Scalable Synthesis of Antiviral Phenyl Selenides: A Technical Breakdown for Industrial Production
Scalable Synthesis of Antiviral Phenyl Selenides: A Technical Breakdown for Industrial Production
The pharmaceutical industry is constantly seeking robust, scalable pathways to produce high-value bioactive intermediates, particularly those with demonstrated antiviral potential. Patent CN111995607A introduces a groundbreaking preparation method for antiviral phenyl selenides, specifically targeting the structural modification of naturally occurring flavonoids like luteolin and quercetin. This technology represents a significant leap forward in medicinal chemistry, offering a streamlined route to synthesize phenyl luteolin selenide and phenyl quercetin selenide with improved efficiency and reduced environmental burden. By leveraging a unique combination of electrophilic and nucleophilic substitution reactions in an aqueous environment, this process addresses critical pain points in the manufacturing of complex selenium-containing organic compounds. For R&D directors and supply chain leaders, understanding the nuances of this patent is essential for evaluating its potential to secure a reliable supply of next-generation antiviral intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the introduction of selenium moieties into complex flavonoid scaffolds has been fraught with significant technical and economic challenges that hinder large-scale adoption. Conventional methods often rely heavily on volatile organic solvents, stringent anhydrous conditions, and expensive transition metal catalysts that are difficult to remove to pharmaceutical-grade standards. These processes frequently suffer from poor atom economy, generating substantial hazardous waste streams that complicate disposal and increase the overall carbon footprint of the manufacturing operation. Furthermore, the sensitivity of flavonoid structures to oxidative degradation under harsh reaction conditions often leads to low yields and complex impurity profiles, necessitating costly and time-consuming purification steps such as preparative HPLC. The reliance on non-aqueous systems also poses safety risks related to flammability and toxicity, creating additional regulatory hurdles for commercial production facilities aiming for green chemistry compliance.
The Novel Approach
In stark contrast to these legacy techniques, the methodology disclosed in CN111995607A utilizes a remarkably simple yet effective aqueous-based strategy that fundamentally alters the cost and safety profile of phenyl selenide production. The core innovation lies in the use of borax as a reaction protective agent within a deionized water medium, which not only stabilizes the sensitive flavonoid substrates but also facilitates the nucleophilic substitution under mild alkaline conditions. This approach eliminates the need for expensive organic solvents in the key coupling step, drastically reducing raw material costs and simplifying the downstream workup procedure to a straightforward acid precipitation. By operating at moderate temperatures ranging from 60°C to 100°C and utilizing readily available reagents like sodium borohydride and dichloromethane, the process demonstrates exceptional feasibility for commercial scale-up. This shift towards aqueous chemistry aligns perfectly with modern sustainability goals while simultaneously enhancing the economic viability of producing high-purity antiviral intermediates.
Mechanistic Insights into Aqueous Nucleophilic Substitution
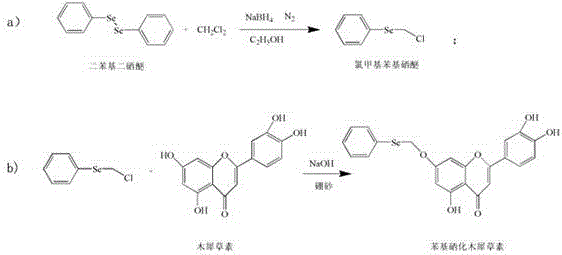
The chemical elegance of this synthesis is best understood through a detailed examination of the two-step reaction mechanism, which begins with the generation of a highly reactive selenium electrophile. In the first stage, diphenyl diselenide undergoes a reduction mediated by sodium borohydride in absolute ethanol under a nitrogen atmosphere, cleaving the Se-Se bond to generate a nucleophilic phenyl selenide anion in situ. This anion immediately attacks dichloromethane in an electrophilic substitution reaction to form the crucial intermediate, chloromethyl phenyl selenide, which serves as the selenium donor for the subsequent modification. The second stage involves the nucleophilic attack of the flavonoid substrate, where the specific role of borax becomes paramount; it is hypothesized that borax forms a reversible cyclic ester complex with the catechol hydroxyl groups on the B-ring of the flavonoid. This complexation effectively masks the 3',4'-dihydroxy functionality, preventing their participation in side reactions and potentially directing the alkylation to the more accessible 7-hydroxyl group on the A-ring, thereby ensuring regioselectivity and preserving the critical pharmacophore required for antiviral activity.
Following the coupling reaction, the integrity of the final product is maintained through a gentle deprotection and isolation protocol that avoids the use of aggressive reagents. The reaction mixture is treated with dilute hydrochloric acid, which serves a dual purpose: it neutralizes the alkaline medium and hydrolyzes the borax-flavonoid complex, releasing the free hydroxyl groups while simultaneously protonating the product to induce precipitation. This acid-induced crystallization is a highly efficient purification technique that allows for the separation of the target phenyl selenide from water-soluble impurities and inorganic salts without the need for chromatographic separation. The resulting crude product can be further purified by simple washing and vacuum drying to achieve purity levels exceeding 80%, as demonstrated in the patent examples. This mechanistic pathway highlights a sophisticated understanding of flavonoid chemistry, leveraging protective group strategies to achieve high selectivity in a green solvent system, which is a rare and valuable combination in industrial organic synthesis.
