Advanced Synthesis of 2,2-Bis[3-Nitro-4-(2,4-Dinitrophenoxy)Phenyl]Propane for High-Performance Polyimides
The chemical industry's relentless pursuit of advanced materials for aerospace and microelectronics has spotlighted the critical role of specialized aromatic intermediates. Patent CN101234972A introduces a robust preparation method for 2,2-bis[3-nitro-4-(2,4-dinitrophenoxy)phenyl]propane, a pivotal precursor for synthesizing highly branched aromatic polyimides. These polyimides are renowned for their exceptional thermal stability, mechanical strength, and electrical insulation properties, making them indispensable for liquid crystal displays (LCDs) and high-performance composite materials. The disclosed technology addresses the historical lack of efficient synthetic routes for this complex molecule, offering a pathway that combines high yield with operational simplicity. By leveraging a specific nucleophilic aromatic substitution strategy, this method ensures the production of high-purity intermediates essential for next-generation electronic applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of highly nitrated aromatic ethers presents significant challenges due to the sensitivity of nitro groups and the steric hindrance associated with bulky substituents. Conventional approaches often suffer from incomplete conversions, leading to complex impurity profiles that are difficult and costly to purify. Furthermore, many existing methods rely on harsh reaction conditions or expensive catalysts that can degrade the sensitive nitro functionalities, resulting in lower overall yields and compromised product quality. The management of waste streams in these older processes is also problematic, often involving toxic solvents that cannot be easily recovered, thereby increasing both the environmental footprint and the operational costs for manufacturers. These limitations have historically constrained the widespread adoption of such advanced polyimide precursors in cost-sensitive industrial applications.
The Novel Approach
The methodology described in the patent overcomes these hurdles through a carefully optimized nucleophilic substitution reaction that balances reactivity with selectivity. By employing a mixed solvent system comprising water-insoluble organic solvents and strong polar aprotic solvents, the process enhances the solubility of reactants while facilitating the removal of water generated during the reaction. This azeotropic water separation drives the equilibrium towards product formation without requiring excessive temperatures that could decompose the nitro groups. The use of readily available inorganic bases as salt-forming agents further simplifies the procedure, eliminating the need for sophisticated catalytic systems. This novel approach not only streamlines the synthesis but also ensures that the final product meets the stringent purity requirements necessary for high-end electronic material manufacturing.
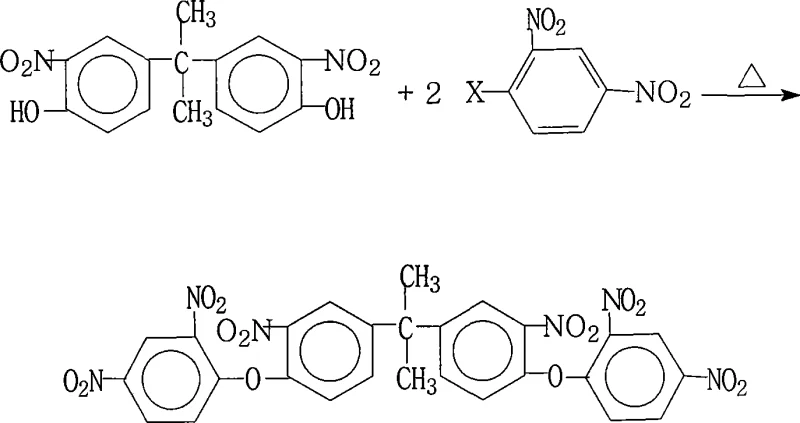
Mechanistic Insights into Nucleophilic Aromatic Substitution
The core of this synthesis lies in the nucleophilic aromatic substitution (SnAr) mechanism, where the phenoxide anion attacks the electron-deficient aromatic ring of the 2,4-dinitrohalobenzene. The presence of two strongly electron-withdrawing nitro groups ortho and para to the leaving halogen atom significantly activates the ring towards nucleophilic attack, lowering the activation energy for the reaction. The choice of halogen (fluorine, chlorine, bromine, or iodine) allows for tuning the reaction kinetics, with fluorine often providing superior rates due to its high electronegativity despite being a poorer leaving group in aliphatic systems. The base deprotonates the hydroxyl groups of the bisphenol derivative, generating the reactive nucleophile in situ, which then displaces the halide to form the stable ether linkage. This mechanistic pathway is highly selective, minimizing side reactions such as hydrolysis or reduction of the nitro groups, which are common pitfalls in less controlled environments.
Impurity control is intrinsically built into this mechanism through the precise control of stoichiometry and reaction conditions. By maintaining a molar ratio of the halobenzene to the bisphenol between 2.0 and 2.2, the process ensures complete conversion of the hydroxyl groups while minimizing the presence of unreacted starting materials or mono-substituted intermediates. The mixed solvent system plays a crucial role here, as the polar aprotic component stabilizes the transition state and the ionic intermediates, while the water-insoluble component facilitates the physical separation of the byproduct salts and water. Post-reaction workup involving water precipitation effectively isolates the product from soluble inorganic salts and residual solvents, yielding a crude solid that is already of high purity. This inherent cleanliness of the reaction profile reduces the burden on downstream purification steps, ensuring a consistent and reliable supply of the intermediate for polymerization.
