Scalable Synthesis of 7-Methyl Ketone Flavonoids via Borax-Mediated Selective Alkylation
Scalable Synthesis of 7-Methyl Ketone Flavonoids via Borax-Mediated Selective Alkylation
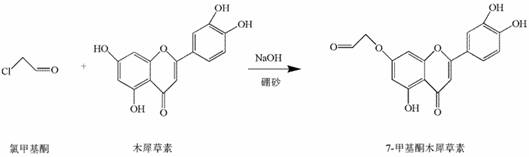
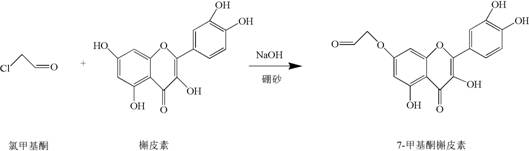
The pharmaceutical and fine chemical industries are constantly seeking efficient pathways to modify natural products for enhanced biological activity. A significant breakthrough in this domain is detailed in Chinese Patent CN111995606B, which discloses a novel preparation method for 7-methyl ketone derivatives of luteolin and quercetin. This technology addresses a critical gap in the functionalization of flavonoids, specifically targeting the 7-position hydroxyl group while preserving the pharmacologically active catechol moieties. By utilizing chloromethyl ketone as a lead compound in an aqueous alkaline environment, this method achieves regioselective nucleophilic substitution without the need for hazardous organic solvents or complex multi-step protection-deprotection sequences. The resulting 7-methyl ketone luteolin and 7-methyl ketone quercetin exhibit superior antiviral properties, particularly against SARS-CoV Mpro, making them high-value candidates for drug development. For procurement and R&D teams, this patent represents a shift towards greener, more cost-effective manufacturing of complex flavonoid intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for modifying flavonoids like luteolin and quercetin often suffer from poor regioselectivity and environmental inefficiency. These polyphenolic compounds possess multiple hydroxyl groups with similar reactivities, particularly the 5, 7, 3', and 4' positions. Conventional alkylation strategies typically require the use of strong bases in anhydrous organic solvents such as DMF or DMSO, accompanied by tedious protection group chemistry to mask the sensitive ortho-dihydroxyl groups on the B-ring. This not only increases the number of synthetic steps and overall production time but also generates substantial volumes of organic waste that require expensive disposal. Furthermore, the use of heavy metal catalysts or harsh reagents can introduce difficult-to-remove impurities, complicating the purification process and jeopardizing the stringent purity specifications required for pharmaceutical intermediates. The reliance on non-aqueous systems also poses significant safety risks during commercial scale-up, limiting the ability of suppliers to offer these derivatives at competitive prices.
The Novel Approach
The methodology outlined in CN111995606B offers a transformative solution by leveraging the unique coordination chemistry of borax in an aqueous medium. Instead of using toxic organic solvents, the reaction proceeds in deionized water with low-concentration sodium hydroxide serving as the catalyst. The key innovation lies in the use of borax as a reversible protecting agent; it selectively complexes with the cis-diol structures (specifically the 3',4'-dihydroxyl groups) on the flavonoid backbone. This complexation effectively deactivates these positions towards nucleophilic attack, thereby directing the chloromethyl ketone exclusively to the 7-hydroxyl position on the A-ring. This "one-pot" strategy eliminates the need for separate protection and deprotection steps, drastically simplifying the workflow. The reaction conditions are mild, typically ranging from 50°C to 90°C, and the workup involves a simple acidification with hydrochloric acid to precipitate the product, ensuring a robust and easily controllable process suitable for industrial application.
Mechanistic Insights into Borax-Mediated Regioselective Alkylation
The success of this synthesis hinges on the precise mechanistic interplay between the flavonoid substrate, the borate ion, and the alkylating agent. In the alkaline environment created by sodium hydroxide (pH 8-10), the phenolic hydroxyl groups of luteolin or quercetin are deprotonated to form phenoxide ions, which are potent nucleophiles. However, without control, alkylation would occur randomly at the most accessible hydroxyls. Borax (sodium tetraborate) dissociates in water to form borate ions, which have a high affinity for forming five- or six-membered cyclic esters with vicinal diols. In the case of luteolin and quercetin, the 3' and 4' hydroxyl groups on the B-ring form a stable borate complex. This steric and electronic shielding prevents the chloromethyl ketone from reacting at these positions. Consequently, the nucleophilic attack is funneled towards the 7-hydroxyl group, which remains free and highly reactive due to its position adjacent to the electron-withdrawing carbonyl at C4, enhancing its acidity and nucleophilicity under these specific conditions.
From an impurity control perspective, this mechanism offers distinct advantages for ensuring high-purity 7-methyl ketone flavonoids. Because the protection is dynamic and reversible, once the alkylation is complete, the subsequent acidification step with hydrochloric acid serves a dual purpose: it protonates the phenoxide to precipitate the product and simultaneously hydrolyzes the borate ester, regenerating the free 3',4'-dihydroxyl groups essential for biological activity. This avoids the formation of permanent byproducts associated with traditional silyl or benzyl protecting groups. The simplicity of the reaction matrix—primarily water, salt, and organic product—facilitates easy removal of inorganic residues through washing. For R&D directors, understanding this mechanism confirms that the process is inherently robust against over-alkylation, a common issue in flavonoid chemistry, thus ensuring a cleaner impurity profile and reducing the burden on downstream purification units.