How to Synthesize Phenyl Selenized Flavonoids Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and minimize impurity formation, particularly during the intermediate generation and the pH-sensitive coupling steps. The process begins with the careful preparation of chloromethyl phenyl selenide, where temperature control during the exothermic reduction with sodium borohydride is critical to prevent the decomposition of the reactive selenium species. Once the intermediate is secured, the focus shifts to the aqueous coupling reactor, where the molar ratios of flavonoid to borax and the precise adjustment of pH to the 8-10 range are vital for optimal conversion rates. Operators must ensure that the sodium hydroxide concentration is maintained between 1.0 and 1.4 mol/L to provide sufficient nucleophilicity without degrading the flavonoid backbone.
- Reduce diphenyl diselenide with sodium borohydride in ethanol under nitrogen, then react with dichloromethane to form chloromethyl phenyl selenide.
- Dissolve luteolin or quercetin in water with borax, adjust pH to 8-10 using sodium hydroxide, and add the selenide intermediate.
- Heat the mixture to 60-100°C for 2-6 hours, then precipitate the final product by acidifying with hydrochloric acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers a compelling value proposition centered around cost stability, operational simplicity, and supply security. The transition from organic solvents to water as the primary reaction medium in the key coupling step represents a massive reduction in solvent procurement costs and waste disposal fees, directly impacting the bottom line of the manufacturing budget. Furthermore, the use of commodity chemicals such as diphenyl diselenide, borax, and sodium hydroxide ensures that the supply chain is not vulnerable to the volatility associated with specialized catalysts or exotic reagents, thereby guaranteeing long-term continuity of supply. The simplified workup procedure, which relies on precipitation rather than complex extraction or chromatography, significantly shortens the batch cycle time, allowing for higher throughput and faster response to market demand fluctuations without compromising product quality.
- Cost Reduction in Manufacturing: The elimination of expensive organic solvents and transition metal catalysts in the main coupling step leads to substantial cost savings in raw material expenditure. By utilizing water as the solvent and borax as a cheap protective agent, the process minimizes the need for costly solvent recovery systems and reduces the energy consumption associated with distillation. Additionally, the high selectivity of the reaction reduces the formation of byproducts, which in turn lowers the cost of goods sold by improving the overall yield and reducing the load on purification units. This economic efficiency makes the commercial production of phenyl selenized flavonoids viable for the first time, opening up new markets for antiviral therapeutics.
- Enhanced Supply Chain Reliability: The reliance on widely available, bulk chemical feedstocks ensures that production schedules are not disrupted by shortages of niche reagents. Diphenyl diselenide and flavonoids like luteolin and quercetin are commercially accessible from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the aqueous reaction conditions also means that the process is less sensitive to minor variations in raw material quality, providing a buffer against supply chain inconsistencies. This resilience is crucial for maintaining steady production volumes and meeting the rigorous delivery timelines expected by pharmaceutical partners.
- Scalability and Environmental Compliance: The inherent safety of using water as a solvent and operating at moderate temperatures simplifies the engineering requirements for scaling up from pilot to commercial production. There is no need for specialized explosion-proof equipment or complex ventilation systems required for volatile organic compounds, which accelerates the timeline for plant commissioning. Moreover, the reduced generation of hazardous organic waste aligns with increasingly strict environmental regulations, minimizing the risk of compliance penalties and enhancing the corporate sustainability profile. This green chemistry approach future-proofs the manufacturing asset against tightening regulatory frameworks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, providing clarity on its practical application and benefits. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What is the role of borax in the synthesis of phenyl selenized flavonoids?
A: Borax acts as a protective agent that likely complexes with the ortho-dihydroxy groups on the flavonoid B-ring, preventing unwanted side reactions and directing selectivity during the nucleophilic substitution.
Q: How does this method improve upon traditional selenium modification techniques?
A: This method utilizes an aqueous medium and mild conditions (60-100°C) instead of harsh organic solvents and high temperatures, significantly reducing environmental impact and operational costs while simplifying purification.
Q: What are the primary biological activities of the resulting phenyl selenides?
A: The synthesized phenyl luteolin selenide and phenyl quercetin selenide exhibit enhanced antiviral activity against SARS-CoV Mpro, along with anti-inflammatory and anticancer properties derived from the selenium modification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenyl Selenide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis route described in CN111995607A for the development of next-generation antiviral agents. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market-ready supply is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of selenium chemistry, including dedicated containment systems and rigorous QC labs capable of verifying stringent purity specifications for complex intermediates. We are committed to delivering high-quality phenyl luteolin selenide and phenyl quercetin selenide that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to optimize your supply chain and accelerate your drug development programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our implementation of this aqueous synthesis technology can drive down your overall manufacturing costs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a reliable, cost-effective source of these critical antiviral intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
