Scaling High-Purity 2,2-Bis[3-(4-Aminobenzamido)-4-Hydroxyphenyl]Hexafluoropropane for Industrial Applications
Scaling High-Purity 2,2-Bis[3-(4-Aminobenzamido)-4-Hydroxyphenyl]Hexafluoropropane for Industrial Applications
The development of advanced heat-resistant polymers relies heavily on the availability of specialized monomers with exceptional thermal stability and mechanical properties. Patent CN116143653A introduces a groundbreaking preparation method for 2,2-bis[3-(4-aminobenzamido)-4-hydroxyphenyl]hexafluoropropane, a critical building block for polyimide resins, polyesterimide resins, and high-performance epoxy curing agents. This technical insight report analyzes the novel synthetic route disclosed in the patent, highlighting its potential to revolutionize the supply chain for high-purity polymer intermediates. The method distinguishes itself through a sophisticated two-stage temperature control strategy during acylation and an efficient catalytic hydrogenation step, ensuring product purity exceeding 99% while maintaining operational simplicity suitable for large-scale manufacturing.
![Chemical structure of 2,2-bis[3-(4-aminobenzamido)-4-hydroxyphenyl]hexafluoropropane showing the central hexafluoroisopropylidene bridge and terminal amine groups](/insights/img/fluorinated-polyimide-monomer-synthesis-supplier-20260314034744-01.png)
For R&D directors and procurement specialists, understanding the nuances of this synthesis is vital for securing a reliable polyimide monomer supplier. The patent details a process that not only achieves high yields but also incorporates solvent recycling protocols, directly addressing the industry's growing demand for sustainable chemical manufacturing. By leveraging strong polar aprotic solvents and optimized molar ratios, the process mitigates common issues associated with fluorinated compound synthesis, such as poor solubility and difficult purification. This report will dissect the mechanistic advantages and commercial implications of adopting this technology for the commercial scale-up of complex polymer additives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fluorinated diamines and related monomers has been plagued by challenges regarding reaction control and impurity management. Conventional acylation methods often employ single-stage temperature profiles, which can lead to uncontrolled exotherms when reacting highly reactive acid chlorides with amino-hydroxy substrates. This lack of thermal regulation frequently results in the formation of oligomeric byproducts and incomplete conversions, necessitating extensive and costly downstream purification steps. Furthermore, traditional methods may struggle with the solubility of fluorinated intermediates, leading to heterogeneous reaction conditions that hinder mass transfer and reduce overall yield. The absence of standardized protocols for this specific hexafluoropropane derivative has previously forced manufacturers to rely on inefficient, multi-step routes that generate significant chemical waste and suffer from batch-to-batch inconsistency.
The Novel Approach
The methodology outlined in patent CN116143653A represents a significant paradigm shift by introducing a segmented temperature control strategy during the acylation phase. Instead of a static reaction condition, the process initiates at a low temperature range of 0-15°C to manage the initial reactivity, followed by a controlled ramp to 40-50°C to drive the reaction to completion. This dynamic approach effectively suppresses side reactions while ensuring full conversion of the starting material. Additionally, the use of specific strong polar aprotic solvents, potentially in mixtures like DMF and NMP, creates a homogeneous medium that enhances reaction kinetics. The subsequent catalytic hydrogenation step is equally refined, utilizing optimized pressure and temperature parameters to selectively reduce nitro groups without compromising the sensitive amide linkages, thereby delivering a product with superior structural integrity and purity.
Mechanistic Insights into Two-Stage Acylation and Catalytic Hydrogenation
The core of this synthesis lies in the precise execution of nucleophilic acyl substitution followed by selective reduction. In the first stage, 2,2-bis(3-amino-4-hydroxyphenyl)hexafluoropropane acts as a nucleophile, attacking the carbonyl carbon of p-nitrobenzoyl chloride. The patent specifies a molar ratio of 1:2.0 to 1:2.14, ensuring a slight excess of the acylating agent to drive the equilibrium towards the bis-amide intermediate. The initial low-temperature phase (0-15°C) is critical for controlling the rate of HCl evolution and preventing localized overheating, which could degrade the fluorinated backbone. As the reaction progresses, raising the temperature to 40-50°C provides the necessary activation energy to overcome steric hindrance around the bulky hexafluoroisopropylidene group, ensuring both amino groups are fully acylated.
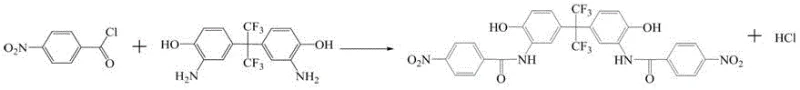
Following the isolation of the dinitro intermediate, the second mechanistic phase involves catalytic hydrogenation. This step utilizes transition metal catalysts, such as nickel-aluminum alloys or palladium on carbon, under hydrogen pressures ranging from 0.3 to 2.5 MPa. The mechanism involves the adsorption of hydrogen onto the catalyst surface, followed by the stepwise reduction of the nitro groups to primary amines. The choice of solvent in this step remains crucial; maintaining a polar environment ensures the intermediate stays in solution, allowing for efficient contact with the catalyst. The patent highlights that operating at temperatures between 50-70°C and pressures of 1.5-2.0 MPa offers the optimal balance between reaction rate and selectivity, preventing the hydrogenolysis of the newly formed amide bonds.

