Advanced Synthesis of Air-Stable Perylene Diimides for Next-Gen Organic Electronics
Advanced Synthesis of Air-Stable Perylene Diimides for Next-Gen Organic Electronics
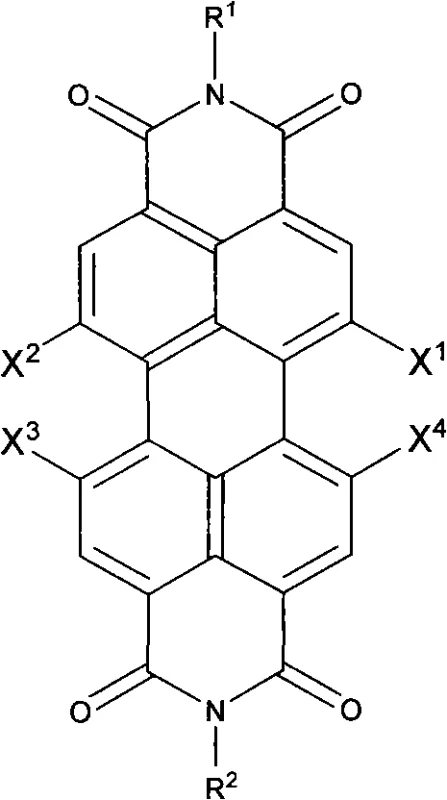
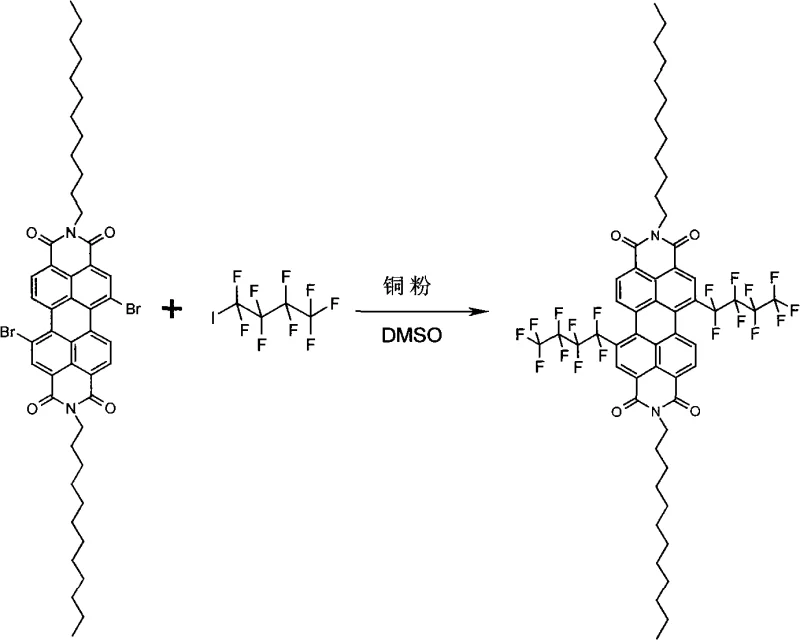
The rapid evolution of organic electronics demands materials that combine high charge carrier mobility with exceptional environmental stability. Patent CN101353349A introduces a groundbreaking methodology for synthesizing perylene-3,4:9,10-tetracarboxylic acid diimides substituted by perfluoroalkyl chains. These compounds represent a significant leap forward in the development of n-type organic semiconductors, addressing the historical instability of electron-transport materials in ambient conditions. By utilizing a robust copper-catalyzed coupling strategy, this technology enables the direct functionalization of the perylene core with fluorinated chains, resulting in materials with superior thermal and photochemical stability. For R&D directors and procurement specialists in the electronic chemicals sector, this synthesis route offers a reliable pathway to producing high-purity intermediates essential for organic field-effect transistors (OFETs), solar cells, and luminescent devices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the modification of perylene diimides to achieve n-type character has been fraught with synthetic challenges and performance compromises. Conventional approaches often rely on introducing electron-withdrawing groups through multi-step sequences that require harsh reaction conditions, expensive reagents, or sensitive organometallic catalysts that are difficult to remove from the final product. Furthermore, many existing fluorination methods result in incomplete substitution or produce mixtures of isomers that are notoriously difficult to separate, leading to batch-to-batch variability in electronic performance. The reliance on precious metal catalysts in some legacy processes also introduces significant cost burdens and supply chain risks, particularly when scaling to commercial quantities. Additionally, without the specific perfluoroalkyl architecture described in recent innovations, many perylene derivatives suffer from rapid degradation upon exposure to oxygen and moisture, limiting their practical utility in unencapsulated devices.
