Scalable Synthesis of Advanced Polyimide Intermediates for Electronic and Aerospace Applications
Scalable Synthesis of Advanced Polyimide Intermediates for Electronic and Aerospace Applications
The rapid evolution of high-performance materials in the aerospace and electronic sectors demands intermediates that offer exceptional thermal stability and mechanical strength. Patent CN101265190A introduces a robust and industrially viable method for preparing 2,2-bis[4-(2,6-dinitro-4-trifluoromethylphenoxy)phenyl]propane, a pivotal building block for next-generation aromatic polyimides. This specific chemical architecture enables the creation of highly branched polymer systems that surpass traditional linear polyimides in terms of temperature resistance and comprehensive performance metrics. For R&D directors and procurement strategists, understanding this synthesis route is critical, as it represents a shift towards more efficient, high-yield manufacturing processes that do not compromise on the stringent purity required for optical and electronic applications. The patent details a streamlined approach that leverages readily available raw materials to produce a complex molecular structure with remarkable precision.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations detailed in this patent, the synthesis of such highly functionalized aromatic ethers often suffered from significant inefficiencies and operational complexities. Traditional routes frequently relied on harsh reaction conditions that necessitated specialized corrosion-resistant equipment, driving up capital expenditure and maintenance costs for manufacturing facilities. Furthermore, older methodologies often struggled with incomplete conversions, leading to difficult purification steps and lower overall yields that negatively impacted the cost of goods sold. The presence of unreacted starting materials or side products could severely degrade the thermal and electrical properties of the final polyimide material, rendering it unsuitable for high-end applications like liquid crystal displays or semiconductor packaging. Additionally, many conventional processes generated substantial amounts of hazardous waste, creating environmental compliance burdens that modern supply chains are increasingly eager to avoid.
The Novel Approach
The methodology described in CN101265190A fundamentally addresses these historical pain points by optimizing the reaction parameters for nucleophilic aromatic substitution. By carefully selecting a mixed solvent system comprising water-insoluble organic solvents and strongly polar aprotic solvents, the process ensures excellent solubility of reactants while facilitating the removal of by-product water. This strategic use of azeotropic distillation drives the reaction equilibrium towards completion without requiring excessive pressure or exotic catalysts. The result is a process that operates under normal atmospheric pressure, drastically reducing safety risks and equipment investment. Moreover, the ability to recover and recycle the solvent system multiple times not only lowers raw material consumption but also minimizes the environmental footprint, making this a highly attractive option for sustainable chemical manufacturing. The simplicity of the work-up procedure, involving merely concentration, cooling, and precipitation, further enhances its appeal for large-scale production.
Mechanistic Insights into Nucleophilic Aromatic Substitution
The core chemical transformation in this synthesis is a classic nucleophilic aromatic substitution (SNAr) reaction, driven by the strong electron-withdrawing nature of the nitro groups on the halobenzene ring. In this mechanism, the phenoxide anion, generated in situ from 2,2-bis(4-hydroxyphenyl)propane and a base such as potassium carbonate, acts as a potent nucleophile. This anion attacks the electron-deficient aromatic ring of the 2,6-dinitro-4-trifluoromethylhalobenzene at the position ortho or para to the nitro groups, where the electron density is lowest. The presence of two nitro groups significantly activates the ring, lowering the activation energy required for the substitution of the halogen atom. This electronic activation is crucial for achieving high reaction rates at moderate temperatures, preventing the degradation of sensitive functional groups that might occur under more forcing conditions.
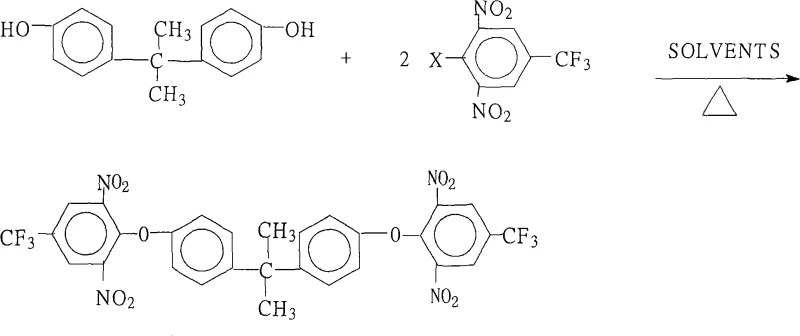
Following the initial attack, a Meisenheimer complex intermediate is formed, which subsequently eliminates the halide ion to restore aromaticity, yielding the desired diphenyl ether linkage. The choice of halogen (fluorine, chlorine, bromine, or iodine) allows for fine-tuning of the reaction kinetics, with fluorine often providing superior reactivity in SNAr reactions due to its high electronegativity despite being a poorer leaving group in other contexts. The trifluoromethyl group further enhances the electrophilicity of the ring and imparts valuable hydrophobic and thermal properties to the final polymer. Understanding this mechanistic pathway is vital for quality control, as it highlights the importance of maintaining anhydrous conditions during the reflux stage to prevent hydrolysis of the activated halobenzene, which would otherwise lead to yield loss and impurity formation.