How to Synthesize 2,2-Bis[3-(4-Aminobenzamido)-4-Hydroxyphenyl]Hexafluoropropane Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters defined in the patent to achieve the reported purity levels above 99%. The process begins with the dissolution of the starting diamine in a selected polar aprotic solvent system, followed by the careful, dropwise addition of p-nitrobenzoyl chloride under inert atmosphere conditions. The temperature must be monitored continuously to maintain the specified two-stage profile, and the reaction mixture should be agitated sufficiently to ensure homogeneity throughout the acylation. Once the intermediate is formed and isolated via precipitation and washing, it is subjected to hydrogenation in a pressure reactor. The detailed标准化 synthesis steps见下方的指南 provide a comprehensive roadmap for laboratory and pilot-scale execution, ensuring reproducibility and safety.
- Perform a two-stage temperature-controlled acylation of 2,2-bis(3-amino-4-hydroxyphenyl)hexafluoropropane with p-nitrobenzoyl chloride in polar aprotic solvents.
- Isolate the intermediate nitro-compound via precipitation and washing to ensure high purity before reduction.
- Execute catalytic hydrogenation of the intermediate using nickel or palladium catalysts under controlled pressure and temperature to yield the final diamine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process offers tangible strategic benefits beyond mere technical feasibility. The ability to recycle solvents repeatedly without significant loss of efficiency translates directly into reduced raw material consumption and lower waste disposal costs. This aligns with global trends towards greener chemistry and helps manufacturers meet increasingly stringent environmental regulations. Furthermore, the high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, which simplifies the purification workflow and reduces the burden on quality control laboratories. These factors collectively contribute to a more resilient and cost-effective supply chain for high-performance polymer intermediates.
- Cost Reduction in Manufacturing: The elimination of complex purification steps and the ability to recover and reuse expensive polar aprotic solvents significantly lower the variable costs associated with production. By optimizing the molar ratios and reaction conditions, the process maximizes atom economy, ensuring that a higher proportion of raw materials end up in the final product rather than as waste. This efficiency gain allows for competitive pricing structures without compromising on the stringent quality standards required for electronic and aerospace grade materials.
- Enhanced Supply Chain Reliability: The robustness of the two-stage temperature control makes the process less susceptible to minor fluctuations in ambient conditions or raw material quality, leading to consistent batch outputs. This reliability is crucial for long-term supply contracts where consistency is paramount. Additionally, the use of commercially available catalysts and solvents reduces the risk of supply bottlenecks, ensuring that production schedules can be maintained even during periods of market volatility.
- Scalability and Environmental Compliance: The reaction conditions, particularly the moderate temperatures and pressures used in the hydrogenation step, are well-suited for scaling from kilogram to multi-ton quantities using standard industrial equipment. The process generates minimal hazardous byproducts, primarily HCl which can be neutralized or captured, and the solvent recovery system drastically reduces the volume of liquid waste. This facilitates easier permitting and compliance with environmental safety standards, accelerating the time to market for new polymer formulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of this fluorinated monomer. These insights are derived directly from the experimental data and claims presented in patent CN116143653A, providing a factual basis for decision-making. Understanding these details is essential for evaluating the feasibility of integrating this material into existing production lines or new product developments.
Q: What is the primary advantage of the two-stage temperature control in the acylation step?
A: The two-stage temperature profile, starting at 0-15°C and rising to 40-50°C, allows for precise control over the exothermic reaction rate, minimizing side reactions and significantly improving the purity of the intermediate compared to single-stage processes.
Q: Which solvents are recommended for maximizing yield and purity in this synthesis?
A: Strong polar aprotic solvents such as N,N-dimethylformamide (DMF), N,N-dimethylacetamide (DMAc), or N-methyl-2-pyrrolidone (NMP) are essential. A mixture of DMF and NMP has shown particularly favorable results for maintaining homogeneity and facilitating product isolation.
Q: How does this method address environmental concerns in fluorinated monomer production?
A: The process utilizes solvent recovery systems that allow for the repeated recycling of organic solvents, drastically reducing waste generation. Additionally, the high selectivity of the catalytic hydrogenation step minimizes the formation of hazardous byproducts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2-Bis[3-(4-Aminobenzamido)-4-Hydroxyphenyl]Hexafluoropropane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality monomers play in the performance of next-generation polymers. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2,2-bis[3-(4-aminobenzamido)-4-hydroxyphenyl]hexafluoropropane performs consistently in your final applications. Our infrastructure is designed to handle complex fluorinated chemistries with the utmost safety and efficiency.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced synthesis capabilities can support your R&D and production goals. Let us be your partner in driving innovation through superior chemical solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