The Novel Approach
The methodology outlined in the patent data presents a streamlined, single-pot solution that drastically simplifies the manufacturing landscape for these advanced materials. By employing inexpensive copper powder as a heterogeneous catalyst in high-boiling polar aprotic solvents like DMSO or NMP, the process eliminates the need for complex ligand systems or air-sensitive reagents. This direct coupling of iodoperfluoroalkanes with halogenated perylene precursors proceeds efficiently at temperatures between 90°C and 150°C, ensuring high conversion rates while maintaining the integrity of the sensitive perylene core. The versatility of this approach allows for precise tuning of the material properties by varying the length of the perfluoroalkyl chain or the imide substituents, providing a modular platform for material optimization. This robustness translates directly into operational efficiency, reducing the technical barriers for commercial scale-up of complex organic semiconductors.
Mechanistic Insights into Copper-Catalyzed Perfluoroalkylation
The core of this technological advancement lies in the mechanistic efficiency of the copper-mediated radical coupling process. Under inert atmosphere conditions, the copper powder activates the carbon-iodine bond of the iodoperfluoroalkane, generating a perfluoroalkyl radical species that is highly reactive towards the electron-deficient perylene aromatic system. This radical mechanism bypasses the steric hindrance issues often encountered in nucleophilic substitutions on crowded aromatic cores, allowing for the successful introduction of bulky fluorinated chains at the 1, 6, 7, or 12 positions. The use of polar solvents facilitates the stabilization of intermediate species and ensures homogeneous heat transfer throughout the reaction mixture, which is critical for maintaining consistent reaction kinetics over the extended 24-hour reaction period. Understanding this mechanism is vital for process chemists aiming to optimize reaction parameters for specific derivative targets.
From an impurity control perspective, the simplicity of the reagent system contributes to a cleaner crude product profile. Since the catalyst is a solid metal powder, it can be easily separated from the reaction mixture via filtration, minimizing the risk of metal contamination in the final electronic grade material. The high selectivity of the radical addition minimizes the formation of side products such as poly-fluorinated adducts or reduced perylene species, which are common pitfalls in less controlled fluorination reactions. Furthermore, the stability of the resulting carbon-fluorine bonds ensures that the product does not undergo defluorination during the workup or purification stages, such as silica gel column chromatography. This inherent chemical stability simplifies the downstream processing requirements, reducing solvent consumption and waste generation associated with extensive purification protocols.
How to Synthesize Perfluoroalkyl-Perylene Diimides Efficiently
Implementing this synthesis route requires careful attention to temperature control and reagent stoichiometry to maximize yield and purity. The process begins with the activation of the copper catalyst in the chosen solvent, followed by the sequential addition of the fluorinating agent and the perylene substrate. This ordered addition is crucial to prevent the premature decomposition of the iodoperfluoroalkane or the formation of homocoupling byproducts. Detailed standardized operating procedures regarding the specific molar ratios of copper to substrate and the precise heating ramps are essential for reproducibility. For a comprehensive breakdown of the experimental conditions and workup procedures validated across multiple examples, please refer to the technical guide below.
- Prepare the catalyst mixture by adding copper powder to a high-boiling polar aprotic solvent such as DMSO or NMP under inert gas protection.
