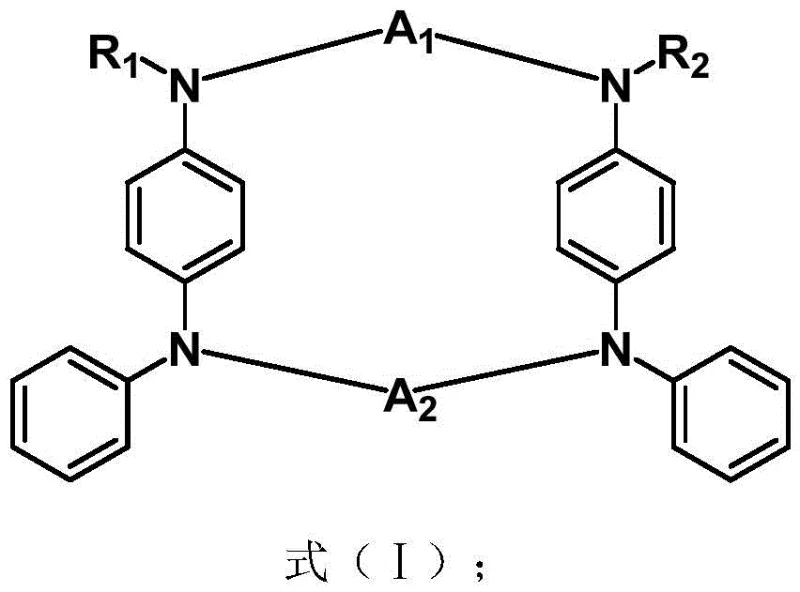
Advanced Synthesis of Luminescent Macrocyclic TADF Compounds for Next-Gen OLED Displays
The rapid evolution of organic light-emitting diode (OLED) technology demands emissive materials that combine high efficiency with long-term stability, driving significant interest in Thermally Activated Delayed Fluorescence (TADF) compounds. Patent CN113816968B introduces a groundbreaking class of luminescent macrocyclic TADF compounds that address the critical limitations of traditional linear emitters. By utilizing an aniline derivative as a robust donor group coupled with a diverse range of interchangeable electron acceptor structures, this invention provides a versatile platform for constructing high-performance organic optoelectronic materials. The core innovation lies in the macrocyclic architecture, which enforces structural rigidity and optimizes the spatial arrangement of donor and acceptor units to maximize photoluminescence quantum yield. For procurement and R&D teams seeking a reliable OLED material supplier, this technology represents a significant leap forward in metal-free emissive design, offering a pathway to reduce dependency on scarce heavy metals while maintaining superior device performance metrics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional OLED emitters have largely relied on fluorescent or phosphorescent materials, each carrying distinct disadvantages that hinder next-generation display manufacturing. Fluorescent materials suffer from inherently low internal quantum efficiency, theoretically capped at 25% because they cannot utilize triplet excitons, leading to higher power consumption and heat generation in commercial devices. While phosphorescent materials utilizing heavy metals like iridium or platinum can achieve nearly 100% internal quantum efficiency, they introduce severe supply chain vulnerabilities due to the scarcity and high cost of these precious metals. Furthermore, phosphorescent emitters often exhibit significant efficiency roll-off at high brightness levels and suffer from stability issues over prolonged operation. Existing linear TADF oligomers attempt to bridge this gap but often lack the structural rigidity required to prevent non-radiative decay, resulting in broader emission spectra and lower color purity compared to the rigid macrocyclic systems proposed in this patent.
The Novel Approach
The novel approach detailed in patent CN113816968B overcomes these hurdles by engineering a continuous D-A-D-A conjugated macrocyclic system that fundamentally alters the photophysical landscape. By cyclizing the donor-acceptor chain, the molecule achieves a constrained conformation that minimizes vibrational relaxation and enhances radiative decay rates. This macrocyclic topology allows for the formation of a large dihedral angle between the donor and acceptor moieties, which is critical for reducing the overlap between the Highest Occupied Molecular Orbital (HOMO) and the Lowest Unoccupied Molecular Orbital (LUMO). This reduction in orbital overlap directly decreases the singlet-triplet energy gap (Delta EST), facilitating rapid Reverse Intersystem Crossing (RISC) and enabling the harvesting of both singlet and triplet excitons without the need for expensive heavy metal catalysts. Additionally, the introduction of ketone-based electron acceptors induces molecular vibrations that trigger an Aggregation-Induced Emission (AIE) effect, effectively suppressing exciton annihilation in the solid state and ensuring high luminous intensity even in concentrated film formulations.