How to Synthesize 7-Methyl Ketone Luteolin Efficiently
The synthesis protocol described in the patent provides a clear roadmap for producing these high-value derivatives with minimal equipment requirements. The process begins by charging a reactor with the flavonoid substrate (luteolin or quercetin), borax, and deionized water, followed by pH adjustment to create the reactive species. The addition of chloromethyl ketone initiates the substitution, which proceeds smoothly under moderate heating. The beauty of this method lies in its operational simplicity; there is no need for inert gas atmospheres or anhydrous conditions, which are typically costly to maintain. The reaction progress can be monitored easily, and upon completion, the product is isolated via a straightforward acid-base workup. For detailed standard operating procedures and specific molar ratios optimized for maximum yield, please refer to the technical guide below.
- Dissolve luteolin or quercetin with borax in deionized water and adjust pH to 8-10 using sodium hydroxide to form the protective borate complex.
- Add chloromethyl ketone to the mixture and maintain the reaction at 50-90°C for 3-7 hours to facilitate nucleophilic substitution at the 7-position.
- Acidify the reaction mixture with hydrochloric acid to precipitate the product, followed by washing and vacuum drying to obtain the pure derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this borax-mediated aqueous synthesis offers compelling economic and logistical benefits. The shift from organic solvents to water as the reaction medium fundamentally alters the cost structure of manufacturing. Water is not only inexpensive but also eliminates the need for solvent recovery systems and the associated energy consumption for distillation. Furthermore, the reagents involved—sodium hydroxide, borax, and hydrochloric acid—are commodity chemicals available globally in bulk quantities, insulating the supply chain from the volatility often seen with specialized organic reagents. This accessibility ensures a stable supply of raw materials, reducing the risk of production delays due to sourcing issues. The simplified workflow also translates to shorter batch cycles, allowing manufacturers to respond more agilely to market demand fluctuations without compromising on quality or safety standards.
- Cost Reduction in Manufacturing: The elimination of expensive organic solvents and complex protecting group reagents leads to substantial cost savings in raw material procurement. Additionally, the aqueous nature of the reaction significantly reduces waste disposal costs, as the effluent is primarily saline water rather than toxic organic waste requiring incineration. The process operates at atmospheric pressure and moderate temperatures, lowering energy consumption compared to high-pressure or cryogenic alternatives. These factors combine to lower the overall cost of goods sold (COGS), enabling more competitive pricing for the final pharmaceutical intermediates without sacrificing margin.
- Enhanced Supply Chain Reliability: By relying on widely available, non-hazardous raw materials, the supply chain becomes more resilient to disruptions. The robustness of the aqueous system means that production can be scaled across multiple facilities with standard stainless steel reactors, avoiding the need for specialized glass-lined or Hastelloy equipment often required for corrosive or sensitive organic reactions. This flexibility allows for diversified manufacturing locations, mitigating geopolitical or logistical risks. The consistent quality of the product, ensured by the high regioselectivity of the borax protection, reduces the incidence of batch failures, further stabilizing the supply of critical intermediates to downstream drug manufacturers.
- Scalability and Environmental Compliance: The process is inherently green, aligning with increasingly strict global environmental regulations. The absence of volatile organic compounds (VOCs) simplifies permitting and compliance reporting. Scaling from laboratory to commercial production (100 kgs to 100 MT) is straightforward because heat and mass transfer issues are minimized in the aqueous phase. The simple precipitation workup avoids the need for large-scale chromatography, which is often a bottleneck in scaling natural product derivatives. This ease of scale-up ensures that supply can be rapidly ramped up to meet clinical or commercial demand, providing a reliable long-term source for high-purity flavonoid derivatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 7-methyl ketone flavonoids based on the patented technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a transparent view of the method's capabilities and limitations. Understanding these details helps stakeholders make informed decisions about integrating these intermediates into their development pipelines.
Q: What is the role of borax in this synthesis?
A: Borax acts as a selective protecting agent by forming stable complexes with the ortho-dihydroxyl groups (such as the 3',4'-positions) on the flavonoid ring. This prevents alkylation at these sites, directing the chloromethyl ketone exclusively to the 7-hydroxyl group, thereby ensuring high regioselectivity without needing complex organic protecting groups.
Q: What yields can be expected from this process?
A: According to the patent data, optimized conditions (such as using 1.2mol/L NaOH at 70°C) can achieve yields of approximately 60% for 7-methyl ketone luteolin and nearly 59% for 7-methyl ketone quercetin, with product purity exceeding 85% after simple acid precipitation.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is highly scalable because it utilizes water as the primary solvent instead of toxic organic solvents. The use of commodity reagents like sodium hydroxide, borax, and hydrochloric acid, combined with a simple precipitation workup, significantly reduces waste treatment costs and operational hazards associated with large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Methyl Ketone Flavonoids Supplier
The technological potential of 7-methyl ketone luteolin and quercetin as antiviral and anti-inflammatory agents is immense, yet realizing this potential requires a manufacturing partner with deep expertise in process chemistry. NINGBO INNO PHARMCHEM stands ready to bridge the gap between patent innovation and commercial reality. As a seasoned CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 7-methyl ketone derivative delivered meets the highest international standards for pharmaceutical intermediates.
We invite you to explore how this innovative synthesis route can optimize your supply chain and reduce your overall project costs. Our technical team is prepared to conduct a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us collaborate to bring these high-value bioactive compounds from the laboratory to the market efficiently and sustainably.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
