners who require guaranteed continuity of supply for their clinical trial materials and eventual commercial launches.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial large-scale production in mind, utilizing reaction conditions that are easily managed in standard stainless steel reactors. The avoidance of hazardous heavy metal catalysts and the use of aqueous workups for the borax complex formation align with green chemistry principles, simplifying waste treatment and regulatory compliance. This environmental compatibility reduces the risk of production shutdowns due to regulatory scrutiny and positions the manufacturer as a sustainable partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these antiviral luteolin derivatives. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these nuances is essential for R&D teams evaluating the feasibility of incorporating these dimers into their drug discovery programs and for procurement officers assessing the long-term viability of the supply source.
Q: What is the primary advantage of using borax protection in this synthesis?
A: Borax selectively protects the ortho-dihydroxyl groups on the flavonoid rings, preventing unwanted side reactions during the copper-catalyzed coupling and ensuring regioselectivity at the C8 position.
Q: How does the antiviral activity of the dimer compare to the monomer?
A: The synthesized luteolin-quercetin and luteolin-luteolin dimers exhibit significantly lower IC50 values against SARS-CoV Mpro compared to the individual luteolin or quercetin monomers, indicating enhanced potency.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights the use of inexpensive reagents like borax and cesium carbonate, along with standard solvent systems like acetone and NMP, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Antiviral Luteolin Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN111995608A for the development of next-generation antiviral therapeutics. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate this laboratory-scale innovation into a robust, commercial-grade manufacturing process. Our facilities are equipped to handle the specific requirements of flavonoid chemistry, including the management of moisture-sensitive coupling reactions and the precise control of crystallization parameters needed to achieve stringent purity specifications. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from preclinical evaluation to full-scale market supply without interruption.
We invite pharmaceutical companies and research institutions to collaborate with us to optimize this synthesis further and secure a stable supply of high-purity luteolin-quercetin and luteolin-luteolin compounds. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized process can reduce your overall development costs. We encourage you to contact us to request specific COA data from our pilot batches and to discuss route feasibility assessments for your unique antiviral drug candidates, ensuring a partnership built on scientific rigor and commercial reliability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