Mechanistic Insights into Macrocyclic TADF Design and RISC Promotion
The exceptional performance of these luminescent macrocyclic compounds stems from a meticulously engineered electronic structure that balances charge transfer characteristics with steric constraints. In the D-A-D-A continuous conjugated macrocycle, the spatial separation of frontier molecular orbitals is maximized through the large dihedral angle formed between the electron-rich aniline donor and the electron-deficient acceptor units. This geometric arrangement is not merely structural but functional, as it drastically lowers the exchange energy between the singlet and triplet states. A small Delta EST value, typically less than 0.3 eV as demonstrated in the patent examples, is the prerequisite for efficient thermal up-conversion of triplet excitons to the singlet state. This process, known as Reverse Intersystem Crossing (RISC), allows the material to harvest the 75% of excitons that are normally lost in fluorescent systems, theoretically achieving 100% internal quantum efficiency. The macrocyclic ring strain further contributes to this effect by locking the molecule into a conformation that prevents planarization, thereby maintaining the charge-transfer state character essential for TADF activity.
Beyond the electronic tuning, the patent highlights a sophisticated mechanism for controlling impurities and enhancing solid-state emission through the Aggregation-Induced Emission (AIE) effect. In conventional solution-processable emitters, high concentrations often lead to aggregation-caused quenching (ACQ), where close packing of molecules dissipates energy as heat rather than light. However, the specific incorporation of ketone carbonyl groups within the acceptor units of this macrocyclic framework induces restricted intramolecular motion upon aggregation. This restriction blocks non-radiative decay channels, forcing the excited state energy to be released as fluorescence. Consequently, these compounds exhibit stronger fluorescence emission in the high-concentration aggregated state found in OLED thin films compared to dilute solutions. This intrinsic AIE characteristic simplifies the device fabrication process by reducing the need for complex host-guest doping strategies to prevent quenching, thereby streamlining the manufacturing workflow for high-purity OLED material production.
How to Synthesize Luminescent Macrocyclic TADF Compounds Efficiently
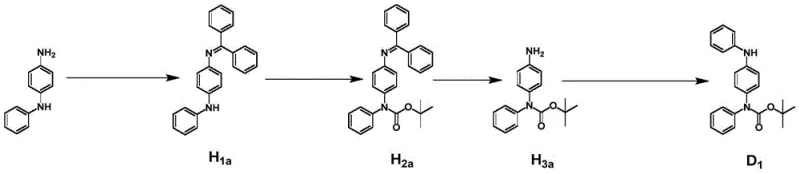
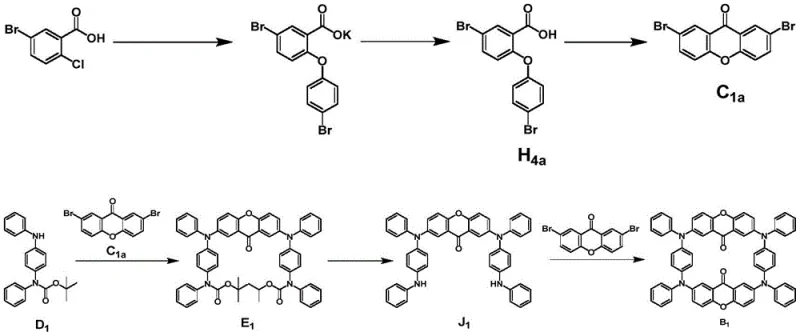
The synthesis of these advanced emitters follows a modular strategy that prioritizes yield and purity through a series of well-defined coupling and cyclization steps. The process begins with the preparation of a protected amine donor, which is crucial for preventing unwanted side reactions during the subsequent nucleophilic substitutions. Following donor synthesis, the route proceeds through the preparation of di-halo acceptor cores, which serve as the electrophilic partners in the final macrocyclization. The key step involves a double Buchwald-Hartwig amination reaction, utilizing palladium catalysis to close the macrocyclic ring. This approach allows for the systematic variation of the acceptor unit to tune emission colors while maintaining a consistent synthetic backbone. Detailed standardized synthesis steps for the donor and the final macrocyclization are provided in the guide below, illustrating the feasibility of scaling this chemistry for industrial production.
- Synthesize the protected donor intermediate by reacting N-phenyl-p-phenylenediamine with benzophenone and protecting groups under basic catalysis.
- Prepare the di-halo acceptor core (e.g., xanthone or thioxanthone derivatives) through cyclization and halogenation reactions.