How to Synthesize 2,2-bis[4-(2,6-dinitro-4-trifluoromethylphenoxy)phenyl]propane Efficiently
Executing this synthesis requires precise control over stoichiometry and thermal conditions to maximize the formation of the target bis-ether while minimizing oligomerization or side reactions. The patent outlines a versatile protocol that accommodates various halogenated starting materials and base combinations, offering flexibility for different production scales. Operators must ensure efficient water removal during the reflux period, typically using a Dean-Stark trap or similar apparatus, as the presence of water can inhibit the formation of the phenoxide nucleophile and reverse the reaction equilibrium.
- Prepare the reaction mixture by combining 2,2-bis(4-hydroxyphenyl)propane and 2,6-dinitro-4-trifluoromethylhalobenzene in a molar ratio of 1.0: 2.0 to 2.2 within a mixed solvent system containing a salt-forming agent.
- Heat the mixture to reflux temperatures between 80°C and 200°C for 6 to 18 hours, utilizing a water-separation apparatus to drive the equilibrium forward by removing generated water.
- Concentrate the reaction solution, cool the system, add water to precipitate the solid product, then filter, wash with hot water, and dry to obtain the final crystalline intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The reliance on commodity chemicals like Bisphenol A and common inorganic bases means that raw material sourcing is stable and less susceptible to the volatility often seen with specialized reagents. This stability translates directly into more predictable pricing models and reduced risk of supply disruptions, which is critical for maintaining continuous production schedules in high-volume manufacturing environments. Furthermore, the operational simplicity of the process reduces the need for highly specialized labor and complex automation, lowering the overall operational expenditure associated with the production of this high-value intermediate.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the ability to operate at atmospheric pressure significantly reduce both capital and operating costs. By avoiding the need for high-pressure reactors and the subsequent removal of trace metal contaminants, manufacturers can achieve substantial cost savings in downstream processing. The high atom economy of the reaction, coupled with the recyclability of the solvent system, ensures that raw material utilization is maximized, further driving down the unit cost of production. These efficiencies make the final polyimide materials more competitive in price-sensitive markets without sacrificing performance quality.
- Enhanced Supply Chain Reliability: The use of widely available starting materials mitigates the risk of bottlenecks that often plague supply chains dependent on niche reagents. Since the process does not require exotic catalysts or strictly controlled inert atmospheres beyond standard nitrogen purging, it can be implemented in a broader range of manufacturing facilities, increasing the potential supplier base. This diversification capability strengthens the resilience of the supply chain against geopolitical or logistical shocks, ensuring a steady flow of critical intermediates to downstream polymer producers.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial production in mind, allowing for seamless transition from pilot batches to multi-ton annual outputs. The reduced generation of hazardous waste and the ability to recycle solvents align with increasingly stringent global environmental regulations, reducing the liability and disposal costs associated with chemical manufacturing. This environmental compatibility not only safeguards the company's reputation but also future-proofs the production line against tightening emissions standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized intermediate. These insights are derived directly from the technical specifications and beneficial effects outlined in the patent documentation, providing a clear picture of what stakeholders can expect from this technology.
Q: What are the primary applications of this chemical intermediate?
A: This compound serves as a critical precursor for synthesizing highly branched aromatic polyimides, which are essential for high-temperature resistant materials in aerospace, microelectronics, and LCD display technologies.
Q: How does this process improve upon traditional synthesis methods?
A: The patented method eliminates the need for complex catalytic systems and operates under atmospheric pressure, significantly simplifying equipment requirements while achieving yields exceeding 96% and purity levels above 99%.
Q: Is the solvent system environmentally sustainable?
A: Yes, the process utilizes recoverable organic solvents that can be recycled repeatedly, minimizing waste generation and aligning with modern green chemistry principles for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2-bis[4-(2,6-dinitro-4-trifluoromethylphenoxy)phenyl]propane Supplier
As the demand for high-performance polyimides continues to surge in the electronics and aerospace industries, securing a dependable source of high-purity intermediates is paramount. NINGBO INNO PHARMCHEM stands ready to support your development and production needs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific solvent systems and thermal requirements of this synthesis, ensuring consistent batch-to-batch quality that meets stringent purity specifications. With our rigorous QC labs and commitment to process optimization, we deliver materials that enable our partners to achieve superior performance in their final polymer applications.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this efficient manufacturing method. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your unique volume requirements, ensuring a seamless transition to a more robust and cost-effective sourcing strategy.
