Advanced Synthesis of Guanidino Luteolin-Chromium(III) Complexes for High-Value Nutraceutical Applications
Advanced Synthesis of Guanidino Luteolin-Chromium(III) Complexes for High-Value Nutraceutical Applications
The global demand for bioactive metal-organic complexes in the nutraceutical and pharmaceutical sectors is driving significant innovation in synthetic methodology. Patent CN110305165B introduces a robust and scalable preparation method for Guanidino Luteolin-Chromium(III) complexes, representing a major advancement in functional food additive manufacturing. This technology leverages the inherent antioxidant properties of luteolin, a flavonoid derived from sources like Grifola frondosa, and enhances them through strategic guanidinylation and chromium coordination. For R&D directors and procurement specialists, this patent offers a clear pathway to producing high-value ingredients that combine the metabolic benefits of trivalent chromium with the anti-inflammatory and anticancer activities of modified flavonoids. The process is designed to be industrially viable, utilizing standard organic solvents and reagents to ensure consistent quality and supply continuity for downstream applications in health foods and therapeutic agents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the supplementation of chromium in dietary and pharmaceutical formulations has relied heavily on inorganic salts such as chromium chloride or picolinate. While effective to a degree, these conventional forms often suffer from poor bioavailability and potential toxicity issues when administered in high doses. Inorganic chromium ions can interact non-specifically with biological macromolecules, leading to oxidative stress rather than the desired metabolic regulation. Furthermore, simple flavonoid extracts, while popular, often lack the targeted cellular penetration required for potent therapeutic effects. The separation and purification of natural flavonoids can also be inconsistent, leading to batch-to-batch variability that complicates regulatory compliance for high-purity pharmaceutical intermediates. These limitations necessitate a more sophisticated chemical approach that stabilizes the metal ion within an organic framework, thereby mitigating toxicity while maximizing biological efficacy through synergistic molecular design.
The Novel Approach
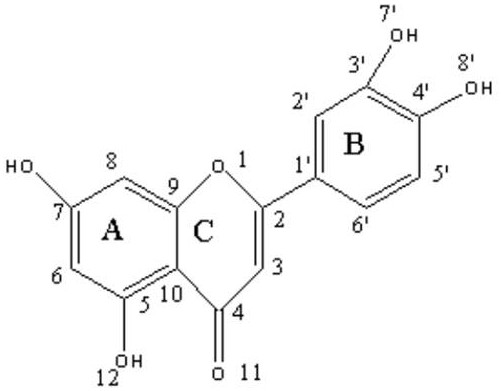
The methodology disclosed in CN110305165B overcomes these hurdles by creating a stable coordination complex where the chromium ion is chelated by a guanidino-modified luteolin ligand. This novel approach begins with the radical bromination of luteolin, introducing a reactive handle that allows for the subsequent attachment of the guanidino group. The guanidino moiety is critical as it imparts strong penetrating power across phospholipid bilayer cell membranes, significantly enhancing the intracellular delivery of the active compound. By converting inorganic chromium into this specific organic trace element form, the invention drastically reduces the risk of heavy metal poisoning while maintaining the essential role of chromium in sugar metabolism. This dual-modification strategy not only improves the stability of the alpha, beta-unsaturated carbonyl structure in the flavonoid C-ring but also creates a high-end product with substantial added value for the functional food and pharmaceutical industries.
Mechanistic Insights into Radical Bromination and Coordination Chemistry
The synthesis initiates with a precise radical substitution reaction where N-bromosuccinimide (NBS) acts as the brominating agent in the presence of dibenzoyl peroxide (BPO) as a radical initiator. This step targets the aromatic rings of the luteolin scaffold, specifically generating bromoluteolin intermediates under controlled thermal conditions ranging from 40°C to 80°C. The use of acetone as a solvent ensures optimal solubility for both the flavonoid substrate and the radical species, facilitating a homogeneous reaction environment. Following this activation, the process employs a nucleophilic substitution mechanism where free guanidine, generated in situ from guanidine hydrochloride and sodium isopropoxide, attacks the brominated position. This step is crucial for installing the bioactive guanidino group, which serves as the primary driver for enhanced cell membrane permeability. The reaction conditions, including the use of potassium iodide as a catalyst and sodium carbonate as a base, are finely tuned to maximize yield while minimizing side reactions that could compromise the integrity of the sensitive flavonoid backbone.
The final stage involves the coordination chemistry between the synthesized guanidino luteolin and chromium acetate. This complexation occurs in an ethanolic system where the pH is carefully adjusted to between 9 and 11 using sodium hydroxide. This alkaline environment is essential for deprotonating the phenolic hydroxyl groups on the luteolin A-ring and the oxygen atoms on the C-ring carbonyl, enabling them to act as effective ligands for the trivalent chromium ion. The resulting coordinate bonds stabilize the metal center, preventing premature oxidation and ensuring the structural integrity of the complex during storage and digestion. The formation of this complex is evidenced by shifts in infrared spectra, particularly in the vibration frequencies of the carbon-carben benzene ring skeleton and carbon-nitrogen bonds. This mechanistic precision ensures that the final product possesses the desired biological activities, including potent inhibition of pancreatic lipase and alpha-glucosidase, making it a superior candidate for managing metabolic disorders compared to its unmodified precursors.