- Perform the final macrocyclization by coupling the deprotected donor with the acceptor using a palladium catalyst and phosphine ligand in toluene.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this macrocyclic TADF technology offers transformative benefits that extend beyond mere performance metrics to fundamental cost and reliability improvements. The elimination of heavy metals such as iridium and platinum from the emissive layer removes a significant bottleneck in the raw material supply chain, mitigating the risks associated with geopolitical instability and price volatility in the precious metals market. Furthermore, the synthetic route relies on commercially available starting materials like aniline derivatives and substituted benzoic acids, which are produced at scale for various chemical industries, ensuring a stable and cost-effective supply base. The modular nature of the synthesis also means that multiple variations of the emitter can be produced using the same core donor intermediate, maximizing asset utilization and reducing the inventory burden for manufacturers seeking a reliable OLED material supplier.
- Cost Reduction in Manufacturing: The transition to metal-free TADF emitters drastically simplifies the cost structure of OLED panel production. By removing the need for expensive organometallic complexes, the raw material cost per gram of emitter is significantly lowered. Additionally, the synthetic pathway described in the patent utilizes standard organic transformation reactions such as nucleophilic substitution and palladium-catalyzed coupling, which are well-understood and easily optimized in large-scale reactors. The absence of stringent requirements for ultra-low metal residue levels, which are critical for phosphorescent materials to prevent device degradation, further reduces the cost of purification and quality control processes. This cumulative effect results in substantial cost savings in electronic chemical manufacturing, making high-efficiency displays more economically viable for mass-market applications.
- Enhanced Supply Chain Reliability: Dependence on a single source for complex phosphorescent dopants poses a significant risk to display manufacturers. This patent platform diversifies the supply chain by enabling the synthesis of emitters from commodity chemicals that are widely available from multiple global vendors. The robustness of the synthetic route, which tolerates a variety of substituents on the acceptor core without requiring completely new process development, ensures continuity of supply even if specific precursor availability fluctuates. Moreover, the stability of the macrocyclic structure implies a longer shelf life for the raw materials, reducing waste and spoilage during storage and transportation. This resilience is critical for maintaining uninterrupted production schedules in the fast-paced consumer electronics sector.
- Scalability and Environmental Compliance: Scaling the production of complex organic molecules often encounters hurdles related to reaction exotherms and purification bottlenecks. The described macrocyclization process operates at moderate temperatures (around 120°C) in common solvents like toluene, which are compatible with existing large-scale chemical infrastructure. The use of heterogeneous catalysts or easily removable homogeneous catalysts in the donor synthesis step simplifies the workup procedure, minimizing solvent usage and waste generation. From an environmental perspective, the shift away from toxic heavy metals aligns with increasingly stringent global regulations on hazardous substances in electronics (such as RoHS and REACH). This proactive compliance reduces the regulatory burden on manufacturers and facilitates smoother market entry for new display products utilizing these high-purity OLED materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this macrocyclic TADF technology in industrial settings. These insights are derived directly from the experimental data and claims within patent CN113816968B, providing a factual basis for evaluating the material's suitability for your specific application needs. Understanding these details is essential for R&D teams planning device integration and procurement officers assessing long-term supply viability.
Q: What is the primary advantage of macrocyclic TADF compounds over linear analogues?
A: Macrocyclic structures enforce rigidity and reduce non-radiative decay pathways. The specific D-A-D-A conjugation in this patent creates a large dihedral angle, minimizing HOMO-LUMO overlap and facilitating efficient Reverse Intersystem Crossing (RISC) without heavy metals.
Q: How does the synthesis route ensure high purity for OLED applications?
A: The process utilizes a strategic protection-deprotection strategy for the amine donor, preventing side reactions during the initial coupling steps. Final purification via silica gel chromatography ensures the removal of palladium residues and oligomeric byproducts.
Q: Can the emission color be tuned using this platform technology?
A: Yes, the patent describes a modular approach where the electron acceptor (A) unit can be varied (e.g., xanthone, thioxanthone, triazine derivatives) while keeping the aniline-based donor constant, allowing for precise tuning of the emission wavelength and photophysical properties.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable TADF Compound Supplier
The technological potential of luminescent macrocyclic TADF compounds is immense, offering a clear path toward high-efficiency, metal-free OLED displays that meet the rigorous demands of modern consumer electronics. As a premier CDMO partner, NINGBO INNO PHARMCHEM possesses the specialized expertise required to translate these complex laboratory-scale syntheses into robust commercial manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot batches to full-scale manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of TADF compound meets the exacting standards required for high-performance optoelectronic applications.
We invite you to collaborate with us to optimize your supply chain and reduce your overall material costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and device architecture. By leveraging our process optimization capabilities, we can help you secure a stable supply of high-quality emitters while minimizing your total cost of ownership. Please contact us today to request specific COA data and route feasibility assessments for your next-generation display projects.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