- Heat the mixture to 90-150°C and introduce the iodoperfluoroalkane reagent, allowing it to react for 30-60 minutes to activate the catalyst surface.
- Add the halogenated perylene-3,4: 9,10-tetracarboxylic diimide substrate and maintain heating for 24 hours to complete the perfluoroalkylation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this copper-catalyzed synthesis offers tangible economic and logistical benefits over traditional noble-metal catalyzed routes. The shift from expensive palladium or platinum catalysts to abundant copper powder represents a fundamental reduction in raw material costs, which is amplified when producing at metric ton scales. Moreover, the use of commodity solvents like DMSO and DMF, which are readily available in bulk quantities globally, mitigates supply chain disruptions associated with specialized or regulated reagents. The robustness of the reaction conditions, which tolerate a range of temperatures and do not require ultra-low temperature cryogenic cooling, further reduces energy consumption and infrastructure requirements in the manufacturing plant.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts removes the need for costly metal scavenging steps and reduces the overall bill of materials significantly. By utilizing a heterogeneous catalyst system, the process avoids the complex ligand synthesis and purification steps often required for homogeneous catalysis, streamlining the production workflow. The high isolated yields reported, ranging from 60% to 90%, ensure that raw material utilization is maximized, minimizing waste disposal costs and improving the overall atom economy of the process. These factors combine to create a highly cost-competitive manufacturing profile for high-value electronic intermediates.
- Enhanced Supply Chain Reliability: The starting materials, including various iodoperfluoroalkanes and halogenated perylene diimides, are commercially available or can be synthesized via established literature methods, ensuring a stable supply base. The process does not rely on proprietary or single-source reagents that could create bottlenecks, allowing for flexible sourcing strategies. Additionally, the ambient stability of the final perfluoroalkyl-substituted products simplifies storage and transportation logistics, as they do not require specialized cold chain management or rigorous exclusion of air during warehousing, unlike many other organic semiconductor precursors.
- Scalability and Environmental Compliance: The reaction operates in standard glass-lined or stainless steel reactors without the need for specialized high-pressure equipment, facilitating easy scale-up from laboratory to pilot and commercial production. The simplified workup procedure involving extraction and filtration reduces the volume of aqueous waste streams compared to processes requiring extensive aqueous quenching of reactive organometallics. Furthermore, the ability to recover and potentially recycle the copper catalyst aligns with green chemistry principles, supporting corporate sustainability goals and reducing the environmental footprint of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these specialized organic materials. The answers are derived directly from the experimental data and structural analysis provided in the patent documentation, ensuring accuracy and relevance for industry stakeholders. Understanding these nuances is critical for integrating these materials into existing product development pipelines and assessing their fit for specific electronic applications.
Q: What represents the key advantage of perfluoroalkyl substitution on perylene cores?
A: The introduction of perfluoroalkyl chains significantly lowers the LUMO energy level, enhancing electron affinity and making the material a stable n-type semiconductor resistant to oxidation in air.
Q: Is the copper powder catalyst reusable in this synthesis protocol?
A: While the patent describes the use of copper powder as a heterogeneous catalyst, standard industrial practices often allow for the recovery and regeneration of metal catalysts to further reduce production costs.
Q: What is the typical yield range for this perfluoroalkylation process?
A: According to the technical data, the purified yield for these complex perylene derivatives typically ranges between 60% and 90%, indicating a highly efficient transformation suitable for scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Perylene Diimides Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance organic semiconductors play in the future of display and energy technologies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from R&D to market is seamless and efficient. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of perylene diimide meets the exacting standards required for organic field-effect transistors and photovoltaic applications. Our commitment to quality assurance means you receive materials with consistent electronic properties, batch after batch.
We invite you to collaborate with us to optimize your supply chain for next-generation electronic materials. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your innovation goals. Let us be your partner in bringing stable, high-efficiency organic electronics to the global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