How to Synthesize Guanidino Luteolin-Chromium(III) Complex Efficiently
The synthesis protocol outlined in the patent provides a reproducible framework for generating this high-value complex, emphasizing the importance of stoichiometric control and temperature management. The process is divided into distinct operational units: precursor activation, ligand synthesis, and metal coordination. Each step requires careful monitoring of molar ratios, such as maintaining a luteolin to NBS ratio between 1:1 and 1:1.5, to ensure complete conversion without excessive halogenation. The subsequent nucleophilic substitution demands anhydrous conditions and precise thermal control to drive the reaction to completion over a period of 6 to 14 hours. Finally, the complexation step relies on pH buffering and extended stirring at elevated temperatures to ensure thermodynamic stability of the chromium-ligand bond. For detailed operational parameters and safety guidelines, please refer to the standardized synthesis steps provided below.
- Perform radical bromination of Luteolin using N-bromosuccinimide (NBS) and dibenzoyl peroxide (BPO) in acetone to generate bromoluteolin intermediates.
- Generate free guanidine in situ by reacting guanidine hydrochloride with sodium isopropoxide in isopropanol under ice bath conditions.
- Execute nucleophilic substitution between bromoluteolin and free guanidine, followed by coordination with chromium acetate at controlled pH levels to finalize the complex.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthesis route offers significant strategic benefits for manufacturers of functional ingredients and pharmaceutical intermediates. The process utilizes widely available and cost-effective raw materials such as luteolin, acetone, ethanol, and chromium acetate, which mitigates the risk of supply chain disruptions associated with exotic or proprietary reagents. The simplicity of the reaction conditions, which do not require extreme pressures or cryogenic temperatures, allows for implementation in standard stainless steel reactors found in most fine chemical facilities. This compatibility with existing infrastructure significantly lowers the barrier to entry for commercial scale-up, enabling rapid transition from pilot plant to full-scale production. Furthermore, the use of common solvents simplifies the recovery and recycling processes, contributing to a more sustainable and environmentally compliant manufacturing operation that aligns with modern green chemistry principles.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of commodity chemicals like sodium carbonate and potassium iodide drive down the overall cost of goods sold. By avoiding complex enzymatic processes or high-pressure hydrogenation steps, the energy consumption per kilogram of product is substantially reduced. The straightforward workup procedure, involving simple distillation and washing, minimizes labor hours and processing time, leading to significant operational expenditure savings. Additionally, the high yield and selectivity of the reaction reduce the volume of waste generated, lowering disposal costs and improving the overall economic efficiency of the production line for cost reduction in functional food additive manufacturing.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable reagents ensures that production schedules are not vulnerable to the degradation issues often associated with sensitive biological catalysts. The modular nature of the synthesis allows for flexible batch sizing, enabling manufacturers to respond quickly to fluctuating market demands without compromising quality. Since the key starting material, luteolin, can be sourced from various botanical extracts or synthesized via established routes, there is no single point of failure in the raw material supply chain. This redundancy guarantees a continuous flow of high-purity guanidino luteolin-chromium complexes, securing the supply chain for downstream clients who require reliable pharmaceutical intermediate supplier partnerships.
- Scalability and Environmental Compliance: The process is inherently scalable, with reaction parameters that translate linearly from laboratory glassware to industrial reactors. The absence of hazardous heavy metal waste, as the chromium is sequestered in the final product, simplifies environmental permitting and wastewater treatment requirements. Solvent recovery systems can be easily integrated to capture acetone and ethanol, further reducing the environmental footprint and raw material costs. This alignment with strict environmental regulations makes the technology attractive for long-term investment, ensuring that commercial scale-up of complex flavonoid derivatives remains viable in increasingly regulated global markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Guanidino Luteolin-Chromium(III) complexes. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing a factual basis for decision-making. Understanding these nuances is critical for stakeholders evaluating the feasibility of integrating this technology into their existing product portfolios or R&D pipelines.
Q: What are the primary biological advantages of the Guanidino Luteolin-Chromium(III) complex over inorganic chromium?
A: The complex significantly reduces the toxicity associated with inorganic trace elements while enhancing bioavailability. The organic ligand structure facilitates better cell membrane penetration and metabolic integration compared to simple salt forms.
Q: How does the patented synthesis route ensure high purity for pharmaceutical applications?
A: The process utilizes specific purification steps including reduced pressure distillation, multiple deionized water washes, and freeze-drying. These steps effectively remove solvent residues and unreacted starting materials, ensuring stringent purity specifications.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights the simplicity of the production process and the use of commercially available reagents like acetone and ethanol. The mild reaction conditions (40-100°C) and standard workup procedures facilitate easy commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Guanidino Luteolin-Chromium(III) Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis route described in CN110305165B for the next generation of metabolic health products. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab concept to market reality is seamless. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of guanidino luteolin-chromium complex meets the highest international standards for safety and efficacy. We understand the complexities of coordinating metal-organic synthesis and have the technical expertise to optimize yields and minimize impurities, delivering a product that stands out in the competitive landscape of high-purity guanidino luteolin-chromium complex supplies.
We invite you to collaborate with us to leverage this innovative technology for your specific application needs. Whether you are developing a new dietary supplement or a specialized pharmaceutical formulation, our team can provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions about your supply chain strategy. By partnering with us, you gain access to a reliable source of advanced chemical intermediates that combine scientific rigor with commercial viability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