How to Synthesize 2,2-Bis[3-Nitro-4-(2,4-Dinitrophenoxy)Phenyl]Propane Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing this valuable intermediate with high efficiency and reproducibility. The process begins with the precise weighing and mixing of 2,2-bis(3-nitro-4-hydroxyphenyl)propane and the chosen 2,4-dinitrohalobenzene in the presence of a carbonate or hydroxide base. The reaction is conducted in a reactor equipped for reflux and water separation, typically using a Dean-Stark trap or similar apparatus to continuously remove the water formed during the etherification. Maintaining the temperature within the specified range of 80°C to 200°C is critical to ensure optimal reaction kinetics without thermal degradation. Following the reaction period of 6 to 18 hours, the mixture is concentrated to recover solvents, and the product is precipitated by the addition of water, followed by filtration and drying. Detailed standardized synthesis steps are provided in the guide below to ensure consistent results across different production scales.
- Mix 2,2-bis(3-nitro-4-hydroxyphenyl)propane and 2,4-dinitrohalobenzene in a molar ratio of 1.0: 2.0~2.2 with a salt-forming agent in a mixed organic solvent system.
- Heat the reaction mixture to reflux (80°C~200°C) with water separation for 6 to 18 hours to drive the nucleophilic substitution to completion.
- Concentrate the reaction solution, cool the system, add water to precipitate the solid product, then filter, wash, and dry to obtain high-purity crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers substantial strategic benefits regarding cost stability and operational reliability. The elimination of expensive transition metal catalysts and the reliance on commodity chemicals like carbonates and common organic solvents drastically reduces the raw material cost base. Furthermore, the ability to recover and recycle the organic solvent mixture multiple times significantly lowers the consumption of volatile organic compounds, translating directly into reduced waste disposal costs and improved environmental compliance. The simplicity of the equipment requirements, operating at atmospheric pressure without the need for specialized high-pressure vessels, minimizes capital expenditure and maintenance overheads. These factors collectively contribute to a more resilient supply chain capable of delivering high-quality intermediates at competitive price points.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing inexpensive inorganic bases and avoiding precious metal catalysts, which removes the need for complex and costly metal scavenging steps. The high atom economy of the substitution reaction ensures that the majority of the input mass is converted into the desired product, minimizing material waste. Additionally, the efficient recovery of solvents allows for their repeated use in subsequent batches, further driving down the variable costs associated with each production run. This economic efficiency makes the large-scale production of this polyimide precursor financially viable for mass-market electronic applications.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials, such as substituted phenols and dinitrohalobenzenes, mitigates the risk of supply disruptions often associated with exotic reagents. The robustness of the reaction conditions, which tolerate slight variations in temperature and time without compromising yield, ensures consistent production output even in diverse manufacturing environments. This reliability is crucial for maintaining continuous production schedules for downstream polymer manufacturers who depend on a steady flow of high-purity monomers. The simplified logistics of handling non-corrosive reagents also streamline transportation and storage requirements, enhancing overall supply chain agility.
- Scalability and Environmental Compliance: The method is inherently scalable, having been designed with industrial production in mind, allowing for seamless transition from pilot batches to multi-ton annual capacities. The reduction in hazardous waste generation, achieved through solvent recycling and the absence of heavy metals, aligns with increasingly stringent global environmental regulations. This eco-friendly profile not only reduces regulatory risks but also enhances the sustainability credentials of the final electronic products. The straightforward workup procedure involving simple filtration and washing facilitates rapid batch turnover, maximizing facility utilization and throughput.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized intermediate. These answers are derived directly from the patented technology to provide accurate and actionable insights for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this material into your specific manufacturing workflows. We encourage technical teams to review these points to fully appreciate the capabilities of this synthesis method.
Q: What are the optimal reaction conditions for this synthesis?
A: The patent specifies a temperature range of 80°C to 200°C under reflux with water separation for 6 to 18 hours, utilizing a mixed solvent system of water-insoluble organics and polar aprotic solvents.
Q: Which halogenated starting materials are suitable?
A: The process accommodates various 2,4-dinitrohalobenzenes, including fluoro-, chloro-, bromo-, and iodo- derivatives, offering flexibility in raw material sourcing.
Q: How is solvent waste managed in this process?
A: The method emphasizes solvent recovery and recycling, particularly the organic phases, which significantly reduces waste generation and environmental impact compared to traditional single-use solvent protocols.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2-Bis[3-Nitro-4-(2,4-Dinitrophenoxy)Phenyl]Propane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the fabrication of advanced polyimides for the electronics and aerospace sectors. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with precision and consistency. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of 2,2-bis[3-nitro-4-(2,4-dinitrophenoxy)phenyl]propane meets the exacting standards required for high-performance applications. Our commitment to quality assurance means that you can rely on us as a long-term partner for your complex chemical needs.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can benefit your specific projects. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of switching to this efficient manufacturing method. We are prepared to provide specific COA data and route feasibility assessments to support your R&D and sourcing decisions. Contact us today to secure a reliable supply of this vital intermediate and accelerate your development of next-generation electronic materials.
